ASTM C1679-22
(Practice)Standard Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry
Standard Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry
SIGNIFICANCE AND USE
5.1 Thermal power curves are used to evaluate the isothermal hydration kinetics of the combined mixture of different materials during the early period after being mixed with water. These isothermal power curves, or hydration profiles, may provide indications relative to setting characteristics, compatibility of different materials, sulfate balance and early strength development. The isothermal hydration profiles can also be used to evaluate the effects of compositions, proportions, and time of addition of materials as well as curing temperature. Special care must be used in evaluating extended retardation with paste specimens, which have been shown to overestimate the retardation of some mixtures containing cement, SCM, and admixtures.
5.2 This procedure can be used to measure the effect of chemical admixtures on the cement hydration profile. In many cases, the addition of chemical admixture changes the kinetics of cement hydration.
5.3 Although this technique has been used historically to understand issues related to setting and slump loss, it must be emphasized that isothermal calorimetry results cannot predict concrete performance definitely, either positively or negatively. Extensive verification in concrete at planned dosages and temperatures, and at higher dosages, is needed. Isothermal calorimetry is an effective tool to identify sensitivities, so that concrete testing can be efficiently planned and performed.
5.4 This practice provides a means of assessing the relative hydration performance of various test mixtures compared with control mixtures that are prepared in a similar manner.
5.5 The procedure and apparatus can be used to monitor the thermal power from pastes and mortars alone or in combination with chemical admixtures.
5.6 The isothermal calorimeter described here can be used to measure the thermal power and heat of hydration of mortars prepared independently or obtained by wet sieving from concrete in accordance with Practice C172/C1...
SCOPE
1.1 This practice describes the apparatus and procedure for measuring relative differences in hydration kinetics of hydraulic cementitious mixtures, either in paste or mortar (see Note 1), including those containing admixtures, various supplementary cementitious materials (SCM), and other fine materials by measuring the thermal power using an isothermal calorimeter.
Note 1: Paste specimens are often preferred for mechanistic research when details of individual reaction peaks are important or for particular calorimetry configurations. Mortar specimens may give results that have better correlation with concrete setting and early strength development and are often preferred to evaluate different mixture proportions for concrete. Both paste and mortar studies have been found to be effective in evaluating concrete field problems due to incompatibility of materials used in concrete mixtures.
1.2 This practice does not cover the measurement of heat of hydration. Heat of hydration can be determined according to Test Method C1702.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (Warning—Fresh hydraulic cementitious mixtures are caustic and may cause chemical burns to skin and tissue upon prolonged exposure.2)
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Dec-2022
- Technical Committee
- C09 - Concrete and Concrete Aggregates
Relations
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-Aug-2020
- Effective Date
- 15-Jul-2020
- Effective Date
- 01-Apr-2020
- Effective Date
- 15-Dec-2019
- Effective Date
- 15-Nov-2019
- Effective Date
- 01-Jun-2019
- Effective Date
- 01-May-2019
- Effective Date
- 01-Jan-2019
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Jul-2018
- Effective Date
- 01-May-2018
- Effective Date
- 01-May-2018
- Effective Date
- 01-Jan-2018
- Effective Date
- 15-Mar-2017
Overview
ASTM C1679-22: Standard Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry provides a standardized approach for assessing the hydration behavior of hydraulic cement mixtures. Issued by ASTM, this practice describes the apparatus and procedures required to measure the relative differences in hydration kinetics for cement pastes and mortars, including those containing supplementary cementitious materials (SCM) and chemical admixtures. Isothermal calorimetry offers critical insights into cementitious system behavior, especially in the early stages after mixing with water, aiding in the evaluation of setting characteristics, compatibility, sulfate balance, and early strength development.
Key Topics
Isothermal Hydration Profiles: Isothermal calorimetry generates thermal power curves that map the progress of cement hydration over time. These profiles are invaluable for analyzing early-age behavior, revealing key information about mixture setting, compatibility of materials, and sulfate balance.
Assessment of Admixture Effects: The practice outlines how changes in admixture type or dosage can significantly affect hydration kinetics. By measuring the thermal output, users can assess retarding or accelerating impacts, informing material selection and mix design.
Mixture Comparisons: ASTM C1679-22 enables side-by-side comparison of different test mixtures-such as control blends versus those with SCMs or admixtures-to identify sensitivities and optimize formulations.
Monitoring and Evaluation: The standard involves careful monitoring using calibrated isothermal calorimeters and repeatable specimen preparation. It also provides guidance on measuring and interpreting thermal equilibrium and repeatability in results.
Safety and Compliance: Special attention is given to safety and handling, as fresh cementitious mixtures are caustic. The document also aligns with internationally recognized standards principles, ensuring global applicability.
Applications
Isothermal calorimetry as standardized in ASTM C1679-22 is widely used in laboratory, quality control, and research settings within the cement, concrete, and construction materials industries. Typical applications include:
Evaluating New Mix Designs: Quickly assess the early-age kinetics of paste, mortar, or mortar obtained from concrete, especially when using novel SCMs or admixtures. This helps in selecting optimal proportions for performance and durability.
Troubleshooting Material Compatibility: Identify incompatibilities between cements, admixtures, and SCMs that may affect setting time, strength development, or sulfate balance, preventing field issues.
Performance Optimization: Understand the influence of temperature, mixing procedure, and order of addition to optimize field performance, setting, and strength gain.
Predictive Testing: While calorimetry does not directly predict concrete performance, it is useful for planning confirmatory concrete tests, reducing trial-and-error and shortening product development cycles.
Sulfate Balance Assessment: Investigate sulfate demand in mixtures to avoid premature setting or extended retardation, supporting robust mix formulations for various field conditions.
Related Standards
ASTM C1679-22 refers to and is complemented by several related ASTM standards, providing a comprehensive framework for cementitious materials testing:
- ASTM C1702: Measurement of Heat of Hydration using Isothermal Conduction Calorimetry
- ASTM C125, C219: Terminology for Concrete, Aggregates, and Cements
- ASTM C305: Mixing of Hydraulic Cement Pastes and Mortars
- ASTM C403: Time of Setting by Penetration Resistance
- ASTM C778: Standard Sand Specification
- ASTM C1602: Mixing Water for Hydraulic Cement Concrete
- ASTM C511: Requirements for Mixing Rooms and Storage Environments
Professionals working with cementitious materials should reference these documents alongside ASTM C1679-22 for comprehensive quality assurance and testing protocols.
Keywords: hydration kinetics, cement hydration, isothermal calorimetry, admixture compatibility, sulfate balance, ASTM C1679, cementitious mixtures, supplementary cementitious materials (SCM), construction materials testing, setting characteristics
Buy Documents
ASTM C1679-22 - Standard Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry
REDLINE ASTM C1679-22 - Standard Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry
Get Certified
Connect with accredited certification bodies for this standard

ICC Evaluation Service
Building products evaluation and certification.

QAI Laboratories
Building and construction product testing and certification.

Aboma Certification B.V.
Specialized in construction, metal, and transport sectors.
Sponsored listings
Frequently Asked Questions
ASTM C1679-22 is a standard published by ASTM International. Its full title is "Standard Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry". This standard covers: SIGNIFICANCE AND USE 5.1 Thermal power curves are used to evaluate the isothermal hydration kinetics of the combined mixture of different materials during the early period after being mixed with water. These isothermal power curves, or hydration profiles, may provide indications relative to setting characteristics, compatibility of different materials, sulfate balance and early strength development. The isothermal hydration profiles can also be used to evaluate the effects of compositions, proportions, and time of addition of materials as well as curing temperature. Special care must be used in evaluating extended retardation with paste specimens, which have been shown to overestimate the retardation of some mixtures containing cement, SCM, and admixtures. 5.2 This procedure can be used to measure the effect of chemical admixtures on the cement hydration profile. In many cases, the addition of chemical admixture changes the kinetics of cement hydration. 5.3 Although this technique has been used historically to understand issues related to setting and slump loss, it must be emphasized that isothermal calorimetry results cannot predict concrete performance definitely, either positively or negatively. Extensive verification in concrete at planned dosages and temperatures, and at higher dosages, is needed. Isothermal calorimetry is an effective tool to identify sensitivities, so that concrete testing can be efficiently planned and performed. 5.4 This practice provides a means of assessing the relative hydration performance of various test mixtures compared with control mixtures that are prepared in a similar manner. 5.5 The procedure and apparatus can be used to monitor the thermal power from pastes and mortars alone or in combination with chemical admixtures. 5.6 The isothermal calorimeter described here can be used to measure the thermal power and heat of hydration of mortars prepared independently or obtained by wet sieving from concrete in accordance with Practice C172/C1... SCOPE 1.1 This practice describes the apparatus and procedure for measuring relative differences in hydration kinetics of hydraulic cementitious mixtures, either in paste or mortar (see Note 1), including those containing admixtures, various supplementary cementitious materials (SCM), and other fine materials by measuring the thermal power using an isothermal calorimeter. Note 1: Paste specimens are often preferred for mechanistic research when details of individual reaction peaks are important or for particular calorimetry configurations. Mortar specimens may give results that have better correlation with concrete setting and early strength development and are often preferred to evaluate different mixture proportions for concrete. Both paste and mortar studies have been found to be effective in evaluating concrete field problems due to incompatibility of materials used in concrete mixtures. 1.2 This practice does not cover the measurement of heat of hydration. Heat of hydration can be determined according to Test Method C1702. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (Warning—Fresh hydraulic cementitious mixtures are caustic and may cause chemical burns to skin and tissue upon prolonged exposure.2) 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Thermal power curves are used to evaluate the isothermal hydration kinetics of the combined mixture of different materials during the early period after being mixed with water. These isothermal power curves, or hydration profiles, may provide indications relative to setting characteristics, compatibility of different materials, sulfate balance and early strength development. The isothermal hydration profiles can also be used to evaluate the effects of compositions, proportions, and time of addition of materials as well as curing temperature. Special care must be used in evaluating extended retardation with paste specimens, which have been shown to overestimate the retardation of some mixtures containing cement, SCM, and admixtures. 5.2 This procedure can be used to measure the effect of chemical admixtures on the cement hydration profile. In many cases, the addition of chemical admixture changes the kinetics of cement hydration. 5.3 Although this technique has been used historically to understand issues related to setting and slump loss, it must be emphasized that isothermal calorimetry results cannot predict concrete performance definitely, either positively or negatively. Extensive verification in concrete at planned dosages and temperatures, and at higher dosages, is needed. Isothermal calorimetry is an effective tool to identify sensitivities, so that concrete testing can be efficiently planned and performed. 5.4 This practice provides a means of assessing the relative hydration performance of various test mixtures compared with control mixtures that are prepared in a similar manner. 5.5 The procedure and apparatus can be used to monitor the thermal power from pastes and mortars alone or in combination with chemical admixtures. 5.6 The isothermal calorimeter described here can be used to measure the thermal power and heat of hydration of mortars prepared independently or obtained by wet sieving from concrete in accordance with Practice C172/C1... SCOPE 1.1 This practice describes the apparatus and procedure for measuring relative differences in hydration kinetics of hydraulic cementitious mixtures, either in paste or mortar (see Note 1), including those containing admixtures, various supplementary cementitious materials (SCM), and other fine materials by measuring the thermal power using an isothermal calorimeter. Note 1: Paste specimens are often preferred for mechanistic research when details of individual reaction peaks are important or for particular calorimetry configurations. Mortar specimens may give results that have better correlation with concrete setting and early strength development and are often preferred to evaluate different mixture proportions for concrete. Both paste and mortar studies have been found to be effective in evaluating concrete field problems due to incompatibility of materials used in concrete mixtures. 1.2 This practice does not cover the measurement of heat of hydration. Heat of hydration can be determined according to Test Method C1702. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (Warning—Fresh hydraulic cementitious mixtures are caustic and may cause chemical burns to skin and tissue upon prolonged exposure.2) 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM C1679-22 is classified under the following ICS (International Classification for Standards) categories: 91.100.10 - Cement. Gypsum. Lime. Mortar. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C1679-22 has the following relationships with other standards: It is inter standard links to ASTM C219-24, ASTM C219-20a, ASTM C305-20, ASTM C219-20, ASTM C125-19a, ASTM C219-19a, ASTM C219-19, ASTM C1738/C1738M-19, ASTM C125-19, ASTM C125-18b, ASTM C125-18a, ASTM C1005-18, ASTM C1738/C1738M-18, ASTM C125-18, ASTM C1005-17. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C1679-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: C1679 − 22
Standard Practice for
Measuring Hydration Kinetics of Hydraulic Cementitious
Mixtures Using Isothermal Calorimetry
This standard is issued under the fixed designation C1679; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
1.1 This practice describes the apparatus and procedure for 2.1 ASTM Standards:
measuring relative differences in hydration kinetics of hydrau- C125 Terminology Relating to Concrete and Concrete Ag-
lic cementitious mixtures, either in paste or mortar (see Note gregates
1), including those containing admixtures, various supplemen- C172/C172M Practice for Sampling Freshly Mixed Con-
tary cementitious materials (SCM), and other fine materials by crete
measuring the thermal power using an isothermal calorimeter. C219 Terminology Relating to Hydraulic and Other Inor-
NOTE 1—Paste specimens are often preferred for mechanistic research
ganic Cements
when details of individual reaction peaks are important or for particular
C305 Practice for Mechanical Mixing of Hydraulic Cement
calorimetry configurations. Mortar specimens may give results that have
Pastes and Mortars of Plastic Consistency
better correlation with concrete setting and early strength development
C403/C403M Test Method for Time of Setting of Concrete
and are often preferred to evaluate different mixture proportions for
Mixtures by Penetration Resistance
concrete. Both paste and mortar studies have been found to be effective in
evaluating concrete field problems due to incompatibility of materials
C511 Specification for Mixing Rooms, Moist Cabinets,
used in concrete mixtures.
Moist Rooms, and Water Storage Tanks Used in the
1.2 This practice does not cover the measurement of heat of
Testing of Hydraulic Cements and Concretes
hydration. Heat of hydration can be determined according to C778 Specification for Standard Sand
Test Method C1702.
C1005 Specification for Reference Masses and Devices for
Determining Mass and Volume for Use in Physical Testing
1.3 The values stated in SI units are to be regarded as
of Hydraulic Cements
standard. No other units of measurement are included in this
C1602/C1602M Specification for Mixing Water Used in the
standard.
Production of Hydraulic Cement Concrete
1.4 This standard does not purport to address all of the
C1702 Test Method for Measurement of Heat of Hydration
safety concerns, if any, associated with its use. It is the
of Hydraulic Cementitious Materials Using Isothermal
responsibility of the user of this standard to establish appro-
Conduction Calorimetry
priate safety, health, and environmental practices and deter-
C1738/C1738M Practice for High-Shear Mixing of Hydrau-
mine the applicability of regulatory limitations prior to use.
lic Cement Pastes
(Warning—Fresh hydraulic cementitious mixtures are caustic
and may cause chemical burns to skin and tissue upon
3. Terminology
prolonged exposure. )
3.1 Definitions—For definitions of terms used in this
1.5 This international standard was developed in accor-
practice, refer to Terminology C125 and Terminology C219.
dance with internationally recognized principles on standard-
3.2 Definitions of Terms Specific to This Standard:
ization established in the Decision on Principles for the
3.2.1 baseline, n—the signal from the calorimeter when
Development of International Standards, Guides and Recom-
there is an inert specimen in the instrument.
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
3.2.2 calcium aluminate, n—various aluminate phases in-
cluding but not limited to the tricalcium aluminate and ferrite
phases in portland cement clinker, calcium aluminate phases
This practice is under the jurisdiction of ASTM Committee C09 on Concrete
occurring in some supplementary cementitious materials, and
and Concrete Aggregates and is the direct responsibility of Subcommittee C09.48 on
Performance of Cementitious Materials and Admixture Combinations.
Current edition approved Dec. 15, 2022. Published February 2023. Originally
approved in 2007. Last previous edition approved in 2017 as C1679 – 17. DOI: For referenced ASTM standards, visit the ASTM website, www.astm.org, or
10.1520/C1679-22. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Section on Safety Precautions, Manual of Aggregate and Concrete Testing, Standards volume information, refer to the standard’s Document Summary page on
Annual Book of ASTM Standards, Vol 04.02. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1679 − 22
calcium-alumino-silicate glasses also occurring in some 3.2.10 main hydration peak, n—the broadest peak in the
supplementary cementitious materials, that are capable of isothermal hydration profile that starts at the end of the
consuming the sulfate phases present in hydrating cementitious dormant period and for a well-balanced mixture lasts for
systems. several hours (see Fig. 1).
3.2.3 calibration coeffıcient, n—a factor that relates the
3.2.11 near isothermal conditions, n—a constant tempera-
value recorded by the data acquisition system to the thermal ture with a permissible variation of 6 1.0 °C.
power output.
3.2.12 specimen holder, n—container within the isothermal
3.2.3.1 Discussion—Normally recorded data are in volts and
calorimeter that conducts the heat from the specimen in the vial
the calibration coefficient has units of watts per volt (W/V).
to the heat flow sensor.
Some calorimeters may have internal automatic calibration and
3.2.13 stock solution, n—a solution of admixture in water
will give the output in watts without the user having to specify
prepared to enable more precise volumetric addition of small
the calibration coefficient.
quantities of admixture, typically made by pipetting known
3.2.4 combined mixture, n—combination of all the materials
volumes of admixture into a volumetric flask and diluting it to
that are introduced into the calorimeter for measuring hydra-
the flask’s fixed volume.
tion kinetics.
3.2.14 sulfate addition, n—the addition of a soluble sulfate
3.2.5 hydration time, n—the elapsed time from initial con-
source (such as gypsum, calcium sulfate hemihydrate, alkali
tact between the cementitious materials and the mix water.
sulfate) to a combined mixture to investigate whether a given
3.2.6 inert specimen, n—specimen placed within the iso-
combination of materials is in sulfate balance.
thermal calorimeter made of a non-reactive material of similar
3.2.15 sulfate balance of mixture, n—the situation when the
thermal properties (mainly heat capacity) as the reacting
size of the main hydration peak is not increased by sulfate
specimen made of the cementitious test mixture.
additions; in some cases where the main peak is increased in
3.2.6.1 Discussion—The output from the calorimeter is the
size by added sulfate, it will also be accelerated in time.
difference between the heat flow from the test specimen and the
3.2.16 sulfate depletion point, n—the onset of accelerated
inert specimen. The use of an inert specimen substantially
calcium aluminate activity that for a portland cement in
decreases the noise and drift of the measured heat flow.
absence of supplementary cementitious material (SCM) and
3.2.7 isothermal calorimeter, n—a calorimeter that mea-
admixture may take place after the main hydration peak.
sures heat flow from a specimen maintained at a constant
3.2.16.1 Discussion—The sulfate depletion point may be-
temperature by intimate thermal contact with a constant
come impossible to detect without further addition of soluble
temperature heat sink.
calcium sulfate for certain cements and more often in com-
3.2.8 isothermal calorimetry, n—an experimental technique
bined mixtures with admixtures or SCMs, or both. In some
to monitor the thermal power output from a specimen kept at
cases other sources of sulfate might be used to mimic potential
near isothermal conditions.
conditions in the system. Among these are anhydrite, arcanite,
3.2.9 isothermal hydration profile, n—the thermal power calcium langbeinite, aphthitalite, syngenite, and others. Fig. 2
plotted as a function of hydration time, which provides an shows an example of the effect of added sulfate on the sulfate
indication of the rate of hydration over time at a given depletion point. Added sulfate may, in some combined mix-
temperature. tures with admixtures or SCMs, or both, accelerate the onset of
NOTE 1—(A) initial thermal power by dissolution of cement and initial cement hydration; (B) dormant period associated with very low thermal power
indicating slow and well-controlled hydration: (C) main hydration peak associated mainly with hydration reactions contributing to setting and early
strength development, with maximum at (D); and (E) sulfate depletion point, followed by (F) accelerated calcium aluminate activity.
FIG. 1 Example of Thermal Power Curve for Isothermal Hydration of Portland Cement
C1679 − 22
FIG. 2 Example of the Effect of Soluble Calcium Sulfate Addition on the Timing of the Sulfate Depletion Point for a Type I Portland Ce-
ment Mixed with Water Only at w/c = 0.45
the main hydration peak. When a combined mixture is at men is transferred and passes across a heat flow sensor. The
sulfate balance, further addition of soluble sulfate will not calorimeter output is calculated from the difference between
increase the size, or accelerate the onset, of the main hydration the outputs from the test specimen heat flow sensor and the
peak. inert specimen heat flow sensor. Because the heat is allowed to
flow away from the specimen, the measurement will take place
3.2.17 thermal equilibrium time, n—the elapsed hydration
at essentially constant temperature (isothermal conditions).
time when the thermal power of replicate mixtures do not differ
by more than 0.2 mW/g of dry material.
4.2 Mixtures with cement, SCM, admixtures, water and
optional fine aggregate are prepared and introduced into an
3.2.18 thermal indicator of setting time, n— the hydration
isothermal calorimeter. Isothermal calorimetry tests are per-
time to reach a thermal power of 50 % of the maximum value
formed on a series of different mixtures for relative comparison
of the main hydration peak.
of the hydration kinetics. The output of the calorimeter is
3.2.19 thermal mass, n—the amount of thermal energy that
evaluated by graphical and mathematical means to evaluate
can be stored by a material (J/K).
retarding and accelerating effects of different combinations of
3.2.19.1 Discussion—The thermal mass of a given material
materials. Calcium sulfate may be added as a probe to
is calculated by multiplying the mass by the specific heat
determine if the addition of admixture, SCMs, or both have
capacity of the material. For the purpose of calculating the
increased the mixture’s demand for sulfate beyond that which
thermal mass used in this standard, the following specific heat
is available in the cement.
capacities can be used: The specific heat capacity of a typical
unhydrated portland cement and water is 0.75 and 4.18 J/(g·K),
5. Significance and Use
respectively. Thus a mixture of A g of cement and B g of water
5.1 Thermal power curves are used to evaluate the isother-
has a thermal mass of (0.75 × A + 4.18 × B) J/K. The specific
mal hydration kinetics of the combined mixture of different
heat capacity of typical quartz and limestone is 0.75 and 0.84
materials during the early period after being mixed with water.
J/(g·K), respectively. The specific heat capacity of most amor-
These isothermal power curves, or hydration profiles, may
phous supplementary cementitious material such as fly ash or
provide indications relative to setting characteristics, compat-
slag is approximately 0.8 J/(g·K).
ibility of different materials, sulfate balance and early strength
3.2.20 thermal power, n—heat production rate measured in
development. The isothermal hydration profiles can also be
watts (W) or joules per second (J/s), usually expressed in
used to evaluate the effects of compositions, proportions, and
relation to the mass of cementitious material, as mW/g or J/s/g.
time of addition of materials as well as curing temperature.
3.2.20.1 Discussion—The thermal power is an indicator of
Special care must be used in evaluating extended retardation
the rate of various chemical reactions between cementitious
with paste specimens, which have been shown to overestimate
materials, other fine particles, mix water and admixtures.
the retardation of some mixtures containing cement, SCM, and
3.2.21 vial, n—container into which the freshly mixed
admixtures.
cementitious mixture is placed for a measurement.
5.2 This procedure can be used to measure the effect of
4. Summary of Practice chemical admixtures on the cement hydration profile. In many
cases, the addition of chemical admixture changes the kinetics
4.1 An isothermal calorimeter consists of heat sink with a
of cement hydration.
thermostat, two heat flow sensors and a specimen vial holder
attached to each sensor. A vial containing a freshly prepared 5.3 Although this technique has been used historically to
mixture is placed in contact with one of the vial holders and a understand issues related to setting and slump loss, it must be
thermally inert material is placed in contact with the other. The emphasized that isothermal calorimetry results cannot predict
heat of hydration released by the reacting cementitious speci- concrete performance definitely, either positively or negatively.
C1679 − 22
Extensive verification in concrete at planned dosages and may vary, but it shall meet the following criteria, and shall be
temperatures, and at higher dosages, is needed. Isothermal verified to do so on a yearly basis or at any time that equipment
calorimetry is an effective tool to identify sensitivities, so that is modified.
concrete testing can be efficiently planned and performed. 6.2.1 The standard deviation in thermal power for six
replicate specimens sampled from one mixture of a portland
5.4 This practice provides a means of assessing the relative
cement paste without chemical admixture at w/c 0.45 tested at
hydration performance of various test mixtures compared with
23 °C 6 4.0 °C shall be no greater than 0.5 mW/g of dry
control mixtures that are prepared in a similar manner.
material measured at the maximum of the main hydration peak
5.5 The procedure and apparatus can be used to monitor the
(see Fig. 1 for identification of the main hydration peak). For
thermal power from pastes and mortars alone or in combination
calorimeters which do not allow external mixing, prepare six
with chemical admixtures.
replicate mixtures.
6.2.2 The baseline measured with an inert specimen of
5.6 The isothermal calorimeter described here can be used
similar thermal mass as the test specimen shall exhibit a low
to measure the thermal power and heat of hydration of mortars
random noise level and be stable against drift. The baseline
prepared independently or obtained by wet sieving from
measured at 23 °C 6 4 °C during a time period of 24 h shall
concrete in accordance with Practice C172/C172M.
have a long-term drift of less than 0.5 mW per day per gram of
inert material used and a baseline random noise level of less
6. Apparatus
than 0.2 mW per gram of inert material used. In practice, the
6.1 Devices for mixing to produce a homogeneous mixture
baseline is measured for 24 h or longer and a straight line is
of cement, SCM, admixtures, water and optional other fine
fitted to the data using a linear least squares procedure. The
materials or aggregate and devices for charging the mixture
long-term drift is the slope of the line and the baseline random
into the specimen vial. noise level is the standard deviation of the data about the
best-fit line.
6.1.1 Weights and Weighing Devices shall conform to the
requirements of Specification C1005. 6.2.3 The vials shall be vapor tight so that endothermic heat
of evaporation does not significantly influence the measure-
6.1.2 Graduated Cylinders shall conform to the require-
ments. This shall be verified with a 24 h measurement with
ments of Specification C1005. The permissible variation for
water instead of the mortar or paste specimen. The difference
graduated cylinders of less than 100-mL capacity shall be
between the measured thermal powers of an empty vial and one
61.0 % of the rated capacity.
containing water shall be less than 0.2 mW per gram of water
6.1.3 Graduated Syringes of suitable capacities to contain
when calculated according to the procedure in 13.1.
the desired volume of liquid admixture or stock solution at
6.2.4 The data acquisition equipment shall be capable of
20 °C. The permissible variation of the measured volume shall
performing continuous logging of the calorimeter output with
not exceed 3 % of the volume to be delivered. When admixture
a time interval between the measurements that is no larger than
quantities required are less than 2 mL, or are viscous in nature,
60 s.
optionally prepare stock solutions at appropriate dilution, to
avoid problems with small volumes measured volumetrically.
7. Materials
Care shall be taken to inspect stock solutions for separation and
any admixture that is prone to separation in stock solution must
7.1 Sand:
be added in an alternative fashion, such as by analytical
7.1.1 Unless specified otherwise, use standard graded sand
syringes.
as defined in Specification C778 for preparing mortar samples.
6.1.4 Mixing Apparatus:
7.1.2 When specified, use job-specific sand when perform-
6.1.4.1 Mortar Preparation—The mixer shall comply with ing a specific mortar test series that is related to field
Practice C305.
application.
6.1.4.2 Paste Preparation—A high shear mixer as described
7.2 Water:
in Practice C1738/C1738M. A handheld household mixer
7.2.1 Unless specified, use potable water as defined in
capable of mixing paste at not less than 400 r/min or other
Specification C1602/C1602M.
mixers for paste or mortar preparation are also permitted.
7.2.2 When specified, use job-specific water when perform-
6.1.4.3 The repeatability of results for any mixing method
ing a specific test series that is related to field application.
must be measured and reported as described in 12.5.
Follow Specification C1602/C1602M in reporting the type of
6.1.5 Vials that can be sealed and fit into the specimen vial
water used.
holders of the calorimeter.
7.3 Sulfate Source:
6.1.6 Pipette, Funnel or Syringe to facilitate charging the
7.3.1 Unless specified otherwise, use reagent grade calcium
sample to the vials. The neck of the device must be large
sulfate dihydrate or hemihydrate prepared from reagent grade
enough to avoid sample segregation.
calcium sulfate dihydrate to verify whether a mixture is in
sulfate balance. See Appendix X6 for examples.
6.2 Calorimeter and Data Acquisition System of suitable
capacity and calibrated to monitor the thermal power of mortar 7.3.2 When specified, use a source-specific calcium sulfate
or paste in a repeatable fashion. The actual design of an for performing a specific test series that is related to field
individual instrument, whether commercial or custom-built, application.
C1679 − 22
8. Temperature and Humidity the noise. If baselines are stable over time as defined in 6.2.3,
they need only be determined at calibrations. If they are not
8.1 Sample Preparation Temperature—Unless specified
stable, they shall be determined before each measurement.
otherwise, maintain the temperature of the air in the vicinity of
all equipment and materials used in accordance with the 11.2 Calibration—Calibration shall be made according to
requirements for cement mixing rooms in Specification C511. manufacturer’s recommendations at regular intervals of one
8.1.1 When specified, different temperature conditions are year or less to determine the calibration coefficient. If the
permitted for research purposes or to simulate field conditions. calibration coefficient differs more than 5 % from one calibra-
8.1.2 To reduce the time to reach thermal equilibrium, it is tion to the next, calibration intervals shall be reduced to 90
permitted to cool the mixture during mixing. It is also days. The length of the time intervals between calibrations
permitted to adjust the temperature of the mix water or the depends on the instrument and the personnel. For the purpose
starting materials, or both, in order to obtain a temperature of of this practice, the calibration coefficient can be assumed to be
the prepared mixture close the temperature of the calorimeter. constant independent of the level of the signal.
This is especially needed when using high shear mixing.
12. Procedure
8.2 Isothermal Calorimeter Test Temperature—The tem-
12.1 Mixing:
perature of the isothermal calorimeter shall be set at
12.1.1 Various mixing methods that have been found useful
23.0 °C 6 4.0 °C and allowed sufficient time to stabilize at that
are described in the Appendix. Because mixing intensity is a
temperature.
variable that may influence the interaction of materials used to
8.2.1 When specified, a different calorimeter test tempera-
prepare cementitious mixtures, in many cases different mixing
ture is permitted for research purposes or to simulate field
procedures (speeds or durations) may be needed, depending on
conditions. Take precaution to prevent condensation of mois-
the goal of the testing. Unless mixing intensity is a defined
ture inside the calorimeter if the calorimeter test temperature is
variable in a testing program, mixtures prepared under different
set below the temperature of the laboratory.
mixing conditions shall not be compared. External mixing may
NOTE 2—Condensation inside the calorimeter cells can be prevented by
result in a temperature increase in the sample that may lead to
controlling the humidity inside the calorimeter, or by controlling the
temporary non-isothermal conditions after loading the speci-
humidity or temperature of the testing environment that is in direct contact
men into the calorimeter. The thermal equilibrium time shall be
with the calorimeter. Condensation will not occur inside the calorimeter
estimated in accordance with 12.4. The results obtained at
cells if the temperature of the testing environment is at or below the
temperature of the thermostat. hydration times shorter than the equilibrium time are less
repeatable and shall only be used if the measured effects are
9. Test Specimens
larger than the variation found when estimating the thermal
equilibrium time (see Note 5).
9.1 The number of specimens and number of test batches
NOTE 5—The isothermal calorimeter will bring the temperatures of
depend on the purpose of the test program. Replicate testing of
specimens into thermal equilibrium, but the time required to do so
specimens sampled from a single prepared batch shall be done
depends on the instrument parameters, specimen size, mixing method, and
initially to establish acceptable repeatability of the apparatus as
materials used. Differences in initial thermal power caused by differences
defined in 6.2. Replicate testing of separately-prepared speci- in initial temperature can optionally be minimized by keeping all materials
at equal temperature, including any water used in cleaning mixing
mens is also required in 12.5 to establish the repeatability of
equipment. If needed, the temperatures of the starting materials may be
results for each mixing method. The number of replicates for
adjusted to compensate for temperature changes during mixing, so that the
routine testing is addressed in 12.2.1.
temperature after mixing is as close as possible to the calorimeter set
NOTE 3—See Appendix X6 for examples of different test programs.
point.
9.2 The specimen size placed in the calorimeter depends on
12.2 Loading Specimen into Calorimeter:
the instrument used as well as the type of mixture (paste or
12.2.1 Two specimens shall be prepared from one batch,
mortar).
unless the standard deviation of the time to reach the maximum
NOTE 4—Typical specimen sizes for paste and mortar are 5 g to 10 g
of the main hydration peak for six replicate separately-
and 50 g to 100 g, respectively. A smaller specimen will reduce the time
prepared specimens has previously been determined according
to reach isothermal conditions if the initial specimen temperature differs
to 12.5.1 to be less than 60 min, in which case one specimen is
from the calorimeter test temperature.
adequate (see Note 6).
10. Composition of Paste or Mortar Mixtures
NOTE 6—A sub-sample of the prepared mixture is used in this method,
and it is necessary to obtain a representative sample. Normally, replicate
10.1 Mixture proportions depend on the purpose of the test
measurements are made initially as specified in 12.5 in order to establish
program (see Note 4).
the error associated with variability in specimen composition, preparation,
10.2 Record mixture proportions for each mixture that is
and sampling.
tested, including complete description of mixing method, order
12.2.2 Wear insulating gloves or use tweezers to minimize
and timing of admixture addition.
warming of the vial and specimen by heat from hands.
12.2.3 Weigh and record the empty mass of the vial to be
11. Preparation of Apparatus
used, or tare the scale to zero with the empty vial on the scale.
11.1 Determination of Baseline—Baselines are determined 12.2.4 Place a representative sample of fresh paste or mortar
during calibrations or before each measurement, preferably into the specimen vial resting on the scale. Typically a plastic
when the calorimeter contains inert specimens, as this reduces disposable pipette is used for paste, with the opening cut to size
C1679 − 22
to match the stiffness of the paste, or a spoon is used for mortar. 12.5 Determination of Repeatability of Results:
Make sure that masses of all specimens that will be compared 12.5.1 The repeatability of the results of any mixing method
with each other do not differ by more than 20 %. Clean and dry must be determined by preparing and testing at least six
the outside of the vials as needed. replicate batches of a paste mixture that has a thermal indicator
12.2.5 Weigh and record the specimen mass to the nearest of setting time that is equal to or exceeds that of the most
0.1 g and seal the vial in order to be vapor tight. retarded mixture in the planned test program. The repeatability
12.2.6 Immediately place the vial into the calorimeter en- shall be recorded as the standard deviation of the time to reach
suring consistency between the specimens ages at the time the maximum of the main hydration peak (see Fig. 4). The
specimens are placed in the calorimeter. Typical loading times repeatability must be measured annually or when the operator
are 1 min to 2 min after the end of the mixing cycle, which for or mixing method has changed.
a 4 min cycle yields 5 min to 6 min after the cementitious
NOTE 8—It is convenient to develop a chart of repeatability versus time
material is added to the water. Record the time the vial was
to reach the maximum of the main hydration peak for a particular mixing
introduced into the calorimeter.
method used. This allows for estimation of repeatability for any given
level of retardation in a particular experiment.
12.3 Measurement of Thermal Power:
12.3.1 Start thermal power measurement immediately after
13. Calculation of Results
specimen has been placed in calorimeter. In the case of internal
13.1 Post-Processing of Data—The aim of the evaluation is
mixing, start thermal power measurement before initial contact
to monitor differences in the evolution of thermal p
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: C1679 − 17 C1679 − 22
Standard Practice for
Measuring Hydration Kinetics of Hydraulic Cementitious
Mixtures Using Isothermal Calorimetry
This standard is issued under the fixed designation C1679; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This practice describes the apparatus and procedure for measuring relative differences in hydration kinetics of hydraulic
cementitious mixtures, either in paste or mortar (see Note 1), including those containing admixtures, various supplementary
cementitious materials (SCM), and other fine materials by measuring the thermal power using an isothermal calorimeter.
NOTE 1—Paste specimens are often preferred for mechanistic research when details of individual reaction peaks are important or for particular calorimetry
configurations. Mortar specimens may give results that have better correlation with concrete setting and early strength development and are often preferred
to evaluate different mixture proportions for concrete. Both paste and mortar studies have been found to be effective in evaluating concrete field problems
due to incompatibility of materials used in concrete mixtures.
1.2 This practice does not cover the measurement of heat of hydration. Heat of hydration can be determined according to Test
Method C1702.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use. (Warning—Fresh hydraulic cementitious mixtures are caustic and may
cause chemical burns to skin and tissue upon prolonged exposure. )
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
C125 Terminology Relating to Concrete and Concrete Aggregates
C172/C172M Practice for Sampling Freshly Mixed Concrete
C219 Terminology Relating to Hydraulic and Other Inorganic Cements
C305 Practice for Mechanical Mixing of Hydraulic Cement Pastes and Mortars of Plastic Consistency
C403/C403M Test Method for Time of Setting of Concrete Mixtures by Penetration Resistance
This practice is under the jurisdiction of ASTM Committee C09 on Concrete and Concrete Aggregates and is the direct responsibility of Subcommittee C09.48 on
Performance of Cementitious Materials and Admixture Combinations.
Current edition approved Dec. 1, 2017Dec. 15, 2022. Published January 2018February 2023. Originally approved in 2007. Last previous edition approved in 20142017
as C1679 – 14.C1679 – 17. DOI: 10.1520/C1679-17.10.1520/C1679-22.
Section on Safety Precautions, Manual of Aggregate and Concrete Testing, Annual Book of ASTM Standards, Vol 04.02.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1679 − 22
C511 Specification for Mixing Rooms, Moist Cabinets, Moist Rooms, and Water Storage Tanks Used in the Testing of Hydraulic
Cements and Concretes
C778 Specification for Standard Sand
C1005 Specification for Reference Masses and Devices for Determining Mass and Volume for Use in Physical Testing of
Hydraulic Cements
C1602/C1602M Specification for Mixing Water Used in the Production of Hydraulic Cement Concrete
C1702 Test Method for Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction
Calorimetry
C1738/C1738M Practice for High-Shear Mixing of Hydraulic Cement Pastes
3. Terminology
3.1 Definitions—For definitions of terms used in this practice, refer to Terminology C125 and Terminology C219.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 baseline, n—the signal from the calorimeter when there is an inert specimen in the instrument.
3.2.2 calcium aluminate, n—various aluminate phases including but not limited to the tricalcium aluminate and ferrite phases in
portland cement clinker, calcium aluminate phases occurring in some supplementary cementitious materials, and calcium-alumino-
silicate glasses also occurring in some supplementary cementitious materials, that are capable of consuming the sulfate phases
present in hydrating cementitious systems.
3.2.3 calibration coeffıcient, n—a factor that relates the value recorded by the data acquisition system to the thermal power output.
3.2.3.1 Discussion—
Normally recorded data are in volts and the calibration coefficient has units of watts per volt (W/V). Some calorimeters may have
internal automatic calibration and will give the output in watts without the user having to specify the calibration coefficient.
3.2.4 combined mixture, n—combination of all the materials that are introduced into the calorimeter for measuring hydration
kinetics.
3.2.5 hydration time, n—the elapsed time from initial contact between the cementitious materials and the mix water.
3.2.6 inert specimen, n—specimen placed within the isothermal calorimeter made of a non-reactive material of similar thermal
properties (mainly heat capacity) as the reacting specimen made of the cementitious test mixture.
3.2.6.1 Discussion—
The output from the calorimeter is the difference between the heat flow from the test specimen and the inert specimen. The use
of an inert specimen substantially decreases the noise and drift of the measured heat flow.
3.2.7 isothermal calorimeter, n—a calorimeter that measures heat flow from a specimen maintained at a constant temperature by
intimate thermal contact with a constant temperature heat sink.
3.2.8 isothermal calorimetry, n—an experimental technique to monitor the thermal power output from a specimen kept at near
isothermal conditions.
3.2.9 isothermal hydration profile, n—the thermal power plotted as a function of hydration time, which provides an indication of
the rate of hydration over time at a given temperature.
3.2.10 main hydration peak, n—the broadest peak in the isothermal hydration profile that starts at the end of the dormant period
and for a well-balanced mixture lasts for several hours (see Fig. 1).
3.2.11 near isothermal conditions, n—a constant temperature with a permissible variation of 6 1.0 °C.
3.2.12 specimen holder, n—container within the isothermal calorimeter that conducts the heat from the specimen in the vial to the
heat flow sensor.
C1679 − 22
NOTE 1—(A) initial thermal power by dissolution of cement and initial cement hydration; (B) dormant period associated with very low thermal power
indicating slow and well-controlled hydration: (C) main hydration peak associated mainly with hydration reactions contributing to setting and early
strength development, with maximum at (D); and (E) sulfate depletion point, followed by (F) accelerated calcium aluminate activity.
FIG. 1 Example of Thermal Power Curve for Isothermal Hydration of Portland Cement
3.2.13 stock solution, n—a solution of admixture in water prepared to enable more precise volumetric addition of small quantities
of admixture, typically made by pipetting known volumes of admixture into a volumetric flask and diluting it to the flask’s fixed
volume.
3.2.14 sulfate addition, n—the addition of a soluble sulfate source (such as gypsum, calcium sulfate hemihydrate, alkali sulfate)
to a combined mixture to investigate whether a given combination of materials is in sulfate balance.
3.2.15 sulfate balance of mixture, n—the situation when the size of the main hydration peak is not increased by sulfate additions;
in some cases where the main peak is increased in size by added sulfate, it will also be accelerated in time.
3.2.16 sulfate depletion point, n—the onset of accelerated calcium aluminate activity that for a portland cement in absence of
supplementary cementitious material (SCM) and admixture may take place after the main hydration peak.
3.2.16.1 Discussion—
The sulfate depletion point may become impossible to detect without further addition of soluble calcium sulfate for certain cements
and more often in combined mixtures with admixtures or SCMs, or both. In some cases other sources of sulfate might be used to
mimic potential conditions in the system. Among these are anhydrite, arcanite, calcium langbeinite, aphthitalite, syngenite, and
others. Fig. 2 shows an example of the effect of added sulfate on the sulfate depletion point. Added sulfate may, in some combined
mixtures with admixtures or SCMs, or both, accelerate the onset of the main hydration peak. When a combined mixture is at sulfate
balance, further addition of soluble sulfate will not increase the size, or accelerate the onset, of the main hydration peak.
FIG. 2 Example of the Effect of Soluble Calcium Sulfate Addition on the Timing of the Sulfate Depletion Point for a Type I Portland Ce-
ment Mixed with Water Only at w/c = 0.45
C1679 − 22
3.2.17 thermal equilibrium time, n—the elapsed hydration time when the thermal power of replicate mixtures do not differ by more
than 0.2 mW/g of dry material.
3.2.18 thermal indicator of setting time, n— the hydration time to reach a thermal power of 50 % of the maximum value of the
main hydration peak.
3.2.19 thermal mass, n—the amount of thermal energy that can be stored by a material (J/K).
3.2.19.1 Discussion—
The thermal mass of a given material is calculated by multiplying the mass by the specific heat capacity of the material. For the
purpose of calculating the thermal mass used in this standard, the following specific heat capacities can be used: The specific heat
capacity of a typical unhydrated portland cement and water is 0.75 and 4.18 J/(g·K), respectively. Thus a mixture of A g of cement
and B g of water has a thermal mass of (0.75 × A + 4.18 × B) J/K. The specific heat capacity of typical quartz and limestone is
0.75 and 0.84 J/(g·K), respectively. The specific heat capacity of most amorphous supplementary cementitious material such as fly
ash or slag is approximately 0.8 J/(g·K).
3.2.20 thermal power, n—heat production rate measured in watts (W) or joules per second (J/s), usually expressed in relation to
the mass of cementitious material, as mW/g or J/s/g.
3.2.20.1 Discussion—
The thermal power is an indicator of the rate of various chemical reactions between cementitious materials, other fine particles,
mix water and admixtures.
3.2.21 vial, n—container into which the freshly mixed cementitious mixture is placed for a measurement.
4. Summary of Practice
4.1 An isothermal calorimeter consists of heat sink with a thermostat, two heat flow sensors and a specimen vial holder attached
to each sensor. A vial containing a freshly prepared mixture is placed in contact with one of the vial holders and a thermally inert
material is placed in contact with the other. The heat of hydration released by the reacting cementitious specimen is transferred
and passes across a heat flow sensor. The calorimeter output is calculated from the difference between the outputs from the test
specimen heat flow sensor and the inert specimen heat flow sensor. Because the heat is allowed to flow away from the specimen,
the measurement will take place at essentially constant temperature (isothermal conditions).
4.2 Mixtures with cement, SCM, admixtures, water and optional fine aggregate are prepared and introduced into an isothermal
calorimeter. Isothermal calorimetry tests are performed on a series of different mixtures for relative comparison of the hydration
kinetics. The output of the calorimeter is evaluated by graphical and mathematical means to evaluate retarding and accelerating
effects of different combinations of materials. Calcium sulfate may be added as a probe to determine if the addition of admixture,
SCMs, or both have increased the mixture’s demand for sulfate beyond that which is available in the cement.
5. Significance and Use
5.1 Thermal power curves are used to evaluate the isothermal hydration kinetics of the combined mixture of different materials
during the early period after being mixed with water. These isothermal power curves, or hydration profiles, may provide indications
relative to setting characteristics, compatibility of different materials, sulfate balance and early strength development. The
isothermal hydration profiles can also be used to evaluate the effects of compositions, proportions, and time of addition of materials
as well as curing temperature. Special care must be used in evaluating extended retardation with paste specimens, which have been
shown to overestimate the retardation of some mixtures containing cement, SCM, and admixtures.
5.2 This procedure can be used to measure the effect of chemical admixtures on the cement hydration profile. In many cases, the
addition of chemical admixture changes the kinetics of cement hydration.
5.3 Although this technique has been used historically to understand issues related to setting and slump loss, it must be emphasized
that isothermal calorimetry results cannot predict concrete performance definitely, either positively or negatively. Extensive
verification in concrete at planned dosages and temperatures, and at higher dosages, is needed. Isothermal calorimetry is an
effective tool to identify sensitivities, so that concrete testing can be efficiently planned and performed.
C1679 − 22
5.4 This practice provides a means of assessing the relative hydration performance of various test mixtures compared with control
mixtures that are prepared in a similar manner.
5.5 The procedure and apparatus can be used to monitor the thermal power from pastes and mortars alone or in combination with
chemical admixtures.
5.6 The isothermal calorimeter described here can be used to measure the thermal power and heat of hydration of mortars prepared
independently or obtained by wet sieving from concrete in accordance with Practice C172/C172M.
6. Apparatus
6.1 Devices for mixing to produce a homogeneous mixture of cement, SCM, admixtures, water and optional other fine materials
or aggregate and devices for charging the mixture into the specimen vial.
6.1.1 Weights and Weighing Devices shall conform to the requirements of Specification C1005.
6.1.2 Graduated Cylinders shall conform to the requirements of Specification C1005. The permissible variation for graduated
cylinders of less than 100-mL capacity shall be 61.0 % of the rated capacity.
6.1.3 Graduated Syringes of suitable capacities to contain the desired volume of liquid admixture or stock solution at 20 °C. 20 °C.
The permissible variation of the measured volume shall not exceed 3 % of the volume to be delivered. When admixture quantities
required are less than 2 mL, 2 mL, or are viscous in nature, optionally prepare stock solutions at appropriate dilution, to avoid
problems with small volumes measured volumetrically. Care shall be taken to inspect stock solutions for separation and any
admixture that is prone to separation in stock solution must be added in an alternative fashion, such as by analytical syringes.
6.1.4 Mixing Apparatus:
6.1.4.1 Mortar Preparation—The mixer shall comply with Practice C305.
6.1.4.2 Paste Preparation—A high shear mixer as described in Practice C1738/C1738M. A handheld household mixer capable of
mixing paste at not less than 400 r/min or other mixers for paste or mortar preparation are also permitted.
6.1.4.3 The repeatability of results for any mixing method must be measured and reported as described in 12.5.
6.1.5 Vials that can be sealed and fit into the specimen vial holders of the calorimeter.
6.1.6 Pipette, Funnel or Syringe to facilitate charging the sample to the vials. The neck of the device must be large enough to avoid
sample segregation.
6.2 Calorimeter and Data Acquisition System of suitable capacity and calibrated to monitor the thermal power of mortar or paste
in a repeatable fashion. The actual design of an individual instrument, whether commercial or custom-built, may vary, but it shall
meet the following criteria, and shall be verified to do so on a yearly basis or at any time that equipment is modified.
6.2.1 The standard deviation in thermal power for six replicate specimens sampled from one mixture of a portland cement paste
without chemical admixture at w/c 0.45 tested at 2323 °C 6 4.0 °C shall be no greater than 0.5 mW/g of dry material measured
at the maximum of the main hydration peak (see Fig. 1 for identification of the main hydration peak). For calorimeters which do
not allow external mixing, prepare six replicate mixtures.
6.2.2 The baseline measured with an inert specimen of similar thermal mass as the test specimen shall exhibit a low random noise
level and be stable against drift. The baseline measured at 2323 °C 6 4 °C during a time period of 24 h shall have a long-term
drift of less than 0.5 mW per day per gram of inert material used and a baseline random noise level of less than 0.2 mW per gram
of inert material used. In practice, the baseline is measured for 24 h or longer and a straight line is fitted to the data using a linear
least squares procedure. The long-term drift is the slope of the line and the baseline random noise level is the standard deviation
of the data about the best-fit line.
6.2.3 The vials shall be vapor tight so that endothermic heat of evaporation does not significantly influence the measurements. This
C1679 − 22
shall be verified with a 24 h measurement with water instead of the mortar or paste specimen. The difference between the measured
thermal powers of an empty vial and one containing water shall be less than 0.2 mW per gram of water when calculated according
to the procedure in 13.1.
6.2.4 The data acquisition equipment shall be capable of performing continuous logging of the calorimeter output with a time
interval between the measurements that is no larger than 60 s.
7. Materials
7.1 Sand:
7.1.1 Unless specified otherwise, use standard graded sand as defined in Specification C778 for preparing mortar samples.
7.1.2 When specified, use job-specific sand when performing a specific mortar test series that is related to field application.
7.2 Water:
7.2.1 Unless specified, use potable water as defined in Specification C1602/C1602M.
7.2.2 When specified, use job-specific water when performing a specific test series that is related to field application. Follow
Specification C1602/C1602M in reporting the type of water used.
7.3 Sulfate Source:
7.3.1 Unless specified otherwise, use reagent grade calcium sulfate dihydrate or hemihydrate prepared from reagent grade calcium
sulfate dihydrate to verify whether a mixture is in sulfate balance. See Appendix X6 for examples.
7.3.2 When specified, use a source-specific calcium sulfate for performing a specific test series that is related to field application.
8. Temperature and Humidity
8.1 Sample Preparation Temperature—Unless specified otherwise, maintain the temperature of the air in the vicinity of all
equipment and materials used in accordance with the requirements for cement mixing rooms in Specification C511.
8.1.1 When specified, different temperature conditions are permitted for research purposes or to simulate field conditions.
8.1.2 To reduce the time to reach thermal equilibrium, it is permitted to cool the mixture during mixing. It is also permitted to
adjust the temperature of the mix water or the starting materials, or both, in order to obtain a temperature of the prepared mixture
close the temperature of the calorimeter. This is especially needed when using high shear mixing.
8.2 Isothermal Calorimeter Test Temperature—The temperature of the isothermal calorimeter shall be set at 23.0 6 4.0 °C
23.0 °C 6 4.0 °C and allowed sufficient time to stabilize at that temperature.
8.2.1 When specified, a different calorimeter test temperature is permitted for research purposes or to simulate field conditions.
Take precaution to prevent condensation of moisture inside the calorimeter if the calorimeter test temperature is set below the
temperature of the laboratory.
NOTE 2—Condensation inside the calorimeter cells can be prevented by controlling the humidity inside the calorimeter, or by controlling the humidity
or temperature of the testing environment that is in direct contact with the calorimeter. Condensation will not occur inside the calorimeter cells if the
temperature of the testing environment is at or below the temperature of the thermostat.
9. Test Specimens
9.1 The number of specimens and number of test batches depend on the purpose of the test program. Replicate testing of
specimens sampled from a single prepared batch shall be done initially to establish acceptable repeatability of the apparatus as
defined in 6.2. Replicate testing of separately-prepared specimens is also required in 12.5 to establish the repeatability of results
for each mixing method. The number of replicates for routine testing is addressed in 12.2.1.
C1679 − 22
NOTE 3—See Appendix X6 for examples of different test programs.
9.2 The specimen size placed in the calorimeter depends on the instrument used as well as the type of mixture (paste or mortar).
NOTE 4—Typical specimen sizes for paste and mortar are 55 g to 10 g and 5050 g to 100 g, respectively. A smaller specimen will reduce the time to reach
isothermal conditions if the initial specimen temperature differs from the calorimeter test temperature.
10. Composition of Paste or Mortar Mixtures
10.1 Mixture proportions depend on the purpose of the test program (see Note 4).
10.2 Record mixture proportions for each mixture that is tested, including complete description of mixing method, order and
timing of admixture addition.
11. Preparation of Apparatus
11.1 Determination of Baseline—Baselines are determined during calibrations or before each measurement, preferably when the
calorimeter contains inert specimens, as this reduces the noise. If baselines are stable over time as defined in 6.2.3, they need only
be determined at calibrations. If they are not stable, they shall be determined before each measurement.
11.2 Calibration—Calibration shall be made according to manufacturer’s recommendations at regular intervals of one year or less
to determine the calibration coefficient. If the calibration coefficient differs more than 5 % from one calibration to the next,
calibration intervals shall be reduced to 90 days. The length of the time intervals between calibrations depends on the instrument
and the personnel. For the purpose of this practice, the calibration coefficient can be assumed to be constant independent of the
level of the signal.
12. Procedure
12.1 Mixing:
12.1.1 Various mixing methods that have been found useful are described in the Appendix. Because mixing intensity is a variable
that may influence the interaction of materials used to prepare cementitious mixtures, in many cases different mixing procedures
(speeds or durations) may be needed, depending on the goal of the testing. Unless mixing intensity is a defined variable in a testing
program, mixtures prepared under different mixing conditions shall not be compared. External mixing may result in a temperature
increase in the sample that may lead to temporary non-isothermal conditions after loading the specimen into the calorimeter. The
thermal equilibrium time shall be estimated in accordance with 12.4. The results obtained at hydration times shorter than the
equilibrium time are less repeatable and shall only be used if the measured effects are larger than the variation found when
estimating the thermal equilibrium time (see Note 5).
NOTE 5—The isothermal calorimeter will bring the temperatures of specimens into thermal equilibrium, but the time required to do so depends on the
instrument parameters, specimen size, mixing method, and materials used. Differences in initial thermal power caused by differences in initial temperature
can optionally be minimized by keeping all materials at equal temperature, including any water used in cleaning mixing equipment. If needed, the
temperatures of the starting materials may be adjusted to compensate for temperature changes during mixing, so that the temperature after mixing is as
close as possible to the calorimeter set point.
12.2 Loading Specimen into Calorimeter:
12.2.1 Two specimens shall be prepared from one batch, unless the standard deviation of the time to reach the maximum of the
main hydration peak for six replicate separately-prepared specimens has previously been determined according to 12.5.1 to be less
than 60 min, in which case one specimen is adequate (see Note 6).
NOTE 6—A sub-sample of the prepared mixture is used in this method, and it is necessary to obtain a representative sample. Normally, replicate
measurements are made initially as specified in 12.5 in order to establish the error associated with variability in specimen composition, preparation, and
sampling.
12.2.2 Wear insulating gloves or use tweezers to minimize warming of the vial and specimen by heat from hands.
12.2.3 Weigh and record the empty mass of the vial to be used, or tare the scale to zero with the empty vial on the scale.
C1679 − 22
12.2.4 Place a representative sample of fresh paste or mortar into the specimen vial resting on the scale. Typically a plastic
disposable pipette is used for paste, with the opening cut to size to match the stiffness of the paste, or a spoon is used for mortar.
Make sure that masses of all specimens that will be compared with each other do not differ by more than 20 %. Clean and dry the
outside of the vials as needed.
12.2.5 Weigh and record the specimen mass to the nearest 0.1 g and seal the vial in order to be vapor tight.
12.2.6 Immediately place the vial into the calorimeter ensuring consistency between the specimens ages at the time specimens are
placed in the calorimeter. Typical loading times are 11 min to 2 min after the end of the mixing cycle, which for a 4-min4 min cycle
yields 55 min to 6 min after the cementitious material is added to the water. Record the time the vial was introduced into the
calorimeter.
12.3 Measurement of Thermal Power:
12.3.1 Start thermal power measurement immediately after specimen has been placed in calorimeter. In the case of internal
mixing, start thermal power measurement before initial contact between the cementitious materials and the mixing water.
12.3.2 Measure the thermal power approximately every 60 s until at least two hours after the maximum of the main hydration peak
(see Fig. 1). The testing duration and data sampling frequency are permitted to be modified according to the nature of the test
program and the rate of change in thermal power.
NOTE 7—Paste studies are generally more repeatable than mortar, because less paste is needed to ensure known and uniform binder content in the
specimen. However, certain mixtures may be sensitive to the mixing action. Paste mixtures with water reducing or retarding admixtures generally have
longer retardation than the corresponding mortar mixtures. Especially when testing for the effect of admixtures with a retarding component, it is
recommended to verify any results obtained in paste by testing in mortar. The larger the specimen, the longer it takes for heat transfer from specimen
to
...

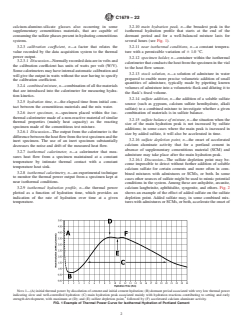
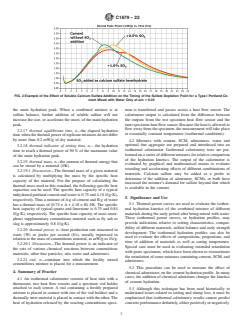




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...