ASTM E2108-16
(Practice)Standard Practice for Calibration of the Electron Binding-Energy Scale of an X-Ray Photoelectron Spectrometer
Standard Practice for Calibration of the Electron Binding-Energy Scale of an X-Ray Photoelectron Spectrometer
SIGNIFICANCE AND USE
5.1 X-ray photoelectron spectroscopy is used extensively for the surface analysis of materials. Elements (with the exception of hydrogen and helium) are identified from comparisons of the binding energies determined from photoelectron spectra with tabulated values. Information on chemical state can be derived from the chemical shifts of measured photoelectron and Auger-electron features with respect to those measured for elemental solids.
5.2 Calibrations of the BE scales of XPS instruments are required for four principal reasons. First, meaningful comparison of BE measurements from two or more XPS instruments requires that the BE scales be calibrated, often with an uncertainty of about 0.1 eV to 0.2 eV. Second, identification of chemical state is based on measurement of chemical shifts of photoelectron and Auger-electron features, again with an uncertainty of typically about 0.1 eV to 0.2 eV; individual measurements, therefore, should be made and literature sources need to be available with comparable or better accuracies. Third, the availability of databases (3) of measured BEs for reliable identification of elements and determination of chemical states by computer software requires that published data and local measurements be made with uncertainties of about 0.1 eV to 0.2 eV. Finally, the growing adoption of quality management systems, such as, ISO 9001:2015, in many analytical laboratories has led to requirements that the measuring and test equipment be calibrated and that the relevant measurement uncertainties be known.
5.3 The actual uncertainty of a BE measurement depends on instrument properties and stability, measurement conditions, and the method of data analysis. This practice makes use of tolerance limits ±δ (chosen, for example, at the 95 % confidence level) that represent the maximum likely uncertainty of a BE measurement, associated with the instrument in a specified time interval following a calibration (ISO 15472:2010). A user should select a...
SCOPE
1.1 This practice describes a procedure for calibrating the electron binding-energy (BE) scale of an X-ray photoelectron spectrometer that is to be used for performing spectroscopic analysis of photoelectrons excited by unmonochromated aluminum or magnesium Kα X-rays or by monochromated aluminum Kα X-rays.
1.2 The calibration of the BE scale is recommended after the instrument is installed or modified in any substantive way. Additional checks and, if necessary, recalibrations are recommended at intervals chosen to ensure that BE measurements are statistically unlikely to be made with an uncertainty greater than a tolerance limit, specified by the analyst, based on the instrumental stability and the analyst’s needs. Information is provided by which the analyst can select an appropriate tolerance limit for the BE measurements and the frequency of calibration checks.
1.3 This practice is based on the assumption that the BE scale of the spectrometer is sufficiently close to linear to allow for calibration by measurements of reference photoelectron lines having BEs near the extremes of the working BE scale. In most commercial instruments, X-ray sources with aluminum or magnesium anodes are employed and BEs are typically measured at least over the 0–1200 eV range. This practice can be used for the BE range from 0 eV to 1040 eV.
1.4 The assumption that the BE scale is linear is checked by a measurement made with a reference photoelectron line or Auger-electron line that appears at an intermediate position. A single check is a necessary but not sufficient condition for establishing linearity of the BE scale. Additional checks can be made with specified reference lines on instruments equipped with magnesium or unmonochromated aluminum X-ray sources, with secondary BE standards, or by following the procedures of the instrument manufacturer. Deviations from BE-scale linearity can occur because of mechanical misalignments, ...
General Information
- Status
- Published
- Publication Date
- 31-Oct-2016
- Technical Committee
- E42 - Surface Analysis
Relations
- Effective Date
- 01-Nov-2016
- Effective Date
- 01-Apr-2022
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Jun-2015
- Effective Date
- 15-Nov-2013
- Effective Date
- 15-Nov-2013
- Effective Date
- 15-Nov-2013
- Effective Date
- 15-Nov-2013
- Effective Date
- 15-Aug-2013
- Effective Date
- 01-May-2012
- Effective Date
- 01-May-2012
- Effective Date
- 01-May-2009
- Effective Date
- 01-May-2009
- Effective Date
- 01-Apr-2008
Overview
ASTM E2108-16 is the Standard Practice for Calibration of the Electron Binding-Energy Scale of an X-Ray Photoelectron Spectrometer (XPS). Published by ASTM International, this standard provides detailed procedures for calibrating the electron binding-energy (BE) scale in XPS instruments. The accurate calibration of the BE scale is essential for reliable surface analysis of materials, enabling precise identification of elements and determination of chemical states based on photoelectron spectra. The standard applies to spectrometers using unmonochromated aluminum or magnesium Kα X-rays, or monochromated aluminum Kα X-rays.
Key Topics
- Importance of Calibration: Ensures that XPS measurements are accurate and comparable across different instruments, with uncertainties typically limited to about 0.1 eV to 0.2 eV.
- Calibration Procedures: Describes methods for calibrating the BE scale using reference photoelectron lines from metals such as copper and gold, and optionally silver.
- Measurement Uncertainty: Discusses sources of uncertainty-such as instrument stability and measurement conditions-and provides ways to set and verify appropriate tolerance limits.
- Frequency of Calibration: Recommends initial calibration after installation or modification, with periodic checks and recalibrations driven by instrument stability and analytical needs.
- BE Scale Linearity: Details procedures for verifying the linearity of the BE scale and provides guidance for addressing non-linearity issues.
- Documentation and Traceability: Encourages consistent documentation of instrumental settings and calibration outcomes for quality management and traceability.
Applications
- Surface Analysis in Materials Science: Accurate BE scale calibration is vital for identifying elemental compositions and chemical states on material surfaces in research and industry.
- Database Consistency: Enables meaningful use of XPS data in published databases and comparison with literature values or results from other laboratories.
- Quality Assurance: Supports laboratories adhering to quality management systems like ISO 9001:2015, ensuring that measurement and test equipment are properly calibrated and that uncertainties are quantified and documented.
- Routine Instrument Maintenance: The standard guides ongoing maintenance and performance checks of XPS instruments to ensure reliable operation and data quality over time.
- Analytical Method Development: Provides a structured approach for establishing routine calibration protocols, accommodating a range of XPS instrument configurations and performance requirements.
Related Standards
- ASTM E902 - Practice for Checking the Operating Characteristics of X-Ray Photoelectron Spectrometers
- ASTM E1016 - Guide for Literature Describing Properties of Electrostatic Electron Spectrometers
- ASTM E1078 - Guide for Specimen Preparation and Mounting in Surface Analysis
- ASTM E1523 - Guide to Charge Control and Charge Referencing Techniques in X-Ray Photoelectron Spectroscopy
- ISO 15472:2010 - Surface Chemical Analysis-X-Ray Photoelectron Spectrometers-Calibration of Energy Scale
- ISO 9001:2015 - Quality Management Systems-Requirements
- ISO 18115-1:2013 - Surface Chemical Analysis-Vocabulary-Part 1: General Terms and Terms Used in Spectroscopy
Keywords: X-Ray Photoelectron Spectrometer calibration, ASTM E2108-16, electron binding-energy scale, XPS instrument calibration, surface analysis standards, BE measurement uncertainty, XPS quality assurance, surface chemical analysis standards, ISO 9001 calibration, XPS routine maintenance.
Buy Documents
ASTM E2108-16 - Standard Practice for Calibration of the Electron Binding-Energy Scale of an X-Ray Photoelectron Spectrometer
REDLINE ASTM E2108-16 - Standard Practice for Calibration of the Electron Binding-Energy Scale of an X-Ray Photoelectron Spectrometer
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E2108-16 is a standard published by ASTM International. Its full title is "Standard Practice for Calibration of the Electron Binding-Energy Scale of an X-Ray Photoelectron Spectrometer". This standard covers: SIGNIFICANCE AND USE 5.1 X-ray photoelectron spectroscopy is used extensively for the surface analysis of materials. Elements (with the exception of hydrogen and helium) are identified from comparisons of the binding energies determined from photoelectron spectra with tabulated values. Information on chemical state can be derived from the chemical shifts of measured photoelectron and Auger-electron features with respect to those measured for elemental solids. 5.2 Calibrations of the BE scales of XPS instruments are required for four principal reasons. First, meaningful comparison of BE measurements from two or more XPS instruments requires that the BE scales be calibrated, often with an uncertainty of about 0.1 eV to 0.2 eV. Second, identification of chemical state is based on measurement of chemical shifts of photoelectron and Auger-electron features, again with an uncertainty of typically about 0.1 eV to 0.2 eV; individual measurements, therefore, should be made and literature sources need to be available with comparable or better accuracies. Third, the availability of databases (3) of measured BEs for reliable identification of elements and determination of chemical states by computer software requires that published data and local measurements be made with uncertainties of about 0.1 eV to 0.2 eV. Finally, the growing adoption of quality management systems, such as, ISO 9001:2015, in many analytical laboratories has led to requirements that the measuring and test equipment be calibrated and that the relevant measurement uncertainties be known. 5.3 The actual uncertainty of a BE measurement depends on instrument properties and stability, measurement conditions, and the method of data analysis. This practice makes use of tolerance limits ±δ (chosen, for example, at the 95 % confidence level) that represent the maximum likely uncertainty of a BE measurement, associated with the instrument in a specified time interval following a calibration (ISO 15472:2010). A user should select a... SCOPE 1.1 This practice describes a procedure for calibrating the electron binding-energy (BE) scale of an X-ray photoelectron spectrometer that is to be used for performing spectroscopic analysis of photoelectrons excited by unmonochromated aluminum or magnesium Kα X-rays or by monochromated aluminum Kα X-rays. 1.2 The calibration of the BE scale is recommended after the instrument is installed or modified in any substantive way. Additional checks and, if necessary, recalibrations are recommended at intervals chosen to ensure that BE measurements are statistically unlikely to be made with an uncertainty greater than a tolerance limit, specified by the analyst, based on the instrumental stability and the analyst’s needs. Information is provided by which the analyst can select an appropriate tolerance limit for the BE measurements and the frequency of calibration checks. 1.3 This practice is based on the assumption that the BE scale of the spectrometer is sufficiently close to linear to allow for calibration by measurements of reference photoelectron lines having BEs near the extremes of the working BE scale. In most commercial instruments, X-ray sources with aluminum or magnesium anodes are employed and BEs are typically measured at least over the 0–1200 eV range. This practice can be used for the BE range from 0 eV to 1040 eV. 1.4 The assumption that the BE scale is linear is checked by a measurement made with a reference photoelectron line or Auger-electron line that appears at an intermediate position. A single check is a necessary but not sufficient condition for establishing linearity of the BE scale. Additional checks can be made with specified reference lines on instruments equipped with magnesium or unmonochromated aluminum X-ray sources, with secondary BE standards, or by following the procedures of the instrument manufacturer. Deviations from BE-scale linearity can occur because of mechanical misalignments, ...
SIGNIFICANCE AND USE 5.1 X-ray photoelectron spectroscopy is used extensively for the surface analysis of materials. Elements (with the exception of hydrogen and helium) are identified from comparisons of the binding energies determined from photoelectron spectra with tabulated values. Information on chemical state can be derived from the chemical shifts of measured photoelectron and Auger-electron features with respect to those measured for elemental solids. 5.2 Calibrations of the BE scales of XPS instruments are required for four principal reasons. First, meaningful comparison of BE measurements from two or more XPS instruments requires that the BE scales be calibrated, often with an uncertainty of about 0.1 eV to 0.2 eV. Second, identification of chemical state is based on measurement of chemical shifts of photoelectron and Auger-electron features, again with an uncertainty of typically about 0.1 eV to 0.2 eV; individual measurements, therefore, should be made and literature sources need to be available with comparable or better accuracies. Third, the availability of databases (3) of measured BEs for reliable identification of elements and determination of chemical states by computer software requires that published data and local measurements be made with uncertainties of about 0.1 eV to 0.2 eV. Finally, the growing adoption of quality management systems, such as, ISO 9001:2015, in many analytical laboratories has led to requirements that the measuring and test equipment be calibrated and that the relevant measurement uncertainties be known. 5.3 The actual uncertainty of a BE measurement depends on instrument properties and stability, measurement conditions, and the method of data analysis. This practice makes use of tolerance limits ±δ (chosen, for example, at the 95 % confidence level) that represent the maximum likely uncertainty of a BE measurement, associated with the instrument in a specified time interval following a calibration (ISO 15472:2010). A user should select a... SCOPE 1.1 This practice describes a procedure for calibrating the electron binding-energy (BE) scale of an X-ray photoelectron spectrometer that is to be used for performing spectroscopic analysis of photoelectrons excited by unmonochromated aluminum or magnesium Kα X-rays or by monochromated aluminum Kα X-rays. 1.2 The calibration of the BE scale is recommended after the instrument is installed or modified in any substantive way. Additional checks and, if necessary, recalibrations are recommended at intervals chosen to ensure that BE measurements are statistically unlikely to be made with an uncertainty greater than a tolerance limit, specified by the analyst, based on the instrumental stability and the analyst’s needs. Information is provided by which the analyst can select an appropriate tolerance limit for the BE measurements and the frequency of calibration checks. 1.3 This practice is based on the assumption that the BE scale of the spectrometer is sufficiently close to linear to allow for calibration by measurements of reference photoelectron lines having BEs near the extremes of the working BE scale. In most commercial instruments, X-ray sources with aluminum or magnesium anodes are employed and BEs are typically measured at least over the 0–1200 eV range. This practice can be used for the BE range from 0 eV to 1040 eV. 1.4 The assumption that the BE scale is linear is checked by a measurement made with a reference photoelectron line or Auger-electron line that appears at an intermediate position. A single check is a necessary but not sufficient condition for establishing linearity of the BE scale. Additional checks can be made with specified reference lines on instruments equipped with magnesium or unmonochromated aluminum X-ray sources, with secondary BE standards, or by following the procedures of the instrument manufacturer. Deviations from BE-scale linearity can occur because of mechanical misalignments, ...
ASTM E2108-16 is classified under the following ICS (International Classification for Standards) categories: 71.040.50 - Physicochemical methods of analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2108-16 has the following relationships with other standards: It is inter standard links to ASTM E2108-10, ASTM E456-13a(2022)e1, ASTM E456-13A(2017)e3, ASTM E456-13A(2017)e1, ASTM E1523-15, ASTM E456-13ae1, ASTM E456-13ae2, ASTM E456-13a, ASTM E456-13ae3, ASTM E456-13, ASTM E456-12e1, ASTM E456-12, ASTM E1078-09, ASTM E1523-09, ASTM E456-08e2. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2108-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2108 − 16
Standard Practice for
Calibration of the Electron Binding-Energy Scale of an
X-Ray Photoelectron Spectrometer
This standard is issued under the fixed designation E2108; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope made with specified reference lines on instruments equipped
with magnesium or unmonochromated aluminum X-ray
1.1 This practice describes a procedure for calibrating the
sources, with secondary BE standards, or by following the
electron binding-energy (BE) scale of an X-ray photoelectron
procedures of the instrument manufacturer. Deviations from
spectrometer that is to be used for performing spectroscopic
BE-scale linearity can occur because of mechanical
analysis of photoelectrons excited by unmonochromated alu-
misalignments, excessive magnetic fields in the region of the
minum or magnesium Kα X-rays or by monochromated
analyzer, or imperfections or malfunctions in the power sup-
aluminum Kα X-rays.
plies. This practice does not check for, nor identify, problems
1.2 The calibration of the BE scale is recommended after
of this type but simply verifies the linearity of the BE scale.
the instrument is installed or modified in any substantive way.
1.5 After an initial check of the BE-scale linearity and
Additional checks and, if necessary, recalibrations are recom-
measurements of the repeatability standard deviation for the
mendedatintervalschosentoensurethatBEmeasurementsare
main calibration lines for a particular instrument, a simplified
statistically unlikely to be made with an uncertainty greater
procedure is given for routine checks of the calibration at
than a tolerance limit, specified by the analyst, based on the
subsequent times.
instrumental stability and the analyst’s needs. Information is
provided by which the analyst can select an appropriate 1.6 This practice is recommended for use with X-ray
tolerance limit for the BE measurements and the frequency of photoelectron spectrometers operated in the constant-pass-
calibration checks. energy or fixed-analyzer-transmission mode and for which the
pass energy is less than 200 eV; otherwise, depending on the
1.3 This practice is based on the assumption that the BE
configurationoftheinstrument,arelativisticequationcouldbe
scale of the spectrometer is sufficiently close to linear to allow
needed for the calibration. The practice should not be used for
for calibration by measurements of reference photoelectron
instruments operated in the constant-retardation-ratio mode at
lineshavingBEsneartheextremesoftheworkingBEscale.In
retardation ratios less than 10, for instruments with an energy
mostcommercialinstruments,X-raysourceswithaluminumor
resolution above 1.5 eV, or in applications for which BE
magnesium anodes are employed and BEs are typically mea-
measurementsaredesiredwithtolerancelimitsof 60.03eVor
sured at least over the 0–1200 eV range. This practice can be
less.
used for the BE range from 0 eV to 1040 eV.
1.7 On instruments equipped with a monochromated alumi-
1.4 TheassumptionthattheBEscaleislinearischeckedby
num Kα X-ray source, a measurement of the position of a
a measurement made with a reference photoelectron line or
specified Auger-electron line can be used, if desired, to
Auger-electron line that appears at an intermediate position.A
determine the average energy of the X-rays incident on the
single check is a necessary but not sufficient condition for
specimen. This information is needed for the determination of
establishinglinearityoftheBEscale.Additionalcheckscanbe
modified Auger parameters.
1.8 The values stated in SI units are to be regarded as
This practice is under the jurisdiction of ASTM Committee E42 on Surface
standard. No other units of measurement are included in this
AnalysisandisthedirectresponsibilityofSubcommitteeE42.03onAugerElectron
Spectroscopy and X-Ray Photoelectron Spectroscopy.
standard.
Current edition approved Nov. 1, 2016. Published December 2016. Originally
1.9 This standard does not purport to address all of the
approved in 2000. Last previous edition approved in 2010 as E2108–10. DOI:
10.1520/E2108-16. safety concerns, if any, associated with its use. It is the
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2108 − 16
responsibility of the user of this standard to establish appro- electron transition of copper (Mg source or unmonochromated
priate safety and health practices and determine the applica- Al source) (1, 2); additional checks can be made, if desired,
bility of regulatory limitations prior to use. with secondary standards. Procedures are given for determin-
ing the components of an uncertainty budget in BE measure-
2. Referenced Documents
ments and for determining the uncertainties of BE measure-
ments(atthe95%confidencelevel)atvarioustimesfollowing
2.1 ASTM Standards:
a calibration. The analyst can thus establish tolerance limits,
E456Terminology Relating to Quality and Statistics
E673TerminologyRelatingtoSurfaceAnalysis(Withdrawn for example, at the same level of confidence, based on the
instrument stability and the analyst’s needs so that BE mea-
2012)
E902Practice for Checking the Operating Characteristics of surements statistically are likely to be made within these limits
during specified time intervals following a calibration. The
X-Ray Photoelectron Spectrometers (Withdrawn 2011)
E1016Guide for Literature Describing Properties of Elec- instrument is then adjusted by following the procedures of the
instrument manufacturer or subsequent BE measurements are
trostatic Electron Spectrometers
E1078Guide for Specimen Preparation and Mounting in corrected by following the procedure outlined in this practice.
For a routine check of the instrumental calibration, either one
Surface Analysis
ortwomeasurementsaremadeforeachofthesamecorelevels
E1523Guide to Charge Control and Charge Referencing
of copper and gold. Fig. 1 is a flow chart that summarizes the
Techniques in X-Ray Photoelectron Spectroscopy
steps of the calibration procedure; references are given to
2.2 ISO Standards:
relevant sections of this practice. An optional procedure is
ISO 9001:2015 Quality management systems—
provided for determining the average energy of the X-rays
Requirements
from a monochromated Al X-ray source, using a measured
ISO 15472:2010Surface chemical analysis—X-ray photo-
position of a copper Auger peak.
electron spectrometers—Calibration of energy scales
ISO 18115-1:2013 Surface chemical analysis—
5. Significance and Use
Vocabulary—Part 1: General terms and terms used in
spectroscopy 5.1 X-ray photoelectron spectroscopy is used extensively
for the surface analysis of materials. Elements (with the
3. Terminology
exception of hydrogen and helium) are identified from com-
3.1 Definitions—SinceTerminologyE673waswithdrawnin parisons of the binding energies determined from photoelec-
tron spectra with tabulated values. Information on chemical
2012, definitions of terms used inAuger and X-ray photoelec-
tron spectroscopy are now based on ISO 18115-1:2013. For state can be derived from the chemical shifts of measured
definitions of terms used in statistics, see Terminology E456. photoelectronandAuger-electronfeatureswithrespecttothose
measured for elemental solids.
3.2 Symbols and Abbreviations—Table 1 shows definitions
of the symbols and abbreviations used in this practice. 5.2 Calibrations of the BE scales of XPS instruments are
required for four principal reasons. First, meaningful compari-
4. Summary of Practice
son of BE measurements from two or more XPS instruments
requires that the BE scales be calibrated, often with an
4.1 A procedure is given for calibrating the BE scale of an
uncertainty of about 0.1 eVto 0.2 eV. Second, identification of
X-ray photoelectron spectrometer equipped with one or more
chemical state is based on measurement of chemical shifts of
of the following sources of characteristic Kα X-rays: magne-
photoelectron and Auger-electron features, again with an
sium (Mg) source; unmonochromated aluminum (Al) source;
uncertainty of typically about 0.1 eV to 0.2 eV; individual
or monochromatedAl source. This procedure is based on ISO
measurements,therefore,shouldbemadeandliteraturesources
15472:2010. In a first calibration for particular operating
need to be available with comparable or better accuracies.
conditions of the instrument, or after the instrument has been
Third, the availability of databases (3) of measured BEs for
modified, measurements are made of the BEs of specified core
reliable identification of elements and determination of chemi-
levels of copper and gold, and these values are then compared
cal states by computer software requires that published data
withcorrespondingreferenceenergies (1). Thelinearityofthe
and local measurements be made with uncertainties of about
BE scale is checked at a single point on the scale using a
0.1 eV to 0.2 eV. Finally, the growing adoption of quality
measurement of the position of either a specified core level of
management systems, such as, ISO 9001:2015, in many
silver (monochromated Al source) or a specified Auger-
analytical laboratories has led to requirements that the measur-
ing and test equipment be calibrated and that the relevant
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
measurement uncertainties be known.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on
5.3 TheactualuncertaintyofaBEmeasurementdependson
the ASTM website.
3 instrument properties and stability, measurement conditions,
The last approved version of this historical standard is referenced on
and the method of data analysis. This practice makes use of
www.astm.org.
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
tolerance limits 6δ (chosen, for example, at the 95% confi-
4th Floor, New York, NY 10036, http://www.ansi.org.
dence level) that represent the maximum likely uncertainty of
https://www.iso.org/obp/ui/#iso:std:iso:18115:-1:ed-2:v1:en.
a BE measurement, associated with the instrument in a
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof
this standard. specified time interval following a calibration (ISO
E2108 − 16
TABLE 1 Definitions of Symbols and Abbreviations
a measured energy scaling error
BE binding energy, in eV
b measured zero offset error, in eV
c number of counts in the i-th channel
i
eV electron volts
E corrected result for the binding energy corresponding to a given E ,ineV
corr meas
E binding energy of a frequently measured element at which the indicated binding energy scale is set, after
elem
calibration, to read correctly, in eV
E kinetic energy of a peak, with reference to the Fermi level, in eV
K
E a measured binding energy, in eV
meas
E average of the measured binding energies for the peak, n,in Table 3,ineV
meas n
E one of a set of measurements of binding energy for the peak, n,in Table 3,ineV
meas ni
E reference values for the position of peak, n,in Table 3, on the binding energy scale, in eV
ref n
E peak binding energy, in eV
p
E binding energy for first data channel at lower binding energy than the channel with the maximum number of counts,
for a peak, in eV
FWHM full width at half maximum peak intensity above the background, in eV
g channel energy separation, in eV
hv effective X-ray energy from an unmonochromated Al X-ray source, in eV
Al
mon
effective X-ray energy from a monochromated Al X-ray source, in eV
hϑ
Al
hv effective X-ray energy from an unmonochromated Mg X-ray source, in eV
Mg
i index to represent channel number, where i = 0 represents the first channel at lower binding energy than the chan-
nel with the maximum number of counts, for a peak, in eV
j number of repeat measurements for a new peak
k number of repeat measurements for the Au 4f ,Cu2p and Ag 3d or Cu L VV peaks in the repeatability stan-
7/2 3/2 5/2 3
dard deviation and linearity determinations
m number of repeat measurements for the Au 4f and Cu 2p peaks in the regular calibrations
7/2 3/2
n designation of the peak identifier in Table 3
p parameter in Eq A1.1, defined in Eq A1.2 and Section A1.2
q parameter in Eq A1.1, defined in Eq A1.3 and Section A1.2
r parameter in Eq A1.1, defined in Eq A1.4 and Section A1.2
t Student’s t value for x degrees of freedom of a two-sided distribution for a confidence level of 95 %
x
U total uncertainty of the calibrated energy scale at a confidence level of 95 %, in eV
c
uncertainty at a confidence level of 95 % arising from the calibration using the Au 4f and Cu 2p peaks at bind-
U (E) 7/2 3/2
ing energy E, assuming perfect scale linearity, in eV
l
uncertainty of ε or ε at a confidence level of 95 % from Eq 7 and Section 8.9.3,ineV
U
2 3
cl
uncertainty of the calibration at a confidence level of 95 % in the absence of a linearity error, from Eq 12 or Eq 13
U
and Section 8.10.4,ineV
XPS X-ray photoelectron spectroscopy
α Auger parameter, in eV
α' modified Auger parameter, in eV
∆ offset energy, given by the average measured binding energy for a calibration peak minus the reference energy, in
n
eV, for n=1,2,3,4in Table 3, for a given X-ray source
∆E correction to be added to E , after calibration, to provide the corrected result for the binding energy, in eV
corr meas
∆E value of ∆E for peaks 1 and 4 in Table 3,ineV
corr n corr
∆E drift of the binding-energy scale following a calibration for peaks 1 and 4 in Table 3,ineV
n
∆φ the average of ∆ and ∆ from Eq 16 and Section 8.11.1.2,ineV
1 4
mon
∆hv
difference between hϑ and hv ,ineV
Al
Al
δ value for the tolerance limit of energy calibration at a confidence level of 95 % (set by the analyst), in eV
ε measured scale linearity error at the Ag 3d peak from Eq 4 and Section 8.9.2,ineV
2 5/2
ε measured scale linearity error at the Cu L VV peak from Eq 5 or Eq 6 and Section 8.9.2,ineV
3 3
σ maximum of σ , σ or σ , and σ ,ineV
R R1 R2 R3 R4
σ repeatability standard deviation for the seven measurements of the binding energy of peak, n,in Table 3,ineV
Rn
σ repeatability standard deviation for a new peak, in eV
Rnew
15472:2010). A user should select a value of δ based on the uncertainty of the calibration procedure. This information is
needs of the analytical work to be undertaken, the likely provided for four common analytical situations. It is important
measurement and data-analysis conditions, the stability of the
to note that some BE measurements may have uncertainties
instrument, and the cost of calibrations. This practice gives
larger than δ as a result of poor counting statistics, large peak
information on the various sources of uncertainty in BE
widths,uncertaintiesassociatedwithpeakfitting,andeffectsof
measurements and on measurements of instrumental stability.
surface charging.
The analyst should initially choose some desired value for δ
5.4 Instrumentsettingstypicallyselectedforanalysisshould
and then make tests, as described in 8.14 to determine from
be used with this practice. Separate calibrations should be
subsequent checks of the calibration whether BE measure-
made if key operating conditions, such as choices of analyzer
ments are made within the limits 6δ. Information is given in
Appendix X1 on how to evaluate for a material of interest the pass energy, aperture sizes, or X-ray source, are varied.
uncertainty of a BE measurement that is associated with the Settingsnotspecifiedinthispracticeareatthediscretionofthe
E2108 − 16
NOTE 1—The number associated with each step indicates the section in which the operation is described.
FIG. 1 Flowchart Indicating Sequence of Operations for the Calibration.
E2108 − 16
user, but those same settings should be recorded and consis- patible with the specimen holder of the X-ray photoelectron
tently used whenever this practice is repeated in order that the spectrometer, and typically are 10 by 10 mm with a thickness
current results will be directly comparable to the previous of 0.2 mm.
results.
6.3 Ion Gun—Thispracticerequiresuseofaniongunonthe
5.5 All of the operations described in Section 8 should be X-ray photoelectron spectrometer for sputter cleaning of the
performed the first time that the BE scale is calibrated or after reference specimens used for the calibration measurements.
anysubstantialmodificationoftheinstrument.Forlaterchecks
6.4 Electron Emission Angle—The electron emission angle
of the calibration, to be performed on a regular schedule, only
should be between 0° and 56° with respect to the surface
the operations in 8.2 – 8.5, 8.10, 8.11, and 8.14 need to be
normal (5). The reference energies used in this practice are
performed. While the measurements described in 8.7 – 8.9 for
only valid for this range of emission angles.
the first calibration require moderate time and effort, they are
essential for ensuring that realistic tolerance limits 6δ have
7. Principle of the Calibration Method
been chosen. The control chart, described in 8.14, is a simple
7.1 For calibration of the XPS instrument with chosen
andeffectivemeansofdemonstratinganddocumentingthatthe
BE scale of the instrument is in calibration, that is, within the instrumentalsettings,forexample,X-raysource,analyzerpass
energy, energy step size, and apertures, measurements are
tolerance limits, for a certain period of time.
made of the binding energies of the Au 4f and Cu 2p
7/2 3/2
5.6 The average energy of the X-rays incident on the
photoelectron peaks. These peaks are chosen because they are
specimen for instruments equipped with a monochromated Al
near the low and high BE limits for many practical analyses,
X-ray source will generally be slightly higher, by up to about
Au and Cu foils of sufficient purity are readily available, the
0.2eV,thantheaverageX-rayenergyforinstrumentsequipped
foil surfaces can be cleaned readily by ion sputtering, and the
with an unmonochromated Al X-ray source (4). The actual
reference energies needed for the calibration have been vali-
energy difference depends on the alignment and thermal
dated for a range of operating conditions (5). In an initial
stabilityoftheX-raymonochromator.Anoptionalprocedureis
calibration of the instrument (or a calibration made after any
giveninAppendixX2todeterminethisenergydifferencefrom
substantive instrumental modifications), a check of the BE-
measurements of the Cu L VV Auger-electron peak. This
scale linearity is made with either the Ag 3d photoelectron
5/2
information is needed for the determination of modifiedAuger
peak (with a monochromatedAl X-ray source) or with the Cu
parameters andAuger-electron kinetic energies on instruments
L VV Auger-electron peak (with an unmonochromated Al or
with the monochromated Al X-ray source.
Mg X-ray source). In this first calibration, a series of seven
independent measurements are made for each of the Au 4f ,
7/2
6. Apparatus
Cu2p andeitherAg3d orCuL VVpeaks(see8.7).These
3/2 5/2 3
6.1 X-ray Photoelectron Spectrometer—The X-ray photo- measurementsaremadetodeterminetherepeatabilitystandard
electron spectrometer is the instrument to be calibrated. The deviationsformeasurementsofthethreepeaks.Thesestandard
essential components of the spectrometer are an X-ray source deviations have contributions from changes in outputs of the
(preferably with an X-ray tube containing one or more anodes spectrometer power supplies, from the sensitivity of the mea-
of aluminum, or magnesium, or both), an ion gun for sample suredBEtosampleposition,andfromcountingstatisticsinthe
sputtering, a specimen holder that can hold three or more recorded spectra (although the latter uncertainty is relatively
specimens, and an electron energy analyzer with which pho- small for the conditions specified in this practice). The mag-
toelectron intensities are measured as a function of photoelec- nitudes of the other two contributions may vary with BE, and
tron binding energy (the analyzer collecting photoelectrons so an overall repeatability standard deviation σ is defined as
r
under a given angular range as specified in 6.4). Some the largest of the repeatability standard deviations for the three
instruments with an Al X-ray source are equipped with an peaks. The calibration will be valid only if the sample-
X-raymonochromator,andalsoachargecompensationdevice. positioning procedure used for regular analyses is the same as
XPS instruments also measure the intensities of Auger elec- the procedure used in the calibration.
trons that are emitted at characteristic energies. See Practice
7.2 IthasbeenfoundininterlaboratorycomparisonsofXPS
E902 for procedures to check the operating characteristics of
measurements that any measured error in measured BEs varies
an X-ray photoelectron spectrometer and Guide E1016 for
approximately linearly with BE. This practice is based on the
information on the properties of electrostatic electron spec-
assumption that the differences between measured BEs and
trometers.
reference BEs for the designated peaks are both small and
6.2 Reference Specimens—The reference specimens, with linearly, or close to linearly, dependent on BE. A test of the
which the spectrometer is calibrated, are polycrystalline foils validity of this assumption is provided (see 8.7 and 8.9)bya
of high-purity (at least 99.8% pure) copper and gold. For measurementatanintermediatepointontheBEscaleusingthe
instruments with a monochromated Al X-ray source, a poly- Ag3d photoelectronpeak(monochromatedAlX-raysource)
5/2
crystalline silver film of similar purity is also required as a or the Cu L VV Auger peak (unmonochromated X-ray
reference specimen. If desired, other high-purity metals can be sources). The Ag 3d measurement is needed for monochro-
5/2
usedtocheckthelinearityoftheBEscalethroughoutitsrange. matedAlX-raysourcesbecausetheaverageX-rayenergyfrom
See Guide E1078 for guidance on specimen preparation and this source may be larger, by up to 0.2 eV, than for an
mounting. The foil dimensions should be chosen to be com- unmonochromatedAlX-raysourceand,asaresult,therelative
E2108 − 16
energies of photoelectron and Auger-electron peaks may vary 7.4 ThemeansbywhichtheBEscaleiscorrectedfollowing
by up to 0.2 eV (4). The Ag 3d peak could be used for the acalibrationoracalibrationcheckdependsonpracticaldetails
5/2
linearity check with unmonochromated X-ray sources, if oftheinstrument.Somemethodsformakingthiscorrectionare
desired,butitwillgenerallybesimplertomakethischeckwith described in 8.11.
the Cu L VV Auger peak because the copper foil is already
7.5 The tolerance limit δ for the calibration at the 95%
required for measurement of the Cu 2p peak. Additional
3/2
confidence limit is an important parameter to be chosen by the
checks of BE-scale linearity can be made, if desired, using
analyst. This parameter also is the tolerance limit, that is, the
appropriate elemental metals and the corresponding recom-
maximum allowed inaccuracy, again at the 95% confidence
mended BEs as secondary standards (6).
limit, for measured BEs for some defined time following a
7.3 If the BE scale is judged to be sufficiently linear (see calibration. The chosen value of δ depends in part on the
8.9), subsequent checks of the calibration can be performed analytical requirements and in part on the stability of the
with the number m of measurements for each of the Au 4f instrumentalBEscale.Table2showsillustrativevaluesforthe
7/2
and Cu 2p photoelectron peaks chosen to be 1 or 2, as various uncertainties that contribute to U for values of δ of
3/2 95
described in 8.10. An expression is given in this section for 0.1eVand0.2eVandforvaluesof mof1or2.Thedifference
calculating the uncertainty U of the BE-scale calibration at betweenδandU isthemaximumdriftoftheinstrumentalBE
95 95
the 95% confidence level (at the time of the calibration). scale that should be allowed between calibration checks.
TABLE 2 Contributions to an Error Budget for Calibration of the BE scale (from ISO 15472:2010)
NOTE 1—The uncertainties are for a 95% confidence level.The numerical values shown are illustrations to indicate the effects of different choices (in
the values of δ and m) on the uncertainty of the calibration U and the required interval between calibrations for assumed values of σ, ε,or ε , and
95 r 2 3
the average drift rate of the BE scale.
Item Symbol Calculated from Examples
If you require a If you require a
high accuracy lower accuracy
Tolerance limits, eV ± δ You choose ±0.1 ±0.2 (Your choice is dictated by the
accuracy you require and the
number of spectra you have
time to acquire in regular cali-
brations.)
Repeatability standard σ Eq 1 and Sec- 0.020 0.020 (Characteristic of your spec-
R
deviation, eV tion 8.8.3 trometer measured at first cali-
bration (see 8.7)).
Number of times each m You choose m=1 m=2 m=1 m=2
pair of spectra is m=1or2
acquired
cl
Uncertainty of calibra- Eq 12 or Eq 13 0.074 0.052 0.074 0.052
U
tion measurements, and Section
eV 8.10.4
Measure of scale non- ε or ε Eq 4 and Sec- 0.020 0.020 0.020 0.020 (Characteristic of your spec-
2 3
linearity, eV tion 8.9.2, Eq 5 trometer measured at first cali-
or Eq 6 and Sec- bration (see 8.7)).
tion 8.9.2
Uncertainty of energy U Eq 11 and Sec- 0.078 0.057 0.078 0.057
scale after tion 8.10.4
calibration, eV
Maximum allowable ±(δ-U ) δ and U ±0.022 ±0.043 ±0.122 ±0.143 (Define the drift allowable be-
95 95
drift between fore you are in danger of ex-
calibrations, eV ceeding your chosen limits ±δ
eV.)
Maximum calibration - Section 8.13 0.9 1.7 4.9 5.7 (Choose a convenient interval
interval (for a steady below this maximum, and less
drift rate of 0.025 eV than four months, with safety
per month), months margin for any erratic behav-
ior.)
Your choice of calibra- - You choose Option not 1 3 4 (No more than 70 % of the
tion interval, months based on practical maximum interval in the pre-
observed drift ceding row to allow a safety
behavior margin.)
E2108 − 16
Information on how to determine the maximum interval should be low enough so that the heights of the carbon and
between calibration checks is given in 8.13. It is strongly oxygen1speaksshallnotexceed3%oftheheightofthemost
recommended that analysts prepare their own version of Table intense metal peak by the time the measurements are com-
2 using numerical values from their measurements and the pleted (see 8.10) or at the end of the working day, whichever
indicated equations and steps. is earlier.
7.6 An analyst unfamiliar with this practice will have to
NOTE 2—Suitable sputtering conditions with an inert gas are 1 min of
make an initial choice for δ, and it is suggested that a value of a 30 µA beam of argon ions covering 1 cm of the surface.
NOTE 3—Illustrative survey spectra for Au, Ag, and Cu may be found
0.1 eV be chosen. To decide whether the selected value of δ is
in Refs (7-11).
realistic, this practice should be followed and a version of
Table 2 should be completed. In general, the value of δ should
8.3.2 It is desirable to complete the measurements in one
bebasedontheaccuracyneedsoftheplannedanalyticalwork,
working day. If a longer time is needed, additional surface
the expected measurement and data-analysis conditions, the
cleaning of the samples probably will be needed (see 8.3.1).
linearity of the BE scale, the stability of the instrument, and a
8.4 Choose the spectrometer settings for which energy
judgment based on the needs and benefits of calibration with
calibration is required—Choose the operating settings of the
the chosen value of δ versus the cost in instrument and staff
spectrometer for which energy calibration is required. The
time in performing calibrations. For example, if δ is chosen to
calibrationprocedure(see8.4–8.13)shallberepeatedforeach
be too small, calibration checks may have to be made too
X-ray source and selected combination of pass energy, retar-
frequently to account for possible drift of the BE scale in the
dationratio,apertures,lenssettings,etc.forwhichacalibration
interval between calibration checks. Other options in such a
is needed. A calibration is only valid for the particular X-ray
situation are to review the operating procedures, for example,
source and operating conditions. Record the chosen operating
the sample-alignment procedure, or the calibration procedure,
settings.
for example, the choice of m), to determine if one or more of
thetermscontributingto U canbereduced.Otherwise,δmay
NOTE 4—The designs of XPS instruments and their electronic controls
vary considerably. As a result, a calibration made for a particular X-ray
need to be increased to an acceptable value.
source and a particular combination of pass energy, apertures, and lens
settings will not necessarily be valid for another X-ray source or another
8. Calibration Procedure
combination of spectrometer settings (4). Many analysts make all or most
8.1 Obtain the Reference Samples—Use gold and copper
of their BE measurements for only one set of conditions; as a result, the
referencesamplesforXPSinstrumentswithunmonochromated instrument may only need calibration for these conditions.
X-ray sources. For instruments with a monochromated Al
8.5 OperatetheInstrument—Operatetheinstrumentaccord-
X-raysource,alsouseasilversampleonthefirstoccasionthat
ing to the manufacturer’s documented instructions. The instru-
the instrument is calibrated for the settings chosen in 8.4; for
ment shall have fully cooled following any bakeout. Ensure
later determinations of the calibration error, as described in
that the X-ray source power, counting rates, spectral scan rate,
8.10, use of the silver sample is not necessary. The samples
and other parameters are within the recommended ranges
shall be polycrystalline and with a purity of at least 99.8%.
specified by the manufacturer. Check that the settings for the
The samples can be foils of typical dimensions 10 by 10 mm
multiplier detector have been adjusted correctly. For multide-
and with thicknesses between 0.1 and 0.2 mm.
tector systems, ensure that any optimizations or checks recom-
mended by the manufacturer have been performed.
NOTE1—IftheCuandAgsamplesappeardiscolored,ashortdipin1%
nitric acid may be used to clean the surfaces with subsequent rinsing in
NOTE 5—Many manufacturers recommend that control and high-
distilledwater.IftheCusamplehasbeenstoredinairformorethanafew
voltage electronics be switched on at least four hours before performing
days, the cleaning with nitric acid will reduce the time needed for later
accurateBEmeasurements.Also,itmaybenecessarytooperatetheX-ray
sputter cleaning (see 8.3.1).
source for some minimum period, for example, 1 h, before making such
8.2 Mount the Samples—MounttheAuandCusamplesand,
measurements. XPS instruments with an X-ray monochromator may also
if required, the Ag sample on the sample holder of the need a warm-up time since the average X-ray energy at the sample may
depend on the ambient temperature or the temperature in the vicinity of
instrumentoronseparatesampleholders,asappropriate,using
the monochromator; records of these temperatures could help to identify
fixing screws or other metallic means to ensure electrical
any problems associated with drifts of measured peak positions.
contact. Double-sided adhesive tape should not be used when
NOTE 6—If the counting rates are too high (12) or the detector voltages
performing energy scale calibration work.
are set incorrectly (12, 13), spectral peaks can be distorted and the
resulting peak positions may be incorrect.
8.3 Clean the Samples:
8.3.1 Achieve ultra-high vacuum in the analytical chamber 8.6 Options for Initial or Subsequent Calibration
and clean the samples by ion sputtering until the heights of the Measurements—For the first time that the instrument has been
carbon and oxygen signals are each less than 2% of the height calibratedforaparticularcombinationofspectrometersettings
of the most intense metal peak in a survey spectrum for each (see8.4),itisnecessarytodeterminetherepeatabilitystandard
deviation for BE measurements, the BE-scale linearity error,
metal (if the silver sample is heavily contaminated it can also
be pre-cleaned using a pencil eraser or metal polish). Record a and the calibration interval. If any of these have not been
survey (widescan) spectrum for each sample to ensure that the determined, follow 8.7 – 8.9; otherwise, proceed to 8.10.Ifthe
only significant peaks are those expected for the particular instrument has been modified, undergone significant repair, or
metal. The pressure of active gases in the analytical chamber been moved, the steps in 8.7 – 8.9 should be followed.
E2108 − 16
TABLE 3 Reference Binding Energies,E for the Peaks Used
8.7 Measurements for the Repeatability Standard Deviation
ref n
in the Calibration Procedure for the Indicated X-Ray Sources
and Scale Linearity:
(1, 2)
8.7.1 The repeatability standard deviation for the peak BE,
NOTE 1—TheAg data included in parentheses are not normally used in
σ , is determined from repeated measurements of theAu 4f ,
R 7/2
the calibration.
Ag 3d or Cu L VV, and Cu 2p peaks as described in this
5/2 3 3/2
Peak Number, n Assignment E (eV)
ref n
sectionandin8.8.Thevalueofσ isvalidonlyfortheselected
R
Al Kα Mg Kα Monochro-
conditions (see 8.4), and involves a significant contribution
matic Al Kα
from the sample-positioning procedure (4, 5). A consistent 1Au4f 83.95 83.95 83.96
7/2
2Ag3d (368.22) (368.22) 368.21
5/2
sample-positioning procedure, following a documented
3CuL VV 567.93 334.90 -
protocol, for example, from the manufacturer’s
4Cu2p 932.63 932.62 932.62
3/2
recommendations, should be followed both for the calibration
measurements and for regular analytical work.
NOTE 7—The sample-positioning procedure will depend on the instru-
angleofelectronemission (5).ThereferenceBEvaluesinTable3areonly
ment design, the type and shape of the samples, and the analytical
validforemissionanglesbetween0°and56° (5).Iflargeremissionangles
requirements.Inmanycases,thecorrectsamplepositionisdeterminedby
maximizingthespectralintensity.Whenoptimizationinvolvesadjustment are used, significant errors will be introduced into the calibration.
NOTE 10—A wide range of spectral scan rates are available on
of two or more interacting parameters, a consistent optimization strategy
is necessary. If the optimization involves an X-ray monochromator, commercial XPS instruments. If the scan rate is too high, the measured
peakBEwillbeshifted.Thescanrateshouldbechosensothatthereisno
changes in the sample position may lead to shifts in the energy of the
recorded peak; thus, it may be necessary to measure peak intensities in an significant peak shift.
energy range of up to 60.5 eV from the nominal peak BE. In this case,
8.7.5 Set the copper sample at the analytical position with
intensity optimization may be more sensitive to sample position at low,
the same emission angle as in 8.7.4. Position the sample with
ratherthanhigh,bindingenergiesor,morerarely,viceversa.Optimization
the documented procedure and record the Cu 2p peak, with
is usually most effective at the BE where the intensity is most sensitive to
3/2
sample position. It may be useful to perform the operations in 8.7 several the same spectrometer settings as those used in 8.7.4, so that
times to refine the sample-positioning procedure and to obtain a lower
more than 40000 counts per channel are acquired in the
value of σ .
R
vicinity of the peak. Scan from at least 1 eV below the peak
8.7.2 The BE-scale linearity error, ε or ε , is determined
2 3 energy to at least 1 eV above the peak energy. Ensure that the
from measurements of the Cu L VV Auger-electron peak for
correct peak has been measured from the wide-scan (survey)
unmonochromated Mg orAl X-ray sources and from measure-
spectrum (see 8.3.1). The reference BE for the Cu 2p peak,
3/2
mentsoftheAg3d photoelectronpeakforamonochromated
peak 4, is given in Table 3.
5/2
Al X-ray source. These measurements are performed at the
8.7.6 If an unmonochromated Al or Mg X-ray source is
same time as the repeatability measurements to reduce the
being used, the Cu L VV peak will be measured next. If a
overall effort and to reduce the measurement uncertainty.
monochromated Al X-ray source is being used, set the silver
8.7.3 The order of data acquisition for the measurements
sample at the analytical position with the same emission angle
described in 8.7.4 – 8.7.7 should be as follows:
as in 8.7.4 and position the sample with the documented
8.7.3.1 Unmonochromated Mg or Al X-rays: Au 4f ,Cu
procedure. Record the Cu L VV peak or the Ag 3d peak,
7/2
3 5/2
2p ,CuL VV, with this sequence repeated six additional
3/2 3 depending on X-ray source, with the same spectrometer
times.
settingsasthoseusedin8.7.4,sothatmorethan40000counts
8.7.3.2 Monochromated Al X-rays: Au 4f ,Cu2p ,Ag
7/2 3/2 per channel are acquired in the vicinity of the peak. Scan from
3d , with this sequence repeated six additional times.
at least 1 eV below the peak energy to at least 1 eV above the
5/2
peak energy. Ensure that the correct peak has been measured
NOTE 8—The Au 4f peak is often the weakest peak although,
7/2
from the wide-scan (survey) spectrum (see 8.3.1). The refer-
depending on the spectrometer, sometimes the Cu L VV peak may be
weaker. Initial measurements with the Au 4f peak should facilitate the
enceBEsfortheAg3d peak,peak2,andtheCuL VVpeak,
7/2
5/2 3
use of a common set of measurement conditions for all peaks.
peak 3, are given in Table 3.
8.7.7 Repeatthestepsin8.7.4–8.7.6anadditionalsixtimes
8.7.4 Set the gold sample at the analytical position with the
angle of emission for the detected electrons with respect to the in order to obtain seven independent measurements for each of
the three peaks. To save time, the energy-scan widths may be
surface normal between 0° and 56°. Position the sample using
the documented procedure and record the Au 4f peak with reduced to the range 60.5 eV about the peak unless a wider
7/2
range is needed if instrumental software is to be used for peak
appropriate X-ray power and channel dwell time so that more
than 40000 counts per channel are acquired in the vicinity of location (see 8.8.1.2).
the peak. Scan the peak with the channel energy interval set at
8.8 Calculate the Repeatability Standard Deviation for the
approximately 0.05 eV or 0.1 eV depending on the method
Peak Binding Energies:
planned for determination of the peak BE (see 8.8.1). Scan
8.8.1 Determine the peak binding energies from the spectra
fromatleast1eVbelowthepeakenergytoatleast1eVabove
recorded in 8.7 using one of the three methods described in
the peak energy. Ensure that the correct peak has been
8.8.1.1,8.8.1.2,or8.8.1.3(specifyinthedocumentationwhich
measured from the wide-scan (survey) spectrum (see 8.3.1).
of the three methods is utilized).
The reference BE for the Au 4f peak, peak 1, is given in
7/2
NOTE 11—The first two methods are useful for instruments that supply
Table 3.
digital data, the third method for instruments that supply only graphical
NOTE 9—The reference BE values for the calibration peaks vary with output.
E2108 − 16
8.8.1.1 Make a least–squares fit of a parabola to six data 8.8.3 Calculatetheaveragebindingenergy,E ,fromthe
meas n
points around the top of the peak. There should be approxi- setofsevenmeasurements, E ,foreachpeak, n.Calculate
meas ni
mately the same number of data points above and below the therepeatabilitystandarddeviation,σ ,ofthesevenmeasure-
R1
energy for the maximum intensity, and the intensities for the ments of theAu 4f peak energy, E , from the equation:
7/2 meas li
first and last data points should be between 85% and 95% of
E 2 E
~ !
7 measli measl
σ 5 (1)
the maximum intensity. Obtain the energy for the peak maxi-
Rl (i51
mum from this fit. If software for this purpose is not conve-
where E is the average value of the E . Calculate
niently available, the simple numerical procedure given in
meas l meas li
Annex A1 can be used. the repeatability standard deviations, σ or σ , and σ , for
R2 R3 R4
the measured positions of the Ag 3d or Cu L VV and Cu
5/2 3
NOTE 12—If the procedure in Annex A1 is utilized, a channel energy
2p peaks, respectively, in a similar way. The overall repeat-
3/2
separation of 0.1 eV (or in the range 0.09 eV to 0.11 eV) should be
abilitystandarddeviation,σ ,istakenasthelargestofσ ,σ ,
selected for XPS with an unmonochromated X-ray source. For XPS with R R1 R2
a monochromatedAl X-ray source, the channel separation should be 0.05 or σ , and σ .
R3 R4
eV (or in the range of 0.045 eV to 0.055 eV) if the FWHM of the peaks
is less than 1.0 eV; otherwise, a channel width between 0.9 eV and 0.11 NOTE 16—Record the value of σ in your version of Table 2.
R
eV can be used.
8.8.4 Review the measured Au 4f and Cu 2p peak
7/2 3/2
NOTE 13—If the procedure in Annex A1 is utilized, the intensities for
energies (from 8.8.2) for any systematic changes with time
thefirstandlastdatapointscould,ifnecessary,bebetween80%and95%
of the maximum intensity. There would then be a slight increase (up to 6 through their order of acquisition.Any such systematic change
meV) in the uncertainty of the derived peak binding energy.
may indicate an inadequate warm-up period or some other
form of drift. In this situation, take appropriate action, for
8.8.1.2 Make a least-squares fit to the data points obtained
example, increase the warm-up period, and repeat 8.7.
in 8.8.1.1 with a suitable lineshape function available with
software on the XPS instrument or on another computer. A
8.8.5 The values of the repeatability standard deviation for
Gaussian function, a Lorentzian function, a Voigt function, or each peak should be less than 0.05 eV for an instrument in
a sum or product of such functions may be suitable functions
goodworkingcondition.Ifσ orσ exceedsthisvalue,check
R1 R4
for this purpose.Aconstant background can be used to aid the the stabilities of the voltages applied to the instrument, the
fitting, if desired, but no asymmetric background, such as a
adequacy of the system ground, and the sample-positioning
sloping line, a Shirley background, or a Tougaard background procedure. If σ is greater than δ/4, it will be necessary either
R
shall be subtracted from the measured intensities or utilized in
to find a way to reduce σ or increase the value of δ.
R
the fitting procedure. Obtain the energy for the peak maximum
NOTE 17—In an interlaboratory comparison in which copper samples
from the fit.
were repositioned following analysis of another sample, 87% of the
resultsshowedvaluesofσ thatwereequaltoorlessthan0.030eV (15).
NOTE14—PhotoelectronpeaksinXPSgenerallyareasymmetrical (14). R4
In another interlaboratory study, values of σ equal to or less than 0.021
The effects of any asymmetry in measured peaks on the determination of R4
eV were found from repeated measurements made without repositioning
the energy for the peak maximum will be insignificant for this practice if
of the samples (4). Values of σ as low as 0.001 eV have been reported
fits are only made to groups of data points that are selected as specified in R4
(4).
8.8.1.1. While fits can be made to a larger number of data points
comprising the peak, it will often be necessary to include an asymmetry
8.9 Check the Linearity of the Binding-Energy Scale:
parameter in the fitting function (14). In such cases, comparisons should
bemadeoftheresultingenergyforthepeakmaximumanditsuncertainty 8.9.1 Subtractthereferenceenergies, E ,giveninTable3
ref n
with the energy and uncertainty for the peak maximum obtained from an
from the corresponding values of the average measured bind-
alternative method, such as those described in 8.8.1.1 and 8.8.1.3. These
ing energies, E , determined in 8.8.3 to obtain the
meas n
comparisonsshouldbemadeforeachmeasuredpeakandforeachselected
measured offset energies, ∆ , for each peak n. Thus,
n
set of operating conditions (see 8.4).
∆ 5 E 2 E (2)
n meas n ref n
8.8.1.3 Draw chords horizontally across the peak at an
intensity of 84
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2108 − 10 E2108 − 16
Standard Practice for
Calibration of the Electron Binding-Energy Scale of an
X-Ray Photoelectron Spectrometer
This standard is issued under the fixed designation E2108; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice describes a procedure for calibrating the electron binding-energy (BE) scale of an X-ray photoelectron
spectrometer that is to be used for performing spectroscopic analysis of photoelectrons excited by unmonochromated aluminum
or magnesium Kα X-rays or by monochromated aluminum Kα X-rays.
1.2 The calibration of the BE scale is recommended after the instrument is installed or modified in any substantive way.
Additional checks and, if necessary, recalibrations are recommended at intervals chosen to ensure that BE measurements are
statistically unlikely to be made with an uncertainty greater than a tolerance limit, specified by the analyst, based on the
instrumental stability and the analyst’sanalyst’s needs. Information is provided by which the analyst can select an appropriate
tolerance limit for the BE measurements and the frequency of calibration checks.
1.3 This practice is based on the assumption that the BE scale of the spectrometer is sufficiently close to linear to allow for
calibration by measurements of reference photoelectron lines having BEs near the extremes of the working BE scale. In most
commercial instruments, X-ray sources with aluminum or magnesium anodes are employed and BEs are typically measured at least
over the 0–10000–1200 eV range. This practice can be used for the BE range from 0 eV to 1040 eV.
1.4 The assumption that the BE scale is linear is checked by a measurement made with a reference photoelectron line or
Auger-electron line that appears at an intermediate position. A single check is a necessary but not sufficient condition for
establishing linearity of the BE scale. Additional checks can be made with specified reference lines on instruments equipped with
magnesium or unmonochromated aluminum X-ray sources, with secondary BE standards, or by following the procedures of the
instrument manufacturer. Deviations from BE-scale linearity can occur because of mechanical misalignments, excessive magnetic
fields in the region of the analyzer, or imperfections or malfunctions in the power supplies. This practice does not check for, nor
identify, problems of this type.type but simply verifies the linearity of the BE scale.
1.5 After an initial check of the BE-scale linearity and measurements of the repeatability standard deviation for the main
calibration lines for a particular instrument, a simplified procedure is given for routine checks of the calibration at subsequent
times.
1.6 This practice is recommended for use with X-ray photoelectron spectrometers operated in the constant-pass-energy or
fixed-analyzer-transmission mode and for which the pass energy is less than 200 eV; otherwise, depending on the configuration
of the instrument, a relativistic equation could be needed for the calibration. The practice should not be used for instruments
operated in the constant-retardation-ratio mode at retardation ratios less than 10, for instruments with an energy resolution above
1.5 eV, or in applications for which BE measurements are desired with tolerance limits of 60.03 eV or less.
1.7 On instruments equipped with a monochromated aluminum Kα X-ray source, a measurement of the position of a specified
Auger-electron line can be used, if desired, to determine the average energy of the X-rays incident on the specimen. This
information is needed for the determination of modified Auger parameters.
1.8 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.9 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
This practice is under the jurisdiction of ASTM Committee E42 on Surface Analysis and is the direct responsibility of Subcommittee E42.03 on Auger Electron
Spectroscopy and X-Ray Photoelectron Spectroscopy.
Current edition approved Nov. 1, 2010Nov. 1, 2016. Published December 2010December 2016. Originally approved in 2000. Last previous edition approved in 20052010
as E2108 – 05.E2108 – 10. DOI: 10.1520/E2108-10.10.1520/E2108-16.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2108 − 16
2. Referenced Documents
2.1 ASTM Standards:
E456 Terminology Relating to Quality and Statistics
E673 Terminology Relating to Surface Analysis (Withdrawn 2012)
E902 Practice for Checking the Operating Characteristics of X-Ray Photoelectron Spectrometers (Withdrawn 2011)
E1016 Guide for Literature Describing Properties of Electrostatic Electron Spectrometers
E1078 Guide for Specimen Preparation and Mounting in Surface Analysis
E1523 Guide to Charge Control and Charge Referencing Techniques in X-Ray Photoelectron Spectroscopy
2.2 ISO Standards:
ISO 9001:20009001:2015 Quality Management Systems—Requirements management systems—Requirements
ISO 15472:200115472:2010 Surface Chemical Analysis—X-Ray Photoelectron Spectrometers—Calibration of Energy Scale-
schemical analysis—X-ray photoelectron spectrometers—Calibration of energy scales
ISO 18115:200118115-1:2013 Surface Chemical Analysis—Vocabulary chemical analysis—Vocabulary—Part 1: General terms
and terms used in spectroscopy
3. Terminology
3.1 Definitions—For definitions ofSince Terminology E673 terms used in X-ray photoelectron spectroscopy and surface
analysis, see Terminologywas withdrawn in 2012, definitions of terms used in Auger and E673 and ISO 18115:2001.X-ray
photoelectron spectroscopy are now based on ISO 18115-1:2013. For definitions of terms used in statistics, see Terminology
E456.
3.2 Symbols and Abbreviations—Table 1 shows definitions of the symbols and abbreviations used in this practice.
4. Summary of Practice
4.1 A procedure is given for calibrating the BE scale of an X-ray photoelectron spectrometer equipped with one or more of the
following sources of characteristic Kα X-rays: magnesium (Mg) source; unmonochromated aluminum (Al) source; or
monochromated Al source. This procedure is based on ISO 15472:2001.15472:2010. In a first calibration for particular operating
conditions of the instrument, or after the instrument has been modified, measurements are made of the BEs of specified core levels
of copper and gold, and these values are then compared with corresponding reference energies (1). The linearity of the BE scale
is checked at a single point on the scale using a measurement of the position of either a specified core level of silver
(monochromated Al source) or a specified Auger-electron transition of copper (Mg source or unmonochromated Al source) (1, 2);
additional checks can be made, if desired, with secondary standards. Procedures are given for determining the components of an
uncertainty budget in BE measurements and for determining the uncertainties of BE measurements (at the 95 % confidence level)
at various times following a calibration. The analyst can thus establish tolerance limits, for example, at the same level of
confidence, based on the instrument stability and the analyst’sanalyst’s needs so that BE measurements statistically are likely to
be made within these limits during specified time intervals following a calibration. The instrument is then adjusted by following
the procedures of the instrument manufacturer or subsequent BE measurements are corrected. corrected by following the procedure
outlined in this practice. For a routine check of the instrumental calibration, either one or two measurements are made for each
of the same core levels of copper and gold. Fig. 1 is a flow chart that summarizes the steps of the calibration procedure; references
are given to relevant sections of this standard.practice. An optional procedure is provided for determining the average energy of
the X-rays from a monochromated Al X-ray source, using a measured position of a copper Auger peak.
5. Significance and Use
5.1 X-ray photoelectron spectroscopy is used extensively for the surface analysis of materials. Elements (with the exception of
hydrogen and helium) are identified from comparisons of the binding energies determined from photoelectron spectra with
tabulated values. Information on chemical state can be derived from the chemical shifts of measured photoelectron and
Auger-electron features with respect to those measured for elemental solids.
5.2 Calibrations of the BE scales of XPS instruments are required for four principal reasons. First, meaningful comparison of
BE measurements from two or more XPS instruments requires that the BE scales be calibrated, often with an uncertainty of about
0.1 eV to 0.2 eV. Second, identification of chemical state is based on measurement of chemical shifts of photoelectron and
Auger-electron features, again with an uncertainty of typically about 0.1 eV to 0.2 eV; individual measurements, therefore, should
be made and literature sources need to be available with comparable or better accuracies. Third, the availability of databases (3)
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
https://www.iso.org/obp/ui/#iso:std:iso:18115:-1:ed-2:v1:en.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
E2108 − 16
TABLE 1 Definitions of Symbols and Abbreviations
a measured energy scaling error
BE binding energy, in eV
b measured zero offset error, in eV
c number of counts in the i-th channel
i
eV electron volts
E corrected result for the binding energy corresponding to a given E , in eV
corr meas
E binding energy of a frequently measured element at which the indicated binding energy scale is set, after
elem
calibration, to read correctly, in eV
E kinetic energy of a peak, with reference to the Fermi level, in eV
K
E a measured binding energy, in eV
meas
E average of the measured binding energies for the peak, n, in Table 3, in eV
meas n
E one of a set of measurements of binding energy for the peak, n, in Table 3, in eV
meas ni
E reference values for the position of peak, n, in Table 3, on the binding energy scale, in eV
ref n
E peak binding energy, in eV
p
E binding energy for first data channel at lower binding energy than the channel with the maximum number of counts,
for a peak, in eV
FWHM full width at half maximum peak intensity above the background, in eV
g channel energy separation, in eV
hv effective X-ray energy from an unmonochromated Al X-ray source, in eV
Al
mon
effective X-ray energy from a monochromated Al X-ray source, in eV
hv
Al
mon
effective X-ray energy from a monochromated Al X-ray source, in eV
hϑ
Al
hv effective X-ray energy from an unmonochromated Mg X-ray source, in eV
Mg
i index to represent channel number, where i = 0 represents the first channel at lower binding energy than the chan-
nel with the maximum number of counts, for a peak, in eV
j number of repeat measurements for a new peak
k number of repeat measurements for the Au 4f , Cu 2p and Ag 3d or Cu L VV peaks in the repeatability stan-
7/2 3/2 5/2 3
dard deviation and linearity determinations
m number of repeat measurements for the Au 4f and Cu 2p peaks in the regular calibrations
7/2 3/2
n designation of the peak identifier in Table 3
p parameter in Eq A1.1 and A1.1 defined in Eq A1.2 and A1.2
p parameter in Eq A1.1, defined in Eq A1.2 and Section A1.2
q parameter in Eq A1.1 and A1.1 defined in Eq A1.3 and A1.3
q parameter in Eq A1.1, defined in Eq A1.3 and Section A1.2
r parameter in Eq A1.1 and A1.1 defined in Eq A1.4 and A1.4
r parameter in Eq A1.1, defined in Eq A1.4 and Section A1.2
t Student’s t value for x degrees of freedom of a two-sided distribution for a confidence level of 95 %
x
t Student’s t value for x degrees of freedom of a two-sided distribution for a confidence level of 95 %
x
U total uncertainty of the calibrated energy scale at a confidence level of 95 %, in eV
c
uncertainty at a confidence level of 95 % arising from the calibration using the Au 4f and Cu 2p peaks at bind-
U (E) 7/2 3/2
ing energy E, assuming perfect scale linearity, in eV
uncertainty of ε or ε at a confidence level of 95 % from Eq 7 and 7, in eV
U 2 3
l
uncertainty of ε or ε at a confidence level of 95 % from Eq 7 and Section 8.9.3, in eV
U 2 3
cl
uncertainty of the calibration at a confidence level of 95 % in the absence of a linearity error, from Eq 12 and 12
U
and Eq 13 and 13, in eV
cl
uncertainty of the calibration at a confidence level of 95 % in the absence of a linearity error, from Eq 12 or Eq 13
U
and Section 8.10.4, in eV
XPS X-ray photoelectron spectroscopy
α Auger parameter, in eV
α' modified Auger parameter, in eV
Δ offset energy, given by the average measured binding energy for a calibration peak minus the reference energy, in
n
eV, for n = 1, 2, 3, 4 in Table 3, for a given X-ray source
ΔE correction to be added to E , after calibration, to provide the corrected result for the binding energy, in eV
corr meas
ΔE value of ΔE for peaks 1 and 4 in Table 3, in eV
corr n corr
ΔE drift of the binding-energy scale following a calibration for peaks 1 and 4 in Table 3, in eV
n
Δφ the average of Δ and Δ from Eq 16 and 16, in eV
1 4
Δφ the average of Δ and Δ from Eq 16 and Section 8.11.1.2, in eV
1 4
mon
Δhv
difference between hv and hv , in eV
Al Al
mon
Δhv
difference between hϑ and hv , in eV
Al Al
δ value for the tolerance limit of energy calibration at a confidence level of 95 % (set by the analyst), in eV
ε measured scale linearity error at the Ag 3d peak from Eq 4 and 4, in eV
2 5/2
ε measured scale linearity error at the Ag 3d peak from Eq 4 and Section 8.9.2, in eV
2 5/2
ε measured scale linearity error at the Cu L VV peak from Eq 5 and 5 or Eq 6 and 6, in eV
3 3
ε measured scale linearity error at the Cu L VV peak from Eq 5 or Eq 6 and Section 8.9.2, in eV
3 3
σ maximum of σ , σ or σ , and σ , in eV
R R1 R2 R3 R4
σ repeatability standard deviation for the seven measurements of the binding energy of peak, n, in Table 3, in eV
Rn
σ repeatability standard deviation for a new peak, in eV
Rnew
of measured BEs for reliable identification of elements and determination of chemical states by computer software requires that
published data and local measurements be made with uncertainties of about 0.1 eV to 0.2 eV. Finally, the growing adoption of
quality management systems, such as, ISO 9001:2000,9001:2015, in many analytical laboratories has led to requirements that the
measuring and test equipment be calibrated and that the relevant measurement uncertainties be known.
E2108 − 16
NOTE 1—The number associated with each step indicates the section in which the operation is described.
FIG. 1 Flowchart Indicating Sequence of Operations for the Calibration.
E2108 − 16
5.3 The actual uncertainty of a BE measurement depends on instrument properties and stability, measurement conditions, and
the method of data analysis. This practice makes use of tolerance limits 6δ (chosen, for example, at the 95 % confidence level)
that represent the maximum likely uncertainty of a BE measurement, associated with the instrument in a specified time interval
following a calibration (ISO 15472:2001).15472:2010). A user should select a value of δ based on the needs of the analytical work
to be undertaken, the likely measurement and data-analysis conditions, the stability of the instrument, and the cost of calibrations.
This practice gives information on the various sources of uncertainty in BE measurements and on measurements of instrumental
stability. The analyst should initially choose some desired value for δ and then make tests, as described in 8.14 to determine from
subsequent checks of the calibration whether BE measurements are made within the limits 6δ. Information is given in Appendix
X1 on how to evaluate for a material of interest the uncertainty of a BE measurement that is associated with the
E2108 − 16
uncertainty of the calibration procedure. This information is provided for four common analytical situations. It is important to note
that some BE measurements may have uncertainties larger than δ as a result of poor counting statistics, large peak widths,
uncertainties associated with peak synthesis,fitting, and effects of surface charging.
5.4 Instrument settings typically selected for analysis should be used with this practice. Separate calibrations should be made
if key operating conditions, such as choices of analyzer pass energy, aperture sizes, or X-ray source, are varied. Settings not
specified in this practice are at the discretion of the user, but those same settings should be recorded and consistently used whenever
this practice is repeated in order that the current results will be directly comparable to the previous results.
5.5 All of the operations described in Section 8 should be performed the first time that the BE scale is calibrated or after any
substantial modification of the instrument. For later checks of the calibration, to be performed on a regular schedule, only the
operations in 8.2 – 8.5, 8.10, 8.11, and 8.14 need to be performed. While the measurements described in 8.7 – 8.9 for the first
calibration require moderate time and effort, they are essential for ensuring that realistic tolerance limits 6δ have been chosen. The
control chart, described in 8.14, is a simple and effective means of demonstrating and documenting that the BE scale of the
instrument is in calibration, that is, within the tolerance limits, for a certain period of time.
5.6 The average energy of the X-rays incident on the specimen for instruments equipped with a monochromated Al X-ray source
will generally be slightly higher, by up to about 0.2 eV, than the average X-ray energy for instruments equipped with an
unmonochromated Al X-ray source (4). The actual energy difference depends on the alignment and thermal stability of the X-ray
monochromator. An optional procedure is given in Appendix X2 to determine this energy difference from measurements of the Cu
L VV Auger-electron peak. This information is needed for the determination of modified Auger parameters and Auger-electron
kinetic energies on instruments with the monochromated Al X-ray source.
6. Apparatus
6.1 X-ray Photoelectron Spectrometer—The X-ray photoelectron spectrometer is the instrument to be calibrated. The essential
components of the spectrometer are an X-ray source (preferably with an X-ray tube containing one or more anodes of aluminum,
or magnesium, or both, both), an ion gun for sample sputtering, a specimen holder that can hold three or more specimens, and an
electron energy analyzer with which photoelectron intensities are measured as a function of photoelectron binding energy. energy
(the analyzer collecting photoelectrons under a given angular range as specified in 6.4). Some instruments with an Al X-ray source
are equipped with an X-ray monochromator. monochromator, and also a charge compensation device. XPS instruments also
measure the intensities of Auger electrons that are emitted at characteristic energies. See Practice E902 for procedures to check
the operating characteristics of an X-ray photoelectron spectrometer and PracticeGuide E1016 for information on the properties
of electrostatic electron spectrometers.
6.2 Reference Specimens—The reference specimens, with which the spectrometer is calibrated, are polycrystalline foils of
high-purity (at least 99.8 % pure) copper and gold. For instruments with a monochromated Al X-ray source, a polycrystalline silver
film of similar purity is also required as a reference specimen. If desired, other high-purity metals can be used to check the linearity
of the BE scale throughout its range. See Guide E1078 for guidance on specimen preparation and mounting. The foil dimensions
should be chosen to be compatible with the specimen holder of the X-ray photoelectron spectrometer, and typically are 10 mm by
10 mm with a thickness of 0.2 mm.
6.3 Ion Gun—This practice requires use of an ion gun on the X-ray photoelectron spectrometer for sputter cleaning of the
reference specimens used for the calibration measurements.
6.4 Electron Emission Angle—The electron emission angle should be between 0° and 56° with respect to the surface normal (5).
The reference energies used in this practice are only valid for this range of emission angles.
7. Principle of the Calibration Method
7.1 For calibration of the XPS instrument with chosen instrumental settings, for example, X-ray source, analyzer pass energy,
energy step size, and apertures, measurements are made of the binding energies of the Au 4f and Cu 2p photoelectron peaks.
7/2 3/2
These peaks are chosen because they are near the low and high BE limits for many practical analyses, Au and Cu foils of sufficient
purity are readily available, the foil surfaces can be cleaned readily by ion sputtering, and the reference energies needed for the
calibration have been validated for a range of operating conditions (5). In an initial calibration of the instrument (or a calibration
made after any substantive instrumental modifications), a check of the BE-scale linearity is made with either the Ag 3d
5/2
photoelectron peak (with a monochromated Al X-ray source) or with the Cu L VV Auger-electron peak (with an unmonochro-
mated Al or Mg X-ray source). In this first calibration, a series of seven independent measurements are made for each of the Au
4f , Cu 2p and either Ag 3d or Cu L VV peaks (see 8.7). These measurements are made to determine the repeatability
7/2 3/2 5/2 3
standard deviations for measurements of the three peaks. These standard deviations have contributions from changes in outputs
of the spectrometer power supplies, from the sensitivity of the measured BE to sample position, and from counting statistics in
the recorded spectra (although the latter uncertainty is relatively small for the conditions specified in this practice). The magnitudes
of the other two contributions may vary with BE, and so an overall repeatability standard deviation σ is defined as the largest of
r
the repeatability standard deviations for the three peaks. The calibration will be valid only if the sample-positioning procedure used
for regular analyses is the same as the procedure used in the calibration.
E2108 − 16
7.2 It has been found in interlaboratory comparisons of XPS measurements that any measured error in measured BEs varies
approximately linearly with BE. This practice is based on the assumption that the differences between measured BEs and reference
BEs for the designated peaks are both small and linearly, or close to linearly, dependent on BE. A test of the validity of this
assumption is provided (see 8.7 and 8.9) by a measurement at an intermediate point on the BE scale using the Ag 3d
5/2
photoelectron peak (monochromated Al X-ray source) or the Cu L VV Auger peak (unmonochromated X-ray sources). The Ag
3d measurement is needed for monochromated Al X-ray sources because the average X-ray energy from this source may be
5/2
larger, by up to 0.2 eV, than for an unmonochromated Al X-ray source and, as a result, the relative energies of photoelectron and
Auger-electron peaks may vary by up to 0.2 eV (4). The Ag 3d peak could be used for the linearity check with unmonochromated
5/2
X-ray sources, if desired, but it will generally be simpler to make this check with the Cu L VV Auger peak because the copper
foil is already required for measurement of the Cu 2p peak. Additional checks of BE-scale linearity can be made, if desired, using
3/2
appropriate elemental metals and the corresponding recommended BEs as secondary standards (6).
7.3 If the BE scale is judged to be sufficiently linear (see 8.9), subsequent checks of the calibration can be performed with the
number m of measurements for each of the Au 4f and Cu 2p photoelectron peaks chosen to be 1 or 2, as described in 8.10.
7/2 3/2
An expression is given in this section for calculating the uncertainty U of the BE-scale calibration at the 95 % confidence level
(at the time of the calibration).
7.4 The means by which the BE scale is corrected following a calibration or a calibration check depends on practical details
of the instrument. Some methods for making this correction are described in 8.11.
7.5 The tolerance limit δ for the calibration at the 95 % confidence limit is an important parameter to be chosen by the analyst.
This parameter also is the tolerance limit, that is, the maximum allowed inaccuracy, again at the 95 % confidence limit, for
measured BEs for some defined time following a calibration. The chosen value of δ depends in part on the analytical requirements
and in part on the stability of the instrumental BE scale. Table 2 shows illustrative values for the various uncertainties that
contribute to U for values of δ of 0.1 eV and 0.2 eV and for values of m of 1 or 2. The difference between δ and U is the
95 95
maximum drift of the instrumental BE scale that should be allowed between calibration checks. Information on how to determine
the maximum interval between calibration checks is given in 8.13. It is strongly recommended that analysts prepare their own
version of Table 2 using numerical values from their measurements and the indicated equations and steps.
7.6 An analyst unfamiliar with this practice will have to make an initial choice for δ, and it is suggested that a value of 0.1 eV
be chosen. To decide whether the selected value of δ is realistic, this practice should be followed and a version of Table 2 should
be completed. In general, the value of δ should be based on the accuracy needs of the planned analytical work, the expected
measurement and data-analysis conditions, the linearity of the BE scale, the stability of the instrument, and a judgment based on
the needs and benefits of calibration with the chosen value of δ versus the cost in instrument and staff time in performing
calibrations. For example, if δ is chosen to be too small, calibration checks may have to be made too frequently to account for
possible drift of the BE scale in the interval between calibration checks. Other options in such a situation are to review the
operating procedures, for example, the sample-alignment procedure, or the calibration procedure, for example, the choice of m),
to determine if one or more of the terms contributing to U can be reduced. Otherwise, δ may need to be increased to an acceptable
value.
8. Calibration Procedure
8.1 Obtain the Reference Samples—Use gold and copper reference samples for XPS instruments with unmonochromated X-ray
sources. For instruments with a monochromated Al X-ray source, also use a silver sample on the first occasion that the instrument
is calibrated for the settings chosen in 8.4; for later determinations of the calibration error, as described in 8.10, use of the silver
sample is not necessary. The samples shall be polycrystalline and with a purity of at least 99.8 %. The samples can be foils of
typical dimensions 10 mm by 10 mm and with thicknesses between 0.1 mm and 0.2 mm.
NOTE 1—If the Cu and Ag samples appear discolored, a short dip in 1 % nitric acid may be used to clean the surfaces with subsequent rinsing in distilled
water. If the Cu sample has been stored in air for more than a few days, the cleaning with nitric acid will reduce the time needed for later sputter cleaning
(see 8.3.1).
8.2 Mount the Samples—Mount the Au and Cu samples and, if required, the Ag sample on the sample holder of the instrument
or on separate sample holders, as appropriate, using fixing screws or other metallic means to ensure electrical contact. Double-sided
adhesive tape should not be used.used when performing energy scale calibration work.
8.3 Clean the Samples:
8.3.1 Achieve ultra-high vacuum in the analytical chamber and clean the samples by ion sputtering until the heights of the
carbon and oxygen signals are each less than 2 % of the height of the most intense metal peak in a survey spectrum for each metal.
metal (if the silver sample is heavily contaminated it can also be pre-cleaned using a pencil eraser or metal polish). Record a survey
(widescan) spectrum for each sample to ensure that the only significant peaks are those expected for the particular metal. The
pressure of active gases in the analytical chamber should be low enough so that the heights of the carbon and oxygen 1 s peaks
shall not exceed 3 % of the height of the most intense metal peak by the time the measurements are completed (see 8.10) or at
the end of the working day, whichever is earlier.
E2108 − 16
TABLE 2 Contributions to an Error Budget for Calibration of the BE scale (from ISO 15472:2001)15472:2010)
NOTE 1—The uncertainties are for a 95 % confidence level. The numerical values shown are illustrations to indicate the effects of different choices (in
the values of δ and m) on the uncertainty of the calibration U and the required interval between calibrations for assumed values of σ , ε , or ε , and
95 r 2 3
the average drift rate of the BE scale.
Item Symbol Calculated from Examples
If you require a If you require a
high accuracy lower accuracy
Tolerance limits, eV ± δ You choose ± 0.1 ± 0.2 (Your choice is dictated by the
accuracy you require and the
number of spectra you have
time to acquire in regular cali-
brations.)
Tolerance limits, eV ± δ You choose ±0.1 ±0.2 (Your choice is dictated by the
accuracy you require and the
number of spectra you have
time to acquire in regular cali-
brations.)
Repeatability standard σ Eq 1 and 1 0.020 0.020 (Characteristic of your spec-
R
deviation, eV trometer measured at first cali-
bration (see 8.7)).
Repeatability standard σ Eq 1 and Sec- 0.020 0.020 (Characteristic of your spec-
R
deviation, eV tion 8.8.3 trometer measured at first cali-
bration (see 8.7)).
Number of times each m You choose m = 1 m = 2 m = 1 m = 2
pair of spectra is m= 1 or 2
acquired
Number of times each m You choose m = 1 m = 2 m = 1 m = 2
pair of spectra is m= 1 or 2
acquired
c1
Uncertainty of calibra- Eq 12 and 12 or 0.074 0.052 0.074 0.052
U
tion measurements, Eq 13 and 13
eV
cl
Uncertainty of calibra- Eq 12 or Eq 13 0.074 0.052 0.074 0.052
U
tion measurements, and Section
eV 8.10.4
Measure of scale non- ε or ε Eq 4 and 4, Eq 5 0.020 0.020 0.020 0.020 (Characteristic of your spec-
2 3
linearity, eV and 5 or trometer measured at first cali-
Eq 6 and 6 bration (see 8.7)).
Measure of scale non- ε or ε Eq 4 and Sec- 0.020 0.020 0.020 0.020 (Characteristic of your spec-
2 3
linearity, eV tion 8.9.2, Eq 5 trometer measured at first cali-
or bration (see 8.7)).
Eq 6 and Sec-
tion 8.9.2
Uncertainty of energy U Eq 11 and 11 0.078 0.057 0.078 0.057
scale after
calibration, eV
Uncertainty of energy U Eq 11 and Sec- 0.078 0.057 0.078 0.057
scale after tion 8.10.4
calibration, eV
Maximum allowable ± (δ-U ) δ and U ± 0.022 ± 0.043 ± 0.122 ± 0.143 (Define the drift allowable be-
95 95
drift between fore you are in danger of ex-
calibrations, eV ceeding your chosen limits ±δ
eV.)
Maximum allowable ± (δ-U ) δ and U ±0.022 ±0.043 ±0.122 ±0.143 (Define the drift allowable be-
95 95
drift between fore you are in danger of ex-
calibrations, eV ceeding your chosen limits ±δ
eV.)
Maximum calibration - Section 8.13 0.9 1.7 4.9 5.7 (Choose a convenient interval
interval (for a steady below this maximum, and less
drift rate of 0.025 eV than four months, with safety
per month), months margin for any erratic behav-
ior.)
Your choice of calibra- - You choose Option not 1 3 4 (No more than 70 % of the
tion interval, months based on practical maximum interval in the pre-
observed drift ceding row to allow a safety
behavior margin.)
E2108 − 16
NOTE 2—Suitable sputtering conditions with an inert gas are 1 min of a 30 μA beam of argon ions covering 1 cm of the surface.
NOTE 3—Illustrative survey spectra for Au, Ag, and Cu may be found in Refs (7-11).
8.3.2 It is desirable to complete the measurements in one working day. If a longer time is needed, additional surface cleaning
of the samples probably will be needed (see 8.3.1).
8.4 Choose the spectrometer settings for which energy calibration is required—Choose the operating settings of the
spectrometer for which energy calibration is required. The calibration procedure (see 8.4 – 8.13) shall be repeated for each X-ray
source and selected combination of pass energy, retardation ratio, apertures, lens settings, etc. for which a calibration is needed.
A calibration only is only valid for the particular X-ray source and operating conditions. Record the chosen operating settings.
NOTE 4—The designs of XPS instruments and their electronic controls vary considerably. As a result, a calibration made for a particular X-ray source
and a particular combination of pass energy, apertures, and lens settings will not necessarily be valid for another X-ray source or another combination
of spectrometer settings (4). Many analysts make all or most of their BE measurements for only one set of conditions; as a result, the instrument may
only need calibration for these conditions.
8.5 Operate the Instrument—Operate the instrument according to the manufacturer’smanufacturer’s documented instructions.
The instrument shall have fully cooled following any bakeout. Ensure that the X-ray source power, counting rates, spectral scan
rate, and other parameters are within the recommended ranges specified by the manufacturer. Check that the settings for the
multiplier detector have been adjusted correctly. For multidetector systems, ensure that any optimizations or checks recommended
by the manufacturer have been performed.
NOTE 5—Many manufacturers recommend that control and high-voltage electronics be switched on at least four hours before performing accurate BE
measurements. Also, it may be necessary to operate the X-ray source for some minimum period, for example, 1 h, before making such measurements.
XPS instruments with an X-ray monochromator may also need a warm-up time since the average X-ray energy at the sample may depend on the ambient
temperature or the temperature in the vicinity of the monochromator; records of these temperatures could help to identify any problems associated with
drifts of measured peak positions.
NOTE 6—If the counting rates are too high (12) or the detector voltages are set incorrectly (12, 13), spectral peaks can be distorted and the resulting
peak positions may be incorrect.
8.6 Options for Initial or Subsequent Calibration Measurements—For the first time that the instrument has been calibrated for
a particular combination of spectrometer settings (see 8.4), it is necessary to determine the repeatability standard deviation for BE
measurements, the BE-scale linearity error, and the calibration interval. If any of these have not been determined, follow 8.7 – 8.9;
otherwise, proceed to 8.10. If the instrument has been modified, undergone significant repair, or been moved, the steps in 8.7 –
8.9 should be followed.
E2108 − 16
8.7 Measurements for the Repeatability Standard Deviation and Scale Linearity:
8.7.1 The repeatability standard deviation for the peak BE, σ , is determined from repeated measurements of the Au 4f , Ag
R 7/2
3d or Cu L VV, and Cu 2p peaks as described in this section and in 8.8. The value of σ is valid only for the selected
5/2 3 3/2 R
conditions (see 8.4), and involves a significant contribution from the sample-positioning procedure (4, 5). A consistent
sample-positioning procedure, following a documented protocol, for example, from the manufacturer’smanufacturer’s
recommendations, should be followed both for the calibration measurements and for regular analytical work.
NOTE 7—The sample-positioning procedure will depend on the instrument design, the type and shape of the samples, and the analytical requirements.
In many cases, the correct sample position is determined by maximizing the spectral intensity. When optimization involves adjustment of two or more
interacting parameters, a consistent optimization strategy is necessary. If the optimization involves an X-ray monochromator, changes in the sample
position may lead to shifts in the energy of the recorded peak; thus, it may be necessary to measure peak intensities in an energy range of up to 60.5
eV from the nominal peak BE. In this case, intensity optimization may be more sensitive to sample position at low, rather than high, binding energies
or, more rarely, vice versa. Optimization is usually most effective at the BE where the intensity is most sensitive to sample position. It may be useful
to perform the operations in 8.7 several times to refine the sample-positioning procedure and to obtain a lower value of σ .
R
8.7.2 The BE-scale linearity error, ε or ε , is determined from measurements of the Cu L VV Auger-electron peak for
2 3 3
unmonochromated Mg or Al X-ray sources and from measurements of the Ag 3d photoelectron peak for a monochromated Al
5/2
X-ray source. These measurements are performed at the same time as the repeatability measurements to reduce the overall effort
and to reduce the measurement uncertainty.
8.7.3 The order of data acquisition for the measurements described in 8.7.4 – 8.7.7 should be as follows:
8.7.3.1 Unmonochromated Mg or Al X-rays: Au 4f , Cu 2p , Cu L VV, with this sequence repeated six additional timestimes.
7/2 3/2 3
8.7.3.2 Monochromated Al X-rays: Au 4f , Cu 2p , Ag 3d , with this sequence repeated six additional timestimes.
7/2 3/2 5/2
NOTE 8—The Au 4f peak is often the weakest peak although, depending on the spectrometer, sometimes the Cu L VV peak may be weaker. Initial
7/2 3
measurements with the Au 4f peak should facilitate the use of a common set of measurement conditions for all peaks.
7/2
8.7.4 Set the gold sample at the analytical position with the angle of emission for the detected electrons with respect to the
surface normal between 0° and 56°. Position the sample using the documented procedure and record the Au 4f peak with
7/2
appropriate X-ray power and channel dwell time so that more than 40 000 counts per channel are acquired in the vicinity of the
peak. Scan the peak with the channel energy interval set at approximately 0.05 eV or 0.1 eV depending on the method planned
for determination of the peak BE (see 8.8.1). Scan from at least 1 eV below the peak energy to at least 1 eV above the peak energy.
Ensure that the correct peak has been measured from the wide-scan (survey) spectrum (see 8.3.1). The reference BE for the Au
4f peak, peak 1, is given in Table 3.
7/2
NOTE 9—The reference BE values for the calibration peaks vary with angle of electron emission (5). The reference BE values in Table 3 are only valid
for emission angles between 0° and 56° (5). If larger emission angles are used, significant errors will be introduced into the calibration.
NOTE 10—A wide range of spectral scan rates are available on commercial XPS instruments. If the scan rate is too high, the measured peak BE will
be shifted. The scan rate should be chosen so that there is no significant peak shift.
8.7.5 Set the copper sample at the analytical position with the same emission angle as in 8.7.4. Position the sample with the
documented procedure and record the Cu 2p peak, with the same spectrometer settings as those used in 8.7.4, so that more than
3/2
40 000 counts per channel are acquired in the vicinity of the peak. Scan from at least 1 eV below the peak energy to at least 1 eV
above the peak energy. Ensure that the correct peak has been measured from the wide-scan (survey) spectrum (see 8.3.1). The
reference BE for the Cu 2p peak, peak 4, is given in Table 3.
3/2
8.7.6 If an unmonochromated Al or Mg X-ray source is being used, the Cu L VV peak will be measured next. If a
monochromated Al X-ray source is being used, set the silver sample at the analytical position with the same emission angle as in
8.7.4 and position the sample with the documented procedure. Record the Cu L VV peak or the Ag 3d peak, depending on X-ray
3 5/2
source, with the same spectrometer settings as those used in 8.7.4, so that more than 40 000 counts per channel are acquired in
the vicinity of the peak. Scan from at least 1 eV below the peak energy to at least 1 eV above the peak energy. Ensure that the
correct peak has been measured from the wide-scan (survey) spectrum (see 8.3.1). The reference BEs for the Ag 3d peak, peak
5/2
2, and the Cu L VV peak, peak 3, are given in Table 3.
TABLE 3 Reference Binding Energies, E for the Peaks Used
ref n
in the Calibration Procedure for the Indicated X-Ray Sources
(1, 2)
NOTE 1—The Ag data included in parentheses are not used normally
used in the calibration.
Peak Number, n Assignment E (eV)
ref n
Al Kα Mg Kα Monochro-
matic Al Kα
1 Au 4f 83.95 83.95 83.96
7/2
2 Ag 3d (368.22) (368.22) 368.21
5/2
3 Cu L VV 567.93 334.90 -
4 Cu 2p 932.63 932.62 932.62
3/2
E2108 − 16
8.7.7 Repeat the steps in 8.7.4 – 8.7.6, in order, a further an additional six times in order to obtain seven independent
measurements for each of the three peaks. To save time, the energy-scan widths may be reduced to the range 60.5 eV about the
peak unless a wider range is needed if instrumental software is to be used for peak location (see 8.8.1.2).
8.8 Calculate the Repeatability Standard Deviation for the Peak Binding Energies:
8.8.1 Determine the peak binding energies from the spectra recorded in 8.7 using one of the three methods described in 8.8.1.1,
8.8.1.2, or 8.8.1.3. (specify in the documentation which of the three methods is utilized).
NOTE 11—The first two methods are useful for instruments that supply digital data, the third method for instruments that supply only graphical output.
8.8.1.1 Make a least–squares fit of a parabola to six data points around the top of the peak. There should be approximately the
same number of data points above and below the energy for the maximum intensity, and the intensities for the first and last data
points should be between 85 % and 95 % of the maximum intensity. Obtain the energy for the peak maximum from this fit. If
software for this purpose is not conveniently available, the simple numerical procedure given in Annex A1 can be used.
NOTE 12—If the procedure in Annex A1 is utilized, a channel energy separation of 0.1 eV (or in the range 0.09 eV to 0.11 eV) should be selected for
XPS with an unmonochromated X-ray source. For XPS with a monochromated Al X-ray source, the channel separation should be 0.05 eV (or in the range
of 0.045 eV to 0.055 eV) if the FWHM of the peaks is less than 1.0 eV; otherwise, a channel width between 0.9 eV and 0.11 eV can be used.
NOTE 13—If the procedure in Annex A1 is utilized, the intensities for the first and last data points could, if necessary, be between 80 % and 95 % of
the maximum intensity. There would then be a slight increase (up to 6 meV) in the uncertainty of the derived peak binding energy.
8.8.1.2 Make a least-squares fit to the data points obtained in 8.8.1.1 with a suitable lineshape function available with software
on the XPS instrument or on another computer. A Gaussian function, a Lorentzian function, a Voigt fun
...



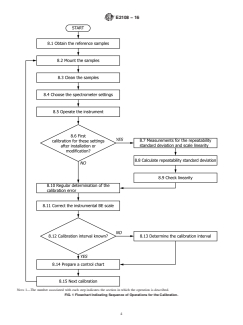



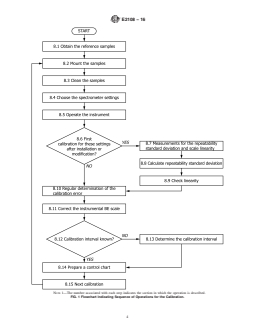
Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...