ASTM C761-18
(Test Method)Standard Test Methods for Chemical, Mass Spectrometric, Spectrochemical, Nuclear, and Radiochemical Analysis of Uranium Hexafluoride
Standard Test Methods for Chemical, Mass Spectrometric, Spectrochemical, Nuclear, and Radiochemical Analysis of Uranium Hexafluoride
SIGNIFICANCE AND USE
4.1 Uranium hexafluoride is a basic material used to prepare nuclear reactor fuel. To be suitable for this purpose the material must meet criteria for uranium content, isotopic composition, metallic impurities, hydrocarbon and halohydrocarbon content. These test methods are designed to determine whether the material meets the requirements described in Specifications C787 and C996.
SCOPE
1.1 These test methods cover or give reference to procedures for subsampling and for chemical, mass spectrometric, spectrochemical, nuclear, and radiochemical analysis of uranium hexafluoride (UF6). Most of these test methods are in routine use to determine conformance to UF6 specifications in the Enrichment and Conversion Facilities.
1.2 The analytical procedures in this document appear in the following order:
Note 1: Subcommittee C26.05 will confer with C26.02 concerning the renumbered section in Test Methods C761 to determine how concerns with renumbering these sections are best addressed in subsequent publications as analytical methods are replaced with stand-alone analytical methods.
Sections
Subsampling of Uranium Hexafluoride
8
Gravimetric Determination of Uranium
9 – 17
Titrimetric Determination of Uranium
18
Preparation of High-Purity U3O8
19
Isotopic Analysis
20
Determination of Hydrocarbons, Chlorocarbons, and Partially Substituted Halohydrocarbons
21 – 27
Determination of Antimony
28
Determination of Bromine
29
Determination of Chlorine
30 – 36
Determination of Silicon and Phosphorus
37 – 43
Determination of Boron and Silicon
44
Determination of Ruthenium
45
Determination of Titanium and Vanadium
46
Spectrographic Determination of Metallic Impurities
47
Determination of Tungsten
48
Determination of Thorium and Rare Earths
49
Determination of Molybdenum
50
Atomic Absorption Determination of Metallic Impurities
51 – 56
Impurity Determination by Spark-Source Mass Spectrography
57
Determination of Boron-Equivalent Neutron Cross Section
58
Determination of Uranium-233 Abundance by Thermal Ionization Mass Spectrometry
59
Determination of Uranium-232 by Alpha Spectrometry
60 – 66
Determination of Fission Product Activity
67
Determination of Plutonium by Ion Exchange and Alpha Counting
68 – 72
Determination of Plutonium by Extraction and Alpha Counting
73 – 80
Determination of Neptunium by Extraction and Alpha Counting
81 – 88
Atomic Absorption Determination of Chromium Soluble In Uranium Hexafluoride
89 – 95
Atomic Absorption Determination of Chromium Insoluble In Uranium Hexafluoride
101 – 102
Determination of Technetium-99 In Uranium Hexafluoride
103 – 110
Method for the Determination of Gamma-Energy Emission Rate from Fission Products in Uranium Hexafluoride
112
Determination of Metallic Impurities by ICP-AES
113 – 122
Determination of Molybdenum, Niobium, Tantalum, Titanium, and Tungsten by ICP-AES
123 – 132
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (For specific safeguard and safety consideration statements, see Section 7.)
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2018
- Technical Committee
- C26 - Nuclear Fuel Cycle
- Drafting Committee
- C26.05 - Methods of Test
Relations
- Effective Date
- 01-Feb-2018
- Effective Date
- 15-Jan-2024
- Effective Date
- 01-Jan-2024
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Jan-2018
- Effective Date
- 01-Jun-2016
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Jun-2015
- Effective Date
- 01-Feb-2015
- Effective Date
- 15-Jun-2014
- Effective Date
- 15-Jun-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Jan-2014
- Effective Date
- 01-Jan-2014
Overview
ASTM C761-18, published by ASTM International, specifies standard test methods for the chemical, mass spectrometric, spectrochemical, nuclear, and radiochemical analysis of uranium hexafluoride (UF₆). Uranium hexafluoride is a fundamental feedstock material used in the nuclear fuel cycle, notably for the enrichment processes in nuclear reactor fuel preparation. Ensuring the quality and purity of UF₆ is crucial for its use in nuclear applications, and this standard provides essential procedures to determine if UF₆ meets the stringent requirements described in ASTM C787 and C996 specifications.
Key Topics
ASTM C761-18 covers a comprehensive set of analytical test methods to evaluate various properties of uranium hexafluoride, focusing on:
- Subsampling: Approaches for obtaining representative samples from bulk UF₆ containers.
- Gravimetric and titrimetric analysis: Determining uranium content by mass or chemical titration.
- Isotopic composition: Methods to analyze uranium-235 content and other isotopes, critical for reactor fuel specifications.
- Impurity detection: Identifying and quantifying metallic, hydrocarbon, halohydrocarbon, and radioactive impurities.
- Spectrochemical and radiochemical analysis: Use of mass spectrometry, atomic absorption, X-ray fluorescence, and ICP-AES for trace contaminants.
- Quality assurance: Ensuring compliance with nuclear material accountability, regulatory safeguards, health, and safety standards.
The procedures address the main impurity elements (chlorine, bromine, silicon, boron, chromium, antimony, titanium, vanadium, thorium, rare earths, technetium-99, and others), as well as key contaminants like hydrocarbons and chlorocarbons, using validated analytical instrumentation techniques.
Applications
These standard test methods are routinely applied within:
- Enrichment and conversion facilities: To confirm that UF₆ meets the required purity, isotopic, and chemical specifications before further processing or enrichment.
- Quality control laboratories: For batch certification, troubleshooting, and process optimization related to uranium hexafluoride handling.
- Regulatory compliance: For demonstrating conformance to international and national nuclear material safety, safeguard, and transport standards.
- Material accountability: Support for nuclear safeguards measurements and traceability, especially important given the regulated nature of nuclear materials.
- Research and development: Analytical protocols are essential for developing new UF₆-related processes and ensuring materials meet evolving nuclear industry needs.
By following ASTM C761-18, laboratories help ensure that uranium hexafluoride is pure, properly characterized, and fit for its intended use in the nuclear fuel cycle, meeting safety, environmental, and regulatory expectations.
Related Standards
ASTM C761-18 references several related standards and practices, including but not limited to:
- ASTM C787: Specification for Uranium Hexafluoride for Enrichment
- ASTM C996: Specification for Uranium Hexafluoride Enriched to Less Than 5% U-235
- ASTM C1689: Practice for Subsampling of Uranium Hexafluoride
- ASTM C1287, C1344, C1380, C1413, C1428, C1474, C1508, C1539: Various methods for isotopic, elemental, and impurity analysis relevant to uranium compounds
- ANSI N14.1: Standard for Packaging of UF₆ for Transport
- ISO 7195: International standard for packaging of uranium hexafluoride
- USEC 651: Good handling and practices for UF₆
Consulting these documents in conjunction with ASTM C761-18 facilitates comprehensive analysis and ensures full compliance with the latest nuclear materials testing methodologies.
Keywords: ASTM C761-18, uranium hexafluoride analysis, UF₆ testing methods, nuclear fuel standards, impurity determination, isotopic analysis, mass spectrometry, spectrochemical analysis, radiochemical analysis, quality control in nuclear materials, ASTM standards for uranium.
Buy Documents
ASTM C761-18 - Standard Test Methods for Chemical, Mass Spectrometric, Spectrochemical, Nuclear, and Radiochemical Analysis of Uranium Hexafluoride
REDLINE ASTM C761-18 - Standard Test Methods for Chemical, Mass Spectrometric, Spectrochemical, Nuclear, and Radiochemical Analysis of Uranium Hexafluoride
Get Certified
Connect with accredited certification bodies for this standard

DNV
DNV is an independent assurance and risk management provider.

Lloyd's Register
Lloyd's Register is a global professional services organisation specialising in engineering and technology.

DNV Energy Systems
Energy and renewable energy certification.
Sponsored listings
Frequently Asked Questions
ASTM C761-18 is a standard published by ASTM International. Its full title is "Standard Test Methods for Chemical, Mass Spectrometric, Spectrochemical, Nuclear, and Radiochemical Analysis of Uranium Hexafluoride". This standard covers: SIGNIFICANCE AND USE 4.1 Uranium hexafluoride is a basic material used to prepare nuclear reactor fuel. To be suitable for this purpose the material must meet criteria for uranium content, isotopic composition, metallic impurities, hydrocarbon and halohydrocarbon content. These test methods are designed to determine whether the material meets the requirements described in Specifications C787 and C996. SCOPE 1.1 These test methods cover or give reference to procedures for subsampling and for chemical, mass spectrometric, spectrochemical, nuclear, and radiochemical analysis of uranium hexafluoride (UF6). Most of these test methods are in routine use to determine conformance to UF6 specifications in the Enrichment and Conversion Facilities. 1.2 The analytical procedures in this document appear in the following order: Note 1: Subcommittee C26.05 will confer with C26.02 concerning the renumbered section in Test Methods C761 to determine how concerns with renumbering these sections are best addressed in subsequent publications as analytical methods are replaced with stand-alone analytical methods. Sections Subsampling of Uranium Hexafluoride 8 Gravimetric Determination of Uranium 9 – 17 Titrimetric Determination of Uranium 18 Preparation of High-Purity U3O8 19 Isotopic Analysis 20 Determination of Hydrocarbons, Chlorocarbons, and Partially Substituted Halohydrocarbons 21 – 27 Determination of Antimony 28 Determination of Bromine 29 Determination of Chlorine 30 – 36 Determination of Silicon and Phosphorus 37 – 43 Determination of Boron and Silicon 44 Determination of Ruthenium 45 Determination of Titanium and Vanadium 46 Spectrographic Determination of Metallic Impurities 47 Determination of Tungsten 48 Determination of Thorium and Rare Earths 49 Determination of Molybdenum 50 Atomic Absorption Determination of Metallic Impurities 51 – 56 Impurity Determination by Spark-Source Mass Spectrography 57 Determination of Boron-Equivalent Neutron Cross Section 58 Determination of Uranium-233 Abundance by Thermal Ionization Mass Spectrometry 59 Determination of Uranium-232 by Alpha Spectrometry 60 – 66 Determination of Fission Product Activity 67 Determination of Plutonium by Ion Exchange and Alpha Counting 68 – 72 Determination of Plutonium by Extraction and Alpha Counting 73 – 80 Determination of Neptunium by Extraction and Alpha Counting 81 – 88 Atomic Absorption Determination of Chromium Soluble In Uranium Hexafluoride 89 – 95 Atomic Absorption Determination of Chromium Insoluble In Uranium Hexafluoride 101 – 102 Determination of Technetium-99 In Uranium Hexafluoride 103 – 110 Method for the Determination of Gamma-Energy Emission Rate from Fission Products in Uranium Hexafluoride 112 Determination of Metallic Impurities by ICP-AES 113 – 122 Determination of Molybdenum, Niobium, Tantalum, Titanium, and Tungsten by ICP-AES 123 – 132 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (For specific safeguard and safety consideration statements, see Section 7.) 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 Uranium hexafluoride is a basic material used to prepare nuclear reactor fuel. To be suitable for this purpose the material must meet criteria for uranium content, isotopic composition, metallic impurities, hydrocarbon and halohydrocarbon content. These test methods are designed to determine whether the material meets the requirements described in Specifications C787 and C996. SCOPE 1.1 These test methods cover or give reference to procedures for subsampling and for chemical, mass spectrometric, spectrochemical, nuclear, and radiochemical analysis of uranium hexafluoride (UF6). Most of these test methods are in routine use to determine conformance to UF6 specifications in the Enrichment and Conversion Facilities. 1.2 The analytical procedures in this document appear in the following order: Note 1: Subcommittee C26.05 will confer with C26.02 concerning the renumbered section in Test Methods C761 to determine how concerns with renumbering these sections are best addressed in subsequent publications as analytical methods are replaced with stand-alone analytical methods. Sections Subsampling of Uranium Hexafluoride 8 Gravimetric Determination of Uranium 9 – 17 Titrimetric Determination of Uranium 18 Preparation of High-Purity U3O8 19 Isotopic Analysis 20 Determination of Hydrocarbons, Chlorocarbons, and Partially Substituted Halohydrocarbons 21 – 27 Determination of Antimony 28 Determination of Bromine 29 Determination of Chlorine 30 – 36 Determination of Silicon and Phosphorus 37 – 43 Determination of Boron and Silicon 44 Determination of Ruthenium 45 Determination of Titanium and Vanadium 46 Spectrographic Determination of Metallic Impurities 47 Determination of Tungsten 48 Determination of Thorium and Rare Earths 49 Determination of Molybdenum 50 Atomic Absorption Determination of Metallic Impurities 51 – 56 Impurity Determination by Spark-Source Mass Spectrography 57 Determination of Boron-Equivalent Neutron Cross Section 58 Determination of Uranium-233 Abundance by Thermal Ionization Mass Spectrometry 59 Determination of Uranium-232 by Alpha Spectrometry 60 – 66 Determination of Fission Product Activity 67 Determination of Plutonium by Ion Exchange and Alpha Counting 68 – 72 Determination of Plutonium by Extraction and Alpha Counting 73 – 80 Determination of Neptunium by Extraction and Alpha Counting 81 – 88 Atomic Absorption Determination of Chromium Soluble In Uranium Hexafluoride 89 – 95 Atomic Absorption Determination of Chromium Insoluble In Uranium Hexafluoride 101 – 102 Determination of Technetium-99 In Uranium Hexafluoride 103 – 110 Method for the Determination of Gamma-Energy Emission Rate from Fission Products in Uranium Hexafluoride 112 Determination of Metallic Impurities by ICP-AES 113 – 122 Determination of Molybdenum, Niobium, Tantalum, Titanium, and Tungsten by ICP-AES 123 – 132 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (For specific safeguard and safety consideration statements, see Section 7.) 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM C761-18 is classified under the following ICS (International Classification for Standards) categories: 27.120.30 - Fissile materials and nuclear fuel technology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C761-18 has the following relationships with other standards: It is inter standard links to ASTM C761-11, ASTM C1295-24, ASTM C859-24, ASTM C1474-19, ASTM C1287-18, ASTM C1561-10(2016), ASTM C996-15, ASTM C787-15, ASTM C1295-15, ASTM C1128-15, ASTM C1295-14, ASTM C859-14a, ASTM C859-14, ASTM C1346-08(2014), ASTM C1477-08(2014). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C761-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:C761 −18
Standard Test Methods for
Chemical, Mass Spectrometric, Spectrochemical, Nuclear,
and Radiochemical Analysis of Uranium Hexafluoride
This standard is issued under the fixed designation C761; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
Determination of Plutonium by Extraction and Alpha Counting 73 – 80
Determination of Neptunium by Extraction and Alpha Counting 81 – 88
1.1 These test methods cover or give reference to proce-
Atomic Absorption Determination of Chromium Soluble In 89 – 95
dures for subsampling and for chemical, mass spectrometric, Uranium Hexafluoride
Atomic Absorption Determination of Chromium Insoluble In 101 – 102
spectrochemical, nuclear, and radiochemical analysis of ura-
Uranium Hexafluoride
nium hexafluoride (UF ). Most of these test methods are in
6 Determination of Technetium-99 In Uranium Hexafluoride 103 – 110
routine use to determine conformance to UF specifications in Method for the Determination of Gamma-Energy Emission Rate 112
from Fission Products in Uranium Hexafluoride
the Enrichment and Conversion Facilities.
Determination of Metallic Impurities by ICP-AES 113 – 122
Determination of Molybdenum, Niobium, Tantalum, Titanium, 123 – 132
1.2 Theanalyticalproceduresinthisdocumentappearinthe
and Tungsten by ICP-AES
following order:
1.3 The values stated in SI units are to be regarded as
NOTE1—SubcommitteeC26.05willconferwithC26.02concerningthe
standard. No other units of measurement are included in this
renumbered section in Test Methods C761 to determine how concerns
standard.
with renumbering these sections are best addressed in subsequent publi-
cations as analytical methods are replaced with stand-alone analytical
1.4 This standard does not purport to address all of the
methods.
safety concerns, if any, associated with its use. It is the
Sections
responsibility of the user of this standard to establish appro-
Subsampling of Uranium Hexafluoride 8
priate safety, health, and environmental practices and deter-
Gravimetric Determination of Uranium 9 – 17
mine the applicability of regulatory limitations prior to use.
Titrimetric Determination of Uranium 18
Preparation of High-Purity U O 19
(Forspecificsafeguardandsafetyconsiderationstatements,see
3 8
Isotopic Analysis 20
Section 7.)
Determination of Hydrocarbons, Chlorocarbons, and Partially 21 – 27
1.5 This international standard was developed in accor-
Substituted Halohydrocarbons
Determination of Antimony 28
dance with internationally recognized principles on standard-
Determination of Bromine 29
ization established in the Decision on Principles for the
Determination of Chlorine 30 – 36
Development of International Standards, Guides and Recom-
Determination of Silicon and Phosphorus 37 – 43
Determination of Boron and Silicon 44
mendations issued by the World Trade Organization Technical
Determination of Ruthenium 45
Barriers to Trade (TBT) Committee.
Determination of Titanium and Vanadium 46
Spectrographic Determination of Metallic Impurities 47
2. Referenced Documents
Determination of Tungsten 48
Determination of Thorium and Rare Earths 49
2.1 The following documents of the issue in effect on date
Determination of Molybdenum 50
of material procurement form a part of this specification to the
Atomic Absorption Determination of Metallic Impurities 51 – 56
Impurity Determination by Spark-Source Mass Spectrography 57
extent referenced herein:
Determination of Boron-Equivalent Neutron Cross Section 58
Determination of Uranium-233 Abundance by Thermal Ionization 59
2.2 ASTM Standards:
Mass Spectrometry
C787Specification for Uranium Hexafluoride for Enrich-
Determination of Uranium-232 by Alpha Spectrometry 60 – 66
ment
Determination of Fission Product Activity 67
Determination of Plutonium by Ion Exchange and Alpha 68 – 72
C799Test Methods for Chemical, Mass Spectrometric,
Counting
Spectrochemical,Nuclear,andRadiochemicalAnalysisof
Nuclear-Grade Uranyl Nitrate Solutions
C859Terminology Relating to Nuclear Materials
These test methods are under the jurisdiction of ASTM Committee C26 on
Nuclear Fuel Cycle and are the direct responsibility of Subcommittee C26.05 on
Methods of Test. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Feb. 1, 2018. Published February 2018. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1973. Last previous edition approved in 2011 as C761–11. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/C0761-18. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C761−18
C996Specification for Uranium Hexafluoride Enriched to C1742 Test Method for Isotopic Analysis of Uranium
Less Than 5% U Hexafluoride by Double Standard Single-Collector Gas
C1128Guide for Preparation of Working Reference Materi- Mass Spectrometer Method
als for Use in Analysis of Nuclear Fuel Cycle Materials D1193Specification for Reagent Water
C1219Test Methods for Arsenic in Uranium Hexafluoride D3084Practice for Alpha-Particle Spectrometry of Water
(Withdrawn 2015) E60Practice for Analysis of Metals, Ores, and Related
C1233Practice for Determining Equivalent Boron Contents Materials by Spectrophotometry
of Nuclear Materials
2.3 American Chemical Society Specification:
C1267Test Method for Uranium by Iron (II) Reduction in
Reagent Chemicals
PhosphoricAcid Followed by Chromium (VI)Titration in
2.4 Other Specifications:
the Presence of Vanadium
Uranium Hexafluoride:Base Charges, Use Charges, Special
C1287Test Method for Determination of Impurities in
Charges, Table of Enriching Services, Specifications, and
Nuclear Grade Uranium Compounds by Inductively
Packaging
Coupled Plasma Mass Spectrometry
USEC 651Good Handling and Practices for UF
C1295Test Method for Gamma Energy Emission from
2.5 ANSI Standards:
Fission and Decay Products in Uranium Hexafluoride and
ANSI N 14.1 Nuclear Material-Uranium Hexafluoride-
Uranyl Nitrate Solution
Packaging for Transport
C1344 Test Method for Isotopic Analysis of Uranium
Hexafluoride by Single-Standard Gas Source Mass Spec-
2.6 ISO Standards:
trometer Method
ISO 7195Nuclear Energy-Packaging of Uranium Hexafluo-
C1346Practice for Dissolution of UF from P-10 Tubes
ride (UF ) for Transport
C1380Test Method for the Determination of Uranium Con-
tent and Isotopic Composition by Isotope Dilution Mass
3. Terminology
Spectrometry
3.1 Definitions:
C1413Test Method for Isotopic Analysis of Hydrolyzed
3.1.1 For definitions of terms relating to the nuclear fuel
Uranium Hexafluoride and Uranyl Nitrate Solutions by
cycle, refer to Terminology C859.
Thermal Ionization Mass Spectrometry
C1428 Test Method for Isotopic Analysis of Uranium
4. Significance and Use
Hexafluoride by Single–Standard Gas Source Multiple
4.1 Uraniumhexafluorideisabasicmaterialusedtoprepare
Collector Mass Spectrometer Method
nuclearreactorfuel.Tobesuitableforthispurposethematerial
C1429 Test Method for Isotopic Analysis of Uranium
must meet criteria for uranium content, isotopic composition,
Hexafluoride by Double-Standard Multi-Collector Gas
metallicimpurities,hydrocarbonandhalohydrocarboncontent.
Mass Spectrometer
These test methods are designed to determine whether the
C1441Test Method for The Analysis of Refrigerant 114,
material meets the requirements described in Specifications
Plus Other Carbon-Containing and Fluorine-Containing
C787 and C996.
Compounds in Uranium Hexafluoride via Fourier-
Transform Infrared (FTIR) Spectroscopy
5. Reagents
C1474Test Method forAnalysis of Isotopic Composition of
Uranium in Nuclear-Grade Fuel Material by Quadrupole
5.1 Purity of Reagents—Reagent grade chemicals shall be
Inductively Coupled Plasma-Mass Spectrometry
used in all procedures. Unless otherwise indicated, all reagents
C1477Test Method for Isotopic Abundance Analysis of
shall conform to the specifications of the Committee on
Uranium Hexafluoride and Uranyl Nitrate Solutions by Analytical Reagents of theAmerican Chemical Society, where
Multi-Collector, Inductively Coupled Plasma-Mass Spec-
such specifications are available. Other grades may be used,
trometry providedthatitisfirstestablishedthatthereagenttobeusedis
C1508Test Method for Determination of Bromine and
of sufficiently high purity to permit its use without lessening
Chlorine in UF and Uranyl Nitrate by X-Ray Fluores- the accuracy of the determination.
cence (XRF) Spectroscopy
5.2 Purity of Water—Unless otherwise indicated, references
C1539Test Method for Determination of Technetium-99 in
to water shall mean reagent water conforming to Specification
Uranium Hexafluoride by Liquid Scintillation Counting
D1193.
C1561Guide for Determination of Plutonium and Neptu-
nium in Uranium Hexafluoride and U-Rich Matrix by
Alpha Spectrometry
Reagent Chemicals, American Chemical Society Specifications, American
C1636Guide for the Determination of Uranium-232 in
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
Uranium Hexafluoride
listed by the American Chemical Society, see Analar Standards for Laboratory
Chemicals,BDHLtd.,Poole,Dorset,U.K.andthe United States Pharmacopeia and
C1689Practice for Subsampling of Uranium Hexafluoride
National Formulary,U.S.PharmacopeialConvention,Inc.(USPC),Rockville,MD.
United States Department of Energy, Oak Ridge, TN 37830.
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
The last approved version of this historical standard is referenced on 4th Floor, New York, NY 10036.
www.astm.org. Type 1 and 2 water have been found to be suitable.
C761−18
6. Rejection correctingforstoichiometrybasedonisotopiccontent,ignition
conditions, and nonvolatile impurities. Ref. (1-4).
6.1 Rejection or acceptance criteria are described in Speci-
fications C787 and C996.
11. Interferences
7. Safety Considerations 11.1 Nonvolatile impurities affect the accuracy of the
method and must be measured by spectrographic analysis with
7.1 Since UF is radioactive, toxic, and highly reactive,
corrections applied.
especiallywithreducingsubstancesandmoisture(seeUranium
Hexafluoride: Handling Procedures and Container Criteria,
12. Apparatus
sections 2.4 through 2.6), appropriate facilities and practices
12.1 Polytrifluorochloroethylene (PTFCE) Sample Tube,
for sampling and analysis must be provided.
TFCE Gasket, Flare Nut, and Plug, see Fig. 1.
7.2 Hydrofluoric acid is a highly corrosive acid that can
12.2 Platinum Boat and Cover—The cover should be plati-
severely burn skin, eyes, and mucous membranes. Hydroflu-
num gauze (52 mesh) and shaped to cover the boat (Fig. 2).
oric acid differs from other acids because the fluoride ion
readily penetrates the skin, causing destruction of deep tissue
12.3 Muffle Furnace, must be capable of operating continu-
layers. Unlike other acids that are rapidly neutralized, hydro-
ously at 875°C and maintain this temperature within 625°C.
fluoric acid reactions with tissue may continue for days if left
The furnace shall be equipped with a steam supply that is
untreated.FamiliarizationandcompliancewiththeSafetyData
passed through a tube furnace to preheat the steam to 875°C.
Sheet is essential.
12.4 Tube Furnace, must be capable of operating continu-
7.3 Committee C26 Safeguards Statement:
ously at 875°C and maintain this temperature within 25°C.
7.3.1 The material (uranium hexafluoride) to which these
12.5 Infrared Heat Lamps, 250 watts.
test methods apply, is subject to nuclear safeguards regulations
12.6 Analytical Balance.
governing its possession and use. The following analytical
procedures in these test methods have been designated as
12.7 Vacuum Oven.
technicallyacceptableforgeneratingsafeguardsaccountability
12.8 Dewar Flask, stainless steel.
measurement data: Gravimetric Determination of Uranium;
12.9 Spatula, platinum.
Titrimetric Determination of Uranium; All Isotopic Analyses.
7.3.2 When used in conjunction with appropriate certified
12.10 PTFCE Rod, 120 mm long and 1.6 mm in diameter.
Reference Materials (CRMs), these procedures can demon-
12.11 Forceps, platinum tipped.
strate traceability to the national measurement base. However,
12.12 Jig,suitableforholdingtheTFCEsampletubesothat
adherence to these procedures does not automatically guaran-
it can be opened with a wrench.
tee regulatory acceptance of the resulting safeguards measure-
ments.Itremainsthesoleresponsibilityoftheuserofthesetest
12.13 Box Wrench, to fit sample tube plug.
methods to assure that its application to safeguards has the
12.14 Beaker, stainless steel, 125 mL capacity.
approval of the proper regulatory authorities.
13. Reagents
SUBSAMPLING OF URANIUM HEXAFLUORIDE
13.1 Liquid Nitrogen.
8. Scope
13.2 Nitric Acid (sp gr 1.42)—concentrated nitric acid
8.1 This test method has been discontinued (see
(HNO ).
ε1
C761–04 ). The subsampling of UF from bulk sample
13.3 Nitric Acid (4M)—Mix 500 mLof concentrated HNO
containersintosmallercontainerssuitableforlaboratoryanaly-
with 1500 mL of distilled water.
ses has been published as a separate Practice C1689.
13.4 Detergent.
GRAVIMETRIC DETERMINATION OF URANIUM
14. Sampling
14.1 AUF sample is taken as described in Practice C1689.
9. Scope
9.1 Practice C1346 is applicable to the hydrolysis of ura-
15. Procedure
nium hexafluoride in polychlorotrifluoroethylene (P10) tubes.
15.1 Inspect the PTFCE sample tube for leaks.
The following test method is then applicable to the direct
NOTE 2—An indication of a leak is a yellow-green residue on the flare
gravimetric determination of uranium.
nut and cap or a yellow discoloration in the tube. Discard the sample if a
leak is indicated.
10. Summary of Test Method
15.2 Allow the sample tube to stand overnight in the
10.1 A sample of uranium hexafluoride is weighed, cooled
laboratory.
in liquid nitrogen, and hydrolyzed with water. The uranyl
fluoride solution produced is evaporated to dryness and con-
verted to uranic oxide by pyrohydrolysis.The uranium content
The boldface numbers in parentheses refer to a list of references at the end of
is determined from the weight of the uranium oxide after these test methods.
C761−18
FIG. 1 Example of a Polychlorotrifluoroethylene P-10 Tube
15.4 Weigh the sample tube to the nearest 0.1 mg.
15.5 Heat the platinum boat and screen in the pyrohydroly-
sis furnace at 875°C for 20 min.
15.6 Cool the platinum boat and store in a desiccator for 40
min. Weigh the boat and screen to the nearest 0.1 mg.
15.7 Freeze the sample by immersing the sample tube in
liquid nitrogen for 10 min.
15.8 Add enough chilled water to the tared platinum boat to
immerse the sample tube (about 50 mL).
15.9 Place the sample tube in the jig and loosen the plug
with the box wrench.
15.10 Removethesampletubefromthejigandunscrewthe
plug while holding the sample tube in an upright position.
15.11 Remove the flare nut from the sample tube and
immerse the tube and gasket in the chilled water in the tared
platinum boat.
15.12 Let the gasket remain in the chilled water about 30
min.
15.13 Remove the gasket with the forceps and rinse well
with deionized water into the boat.
15.14 Place the plug-nut assembly and gasket into a stain-
less steel beaker for drying.
15.15 Allow the tube to remain in the water until the UF
has been hydrolyzed (2 to 4 h).
15.16 Remove the tube from the sample solution by insert-
ing the TFCE rod or platinum spatula into the tube and lifting
FIG. 2 Platinum Boat and Cover
directly above the boat.
15.3 Wipe the sample tube with a lint-free tissue to remove 15.17 Rinse the sample tube with deionized water into the
any moisture or foreign material that might be adhering. boat using extreme care to prevent splashing.
C761−18
15.18 Cover the sample boat containing the UO F solution 15.34 Place the platinum boat in hot 4M HNO for3to4h
2 2 3
with the matching cover shown in Fig. 2. Place under the and rinse with deionized water acetone.
infrared head lamps and evaporate to dryness for 16 h.
16. Calculation
15.19 Shake the excess water from the sample tube and
16.1 Calculate the weight fraction of uranium in the sample
place in the stainless beaker containing the plug-nut assembly
as follows:
and gasket.
gU/g UF 5 ~A 2 ~AB!!~GravimetricFactor!/W (1)
15.20 Dry the sample tube parts in the vacuum oven at
80°C.
where:
15.21 Allow the unassembled parts to sit in the room Gravimetric Factor = gU/g U O which varies with isoto-
3 8
overnight. pic composition. Theoretical stoichi-
ometry for U O cannot be assumed
3 8
15.22 Assemble the empty sample tube and weigh to the
and the actual gU/g U O must be
3 8
nearest 0.1 mg.
established by potentiometric titra-
15.23 Disassemble the sample tube and soak the tube and
tion (1-4). (Tri-diffusion plant com-
gasket in 4M HNO at 75° to 80°C for 1 h.
mittee with DOE approval has estab-
15.24 Rinse with deionized water and place in the stainless lished 0.8479 gU/g U O by titration
3 8
steel beaker. as the factor for natural uranium,
A = grams of U O from the pyrohydro-
3 8
15.25 Clean the metal parts with detergent and rinse with
lysis of UO F ,
2 2
deionized water and acetone.
B = grams of impurity metal oxides per
15.26 Place the metal parts to the stainless steel beaker and
gram of U O ,
3 8
dry all parts in the vacuum oven at 80°C overnight.
W = corrected sample weight in grams.
15.27 Reassemble the sample tube for the next sample. The correction is for the combined effects of cover gas
trapped over the UF in the sample tube and the air buoyancy
15.28 Set the temperatures of the furnace and tube furnace
correction (5).Thefollowingequationhasbeendeterminedfor
at 875°C.
the sample tube in Fig. 1 and the subsampling conditions
15.29 Establish a steam flow to the furnace equal to 1 L of
described in Practice C1689. The correction equation is appli-
water per hour.
cable for sample weights in the range of 7 to 13 g.
15.30 Place the boat into the furnace with the platinum
W 5 1.00047 x 20.0058 (2)
~ !
cover on the boat and pyrohydrolyze the sample for 1 h.
where:
15.31 Remove the boat from the furnace, cool, and place in
x = observed UF sample weight, g.
a desiccator while still warm.
15.32 Desiccatethesamplefor1handweighquicklytothe 17. Precision and Bias
nearest 0.1 mg.
17.1 Precision—The precision within a laboratory and be-
15.33 Transfer a portion of the U O residue to a vial and tween laboratories was established by analyzing 15 samples at
3 8
submit for spectrographic analysis to determine the weight of each laboratory. The sampling scheme is shown in Table 1.
nonvolatile impurities. Within a laboratory, based on 15 measurements made on
C761−18
separate days the relative standard deviation is 0.021%. The method respectively. For multi-collector instruments, Test
results from all the laboratories are shown in Table 2. Methods C1428 and C1429, using single or double standard
can be used.
17.2 Bias—To establish an estimate of bias for the gravi-
metricmethod,aseriesofcomparativeanalysesofUF control 20.3 For hydrolyzed UF , methods using Thermal Ioniza-
6 6
batches were made using the gravimetric and potentiometric tion Mass Spectrometry (TIMS) have been developed and can
titration methods. The potentiometric titration was used as the be used: Test Methods C1413 and C1380. Methods using
reference method because the uranium was measured directly ICP-MS can also be used: Test Methods C1474 and C1477.
using NIST potassium dichromate. The results are shown in
DETERMINATION OF HYDROCARBONS,
Table 3.
CHLOROCARBONS, AND HALOHYDROCARBONS
TITRIMETRIC DETERMINATION OF
21. Scope
URANIUM
21.1 The determination of some forms of hydrocarbons,
18. Scope
chlorocarbons, and halohydrocarbons in UF vapor can be
performedusingTestMethodC1441.Asanalternative,amass
18.1 A sample of the U O produced by the hydrolysis of
3 8
spectrometry technique may be used and is detailed below.
the UF and ignition of the resulting UO F is analyzed
6 2 2
Although this test method is only semiquantitative, it is
according to Test Method C1267.
adequate for certifying that the subject impurities do not
PREPARATION OF HIGH-PURITY U O
exceed 0.01 mol% of the UF .
3 8
22. Summary of Test Method
19. Scope
22.1 UF is admitted to a mass spectrometer through a gas
19.1 High purity U O can be prepared according to Prepa-
3 8
sample leak, and magnetic scanning is employed to record a
rationC1128.Highpurityuraniumisneededforablankmatrix
spectrum of peaks.Arepresentative group of recorded peaks is
for analyses using ICP-MS, ICP-AES, AA, XRF, and MS
compared to the same peaks in a pure UF standard scan to
equipment.
determine whether appreciable ion fragments from subject
ISOTOPIC ANALYSIS
impurities are present.
23. Interferences
20. Scope
20.1 The isotopic composition can be determined on either 23.1 If detectable impurities are present, a complete mass
scan of the range from 12 to 400 is performed. All impurities
gaseous UF or on hydrolyzed UF .
6 6
are then identified from their cracking patterns, and calcula-
20.2 For gaseous UF , using single collector mass spec-
tions are performed using ionization efficiency factors for the
trometer instruments, Test Methods C1344 and C1742 have
compounds present. Since cracking patterns vary with ioniza-
been developed and can be used for single or double standard
tion potential and ionization efficiencies vary with focus
conditions, this measurement can only be performed by one
proficient in analytical mass spectrometery.
Standard reference material, now available as NIST SRM 136e.
24. Apparatus
24.1 A mass spectrometer with resolution adequate to dis-
TABLE 2 Results of Interlaboratory Study—U in UF
tinguish between adjacent peaks at m/e =400 is required. For
Analysis Site %U in UF
example, a 152-mm radius, 60-deg, Nier-type spectrometer
GAT ORGDP PGDP
modified for spectrum recording (6) is suitable. The sample
Subsampled at GAT:
inlet system should be of nickel or Monel, equipped with an
67.600 67.619 67.589
adjustable viscous-flow or molecular leak for delivering the
67.601 67.574 67.575
67.583 67.607 67.612 sample to the ion source.
67.611 67.600 67.612
24.2 The ion source must be fabricated from nonmagnetic
67.618 67.606 Sample Lost
Subsampled at ORGDP:
materialsuchasNichromeV,andmustbedesignedsoitcanbe
67.614 67.580 67.611
disassembled for cleaning. The magnetic field of the analyzer
67.611 67.621 67.598
magnet must be continuously variable from about 200 to 6500
67.587 67.600 67.501
67.599 67.606 67.610
gauss. A single ion collector electrode is suitable, and a
67.617 67.596 67.624
vibrating-reed electrometer and 304-mm strip chart recorder
Subsampled at PGDP:
are optimum for amplifying and recording ion signals.
67.616 67.588 67.591
67.586 67.602 67.620
24.3 It is quite possible that quadrupole or time-of-flight
67.573 67.612 67.612
67.614 67.606 67.612 instruments could be adapted to this measurement.
67.607 67.586
Mean and Standard Deviation:
25. Procedure
67.602 ± 0.014 67.601 ± 0.013 67.603 ± 0.014
25.1 UF Standard Measurements:
C761−18
TABLE 3 Determination of Uranium in Uranium Hexafluoride—Comparison of Gravimetric and Potentiometric Titration Methods
%Uranium
Control UF Number of Bias
Method
A B
Date Measurements Estimate
Mean SD
9/78 Gravimetric 24 67.610 0.009 −0.001
Potentiometric Titration 9 67.611 0.015
5/82–8/82 Gravimetric 30 67.596 0.010 −0.009
Potentiometric Titration 8 67.605 0.011
7/83–9/83 Gravimetric 25 67.610 0.006 +0.005
Potentiometric Titration 8 67.605 0.010
A
Control UF used in 9/78 was a different batch of material from that used in 1982 and 1983.
B
Potentiometric titration results are used as the reference values for the bias estimates.
25.1.1 Select a standard material that has been given repeti-
Mass Number Positively Charged Ion
Fragment
tive flash purifications to rid it of all volatile impurities.
15 CH
Isotopic UF standards usually fall in this category.
26 C H
2 2
27 C H
25.1.2 With the electrometer sensitivity set at ⁄100 of the
2 3
31 CF
mostsensitiveusableoperatingrange,adjustthegasflowtothe
43 C H
3 7
+
ion source to record a mass (Note 3) 333 peak (UF )
5 47 CCl
49 CCl
approximately 80% of full scale (80 divisions).
69 CF
NOTE 3—The term “mass” in this procedure alludes to m/e, the
26.3 Read sample intensities for the representative ion
mass-to-charge ratio (see also Terminology C859).
fragments from the recorder chart.
+
25.1.3 Measure the ratio of mass 333 (UF ) to 147.5
26.4 Subtract the background intensities observed on the
++
(UF ). Mass 333 is measured on a sensitivity range one-
pure standard from respective sample intensities.
hundredth that of mass 147.5. Depending on focus conditions,
26.5 Examine the net intensity at each of the eight mass
a ratio of the order of 10 is obtained. Measure this ratio only
numbers.(Itwillberecalledthatonerecorderchartdivisionof
once per day and use for calculating results of all samples
net intensity is equivalent to about 2 ppm on a UF basis;
analyzed that day.
however, ionization efficiencies of compounds differ, and a
25.1.4 With the electrometer sensitivity set at ⁄100 of the
specific ion fragment may result from many different com-
most sensitive usable operating range, increase the gas flow to
pounds. Thus, the net intensity at a specific mass number is
provide an output signal of approximately 80 divisions at the
only qualitative and not a quantitative measurement of impu-
147.5massposition.Thisgivesadetectionlimitoftheorderof
rity.)
+
2 ppm per chart division: 50 to 100 due to ratio between UF
26.5.1 If the net ion intensity does not exceed 2 ppm at any
++
andUF ,100duetosensitivityshunts,andapproximately80
ofthemasspositions,reportthesampleascontaininglessthan
on the recorder chart.
0.01 mol% of the subject impurities.
25.1.5 Recordascanofmassrangefrom12to150usingthe
26.5.2 Where detectable impurities are apparent, perform a
most sensitive usable operating shunt, and use this scan as a
complete mass scan of the range from 12 to 400, identify
background for all samples analyzed that day.
impurities,andperformcalculationsusingionizationefficiency
factors for the compounds present.
25.2 UF Sample Measurement:
25.2.1 Introducethesampletothespectrometersourcesuch
27. Reliability
that an output intensity of approximately 80 chart divisions is
++
27.1 This simplified procedure was designed specifically to
obtained at the 147.5 mass number (UF ), using ⁄100 the
certify that a UF sample contains less than 0.01 mol%
most sensitive usable operating range.
hydrocarbons, chlorocarbons, and partially substituted halohy-
25.2.2 Recordascanofmassrangefrom12to150usingthe
drocarbons. Thus, the procedure is qualitative rather than
most sensitive usable operating range.
quantitative in cases where the impurity level is below 100
25.2.3 Repeat 25.2.1 and 25.2.2 for each sample to be
ppm.
analyzed that day.
27.2 The detectability limit for any ion fragment is about 2
ppm. The detectability limit for the parent compound could be
26. Calculation
greater or less than 2 ppm depending on ionization efficiency
26.1 Due to mass spectrometer cracking patterns, low-mass
and cracking pattern. If one of the subject compounds were
ion fragments are produced from all compounds, even the
present to 10 ppm or more, it would be evident from monitor-
high-mass ones.
ing the eight masses. Thus, a quoted result of less than 0.01
mol% is conservative.
26.2 Itispracticaltolookforarepresentativegroupofsuch
ion fragments at specific masses. Thus, initially monitor the 27.3 The results are considered quantitative when the im-
following masses for purposes of this procedure: purity being determined is present to a level greater than 100
C761−18
ppm.Insuchinstancestheimpurityisidentifiedandmeasured.
The 95% symmetrical confidence interval for such a measure-
ment is 650% of the quoted impurity.
DETERMINATION OF ANTIMONY
28. Scope
28.1 The Atomic Absorption test method has been discon-
tinued (see C761–96). Antimony can be determined by ICP-
MS. Test Method C1287 can be used.
DETERMINATION OF BROMINE
29. Scope
29.1 The Spectrophotometric test method has been discon-
tinued (see C761–96). Bromine can be determined by X-Ray
spectroscopy. Test Method C1508 can be used.
DETERMINATION OF CHLORINE
FIG. 3 Apparatus for Distillation of Chlorine
30. Scope
30.1 Chlorine can be determined by X-Ray Spectroscopy.
Test Method C1508 can be used. Chlorine can also be
33.5 Potassium Permanganate Solution (1 %)—Prepare a
determined by titrimetry. This test method is described below.
1% solution of KMnO in water.
It is applicable over a range from 10 to 100 ppm chlorine;
however, higher concentrations can be measured by appropri- 33.6 Sodium Acetate (NaC H O ), reagent grade.
2 3 2
ate sample dilution.
33.7 Sodium Thiosulfate Solution (0.025 N)—Prepare a
0.025 N solution of Na S O in water.
2 2 3
31. Summary of Test Method
33.8 Starch Indicator Solution, pH 7.
31.1 The test method consists of treating a hydrolyzed
sample of UF with ferrous sulfate in sulfuric acid solution to
33.9 Sulfuric Acid (sp gr 1.84)—Concentrated sulfuric acid
reduce chlorates, and then with potassium permanganate to
(H SO ).
2 4
liberate free chlorine.The chlorine gas is carried by a nitrogen
stream into a potassium iodide solution, and the liberated 34. Procedure
iodine is titrated with sodium thiosulfate. Bromine, if present,
34.1 Sample Preparation:
is determined separately, and a correction is applied to the
34.1.1 Hydrolyze the sample of UF with distilled water.
chlorine result.
Approximately 250 g of UF from a nickel knockout container
31.2 It is recommended that the potassium iodide-sodium may be hydrolyzed to provide a sample for various chemical
acetate solution be made up fresh once each week. Any color
measurements, or about 20 g from two polychlorotrifluoroeth-
change signals the need for a new solution. As the solution ylene sample tubes may be hydrolyzed to provide the chlorine
ages, the blank result increases; therefore, the same potassium sample. The procedure for hydrolyzing the contents of the
iodide solution is used for both sample and blank. polychlorotrifluoroethylene tubes is described here.
34.1.1.1 Immerse the tubes in liquid nitrogen and cool for
31.3 If the sample solution is allowed to boil too vigorously
10 min.
when chlorine gas is being released, liquid droplets may be
34.1.1.2 Remove the top flare plugs and collars, and place
carried by the nitrogen stream into the potassium iodide
the tubes into a platinum dish or a polychlorotrifluoroethylene
solution, resulting in sample bias.
beaker containing 100 mL of chilled distilled water.
32. Apparatus
34.1.1.3 After hydrolysis of the UF , remove the polychlo-
rotrifluoroethylenetubesandrinsewithdistilledwater.Addthe
32.1 Distillation Apparatus, shown in Fig. 3.
rinse solution to the UO F solution.
2 2
33. Reagents
34.2 Analysis:
33.1 Boric Acid (H BO ), reagent grade, crystal or powder.
34.2.1 Fill the graduated cylinder in Fig. 2 to the 150-mL
3 3
level with KI-NaC H O solution. Then connect the delivery
2 3 2
33.2 Ferrous Sulfate Solution—5 g FeSO ×7H O dis-
4 2
tubesoitstipisnearthebottomofthesolutioninthereceiving
solved in 500 mL of 3.6 M sulfuric acid.
graduate.
33.3 Potassium Iodide (KI), reagent grade.
34.2.2 Dispense 20 g of H BO into the 1-L round-bottom
3 3
33.4 Potassium Iodide-Sodium Acetate Solution—Dissolve flask.
100gofKIand100gofNaC H O ×3H O in distilled water 34.2.3 Transfer the sample solution containing UO F from
2 3 2 2 2 2
and dilute to 2 L. approximately 20 g of UF in 100 mL of solution to the flask.
C761−18
34.2.4 Add 10 mL of concentrated H SO (sp gr 1.84) to 38.2 Reduction of the silico- or phosphomolybdate ions
2 4
the flask, and swirl the contents for mixing. with a combination 1-amino-2-naphthol-4-sulfonic acid, so-
34.2.5 Add10mLoftheFeSO solution,rinsethemouthof dium sulfite, sodium pyrosulfite solution produces the same
theflask,andconnecttheflaskimmediatelytotheapparatusas molybdenum blue complex that is measured
in Fig. 3. spectrophotometrically, directly in the uranium solution at 710
34.2.6 Initiatenitrogenflowthroughthesolutionatarateof nm. One gram of uranium absorbs slightly at this wavelength,
2 to 3 bubbles per second and start the water flow through the butitsabsorbanceiseasilycorrectedbytheuseofanadditional
condenser. aliquot to which no reducing agent is added as the blank.
34.2.7 Heat the contents of the flask until boiling and allow
39. Interferences
to boil for 30 s.
34.2.8 Removetheheat,add10mLof1%KMnO solution
39.1 Phosphate interference in the silicon determination is
through the sidearm, and close the sidearm by clamping the
eliminated by the addition of oxalic acid to decompose any
rubber tube that is attached to the end of the sidearm.
phospho-molybdate formed. Silicon in small amounts does not
34.2.9 Reapply heat and allow the contents of the flask to
interfereinthephosphorusanalysissincesilicomolybdatedoes
simmer for 5 min.
not form at the acid concentration at which the phosphomo-
34.2.10 Remove the heat, but continue the nitrogen purge
lybdic acid is formed.
for an additional 5 min.
39.2 Fluoride, which would be a serious interference in the
34.2.11 Rinse the delivery tube into the receiving graduate
silicon analysis, is complexed with boric acid.Ahigh concen-
and transfer the contents of the graduate to a 300-mL Erlen-
tration of silicon which could interfere in the phosphorus
meyer flask.Add 1 mLof starch solution and titrate the iodine
analysis is prevented by handling hydrolyzed UF samples in
with 0.025 N Na S O solution to the starch end point. (The
2 2 3
platinumorplastic.Otherpotentialinterferencesrarelypresent
iodine may be measured spectrophotometrically rather than
in significant amounts are arsenic and tungsten.
titrimetrically.)
34.2.12 Perform a blank analysis by carrying 100 mL of
40. Apparatus
distilled water through procedural steps, 34.2.1 through
40.1 Polyethylene Bottles, 100 and 500 mL.
34.2.11, and subtracting from the sample titration.
40.2 Polyethylene Beakers, 100 mL.
35. Calculation
40.3 Polyethylene Pipets; 1, 2, 5, and 10 mL.
35.1 Calculatetheconcentrationofchlorineinppmchlorine
40.4 Spectrophotometer, equipped with 1 and 5 cm cells as
on a uranium basis as follows:
described in Practice E60.
Cl, ppm 5 ~V 2 V !~N!~0.03545 310 !/S
1 2
where:
41. Reagents
V = millilitres of thiosulfate for sample,
41.1 Ammonium Hydroxide Silicon-Free—Distill500mLof
V = millilitres of thiosulfate for blank,
saturated NH OH through plastic tubing into 300 mL of
N = normality of Na S O solution,
2 2 3
distilled water.
0.03545 = grams of chlorine per milliequivalents, and
41.2 Ammonium Molybdate Solution (10 %)—Dissolve 100
S = grams of uranium.
g of reagent grade (NH ) Mo O in water, and dilute the
4 6 7 24
36. Reliability
solution to 1 L with distilled water.
36.1 Theprecisionatthe95%confidencelevelis 610%at
NOTE 4—Not all commercially available (NH ) Mo O is suitable.
4 6 7 24
the 100-ppm level.
Material supplied by J. T. Baker Chemical Co. or Baker and Adamson
Products, however, has been found to be satisfactory consistently.
DETERMINATION OF SILICON AND PHOSPHORUS
41.3 Boric Acid Solution (5 %)—Dissolve 25 g of reagent
37. Scope grade H BO in water, and dilute the solution to 500 mL.
3 3
41.4 Oxalic Acid Solution (5 %)—Dissolve 25 g of reagent
37.1 Phosphorus and Silicon can be determined by ICP-MS
(see Test Method C1287). grade H C O in water, and dilute the solution to 500 mL.
2 2 4
(Thissolutionisnotrequiredfordeterminationofphosphorus.)
37.2 Silicon can be analyzed byAtomicAbsorption without
matrix separation. 41.5 Phosphorus Standard Solution (25 µg P/mL)—
Dissolve 4.6422 g of ammonium dihydrogen phosphate
37.3 Phosphorus and Silicon can be analyzed by spectro-
[(NH )H PO ] in distilled water and dilute the solution to 1 L
4 2 4
photometry (as described below). With these procedures about
with distilled water. Transfer 20 mL of this solution to a 1-L
0.5 µg silicon or phosphorus per gram of uranium can be
volumetric flask and dilute to 1 volume with distilled water to
detected.
obtain a solution containing 2.0 µg P/mL.
38. Summary of Test Method
41.6 Reducing Mix—Dissolve 0.1 g of 1-amino-2-naphthol-
38.1 The test methods are based on the development of the 4-sulfonicacid,1.0gofsodiumsulfite(Na SO ),and10.0gof
2 3
colorknownasmolybdenumblueobtainedbythereductionof sodiumpyrosulfite(Na S O )inwater;thendilutethesolution
2 2 5
silico- or phosphomolybdate ions. to 100 mL.
C761−18
41.7 Silicon Standard Solution (2.5 µg Si/mL)—Dissolve 42.2.2.4 Place the TFE-fluorocarbon beaker containing the
10.6 mg of precipitated silica (SiO ) and 0.5 g of sodium sample in a water bath or an oven and heat to 90 to 95°C.
hydroxide (NaOH) in a platinum dish. Transfer the solution to
42.2.2.5 Remove the beaker from the water bath or the
a 2-L plastic bottle and dilute to volume. oven, and add 5 mLof 10% (NH ) Mo O solution immedi-
4 6 7 24
ately.
NOTE5—Siliconinsolutionassodiumsilicateisnotstablewhenstored
42.2.2.6 Adjust the acidity to a pH of 1.2 to 1.3 while the
in polyethylene bottles. New standard solutions should be prepared
solution is still warm by adding silicon-free NH OH or HCl.
monthly.
42.2.2.7 Allow the sample to stand 10 min to permit the
41.8 SulfuricAcid—BoricAcid Solution (10 % H SO —4%
2 4
formation of the silico-molybdate complex.
H BO )—Dissolve20gofreagentgradeH BO inwater.Add
3 3 3 3
42.2.2.8 Add 10 ml of 5% H C O solution to the beaker
2 2 4
50 mL of concentrated H SO (sp gr 1.84) and dilute the
2 4
and swirl.Allow the solution to stand for 2 min to decompose
solution to 500 mL.
any phosphomolybdate.
41.9 Uranium Oxide (U O,UO,orUO ), phosphorus-
3 8 2 3
42.2.2.9 Add 2 mLof reducing mix to the beaker and swirl.
and silicon-free.
42.2.2.10 Addsufficient6 NHClimmediatelytothesample
to obtain a 1 N acid solution.
NOTE 6—All standard solutions should be made and stored in plastic
containers to prevent silicon contamination from glassware. 42.2.2.11 After all the precipitate is dissolved, transfer the
solution to a 50 or 100-mL volumetric flask and dilute to
42. Procedure
volume with 1 N HCl.
42.2.2.12 Determine the absorbance of the solutions in a
42.1 Sample Preparation:
5-cm cell at 710 nm.
42.1.1 Hydrolyze a weighed portion of 6 to 10 g of UF in
aplatinumboatin80mLofdistilledwaterasdescribedin15.7
NOTE 7—The uranyl ion shows a slight absorbance at 710 nm, and
through 15.17. samples must be corrected for this absorbance.This is best determined by
taking an additional aliquot from the sample solution and treating it as
42.1.2 Transfer the solution to a 100-mL plastic bottle and
indicatedintheprocedureuptothepointthepHisadjustedwithNH OH.
dilute to 100 mL.
Any precipitate is dissolved with a minimum of H SO and the solution
2 4
42.1.3 Transfer an aliquot equivalent to1gofUF to a
diluted to 50 mL. The absorbance of this solution is used as an additional
blank correction.
100-mLTFE-fluorocarbonbeaker,andadd1mLof6NH SO .
2 4
(Phosphorusaliquotsshouldcontain1to50µgofphosphorus.)
42.2.2.13 Determine the quantity of silicon in the aliquot
42.1.4 Add 20 mLof 5% H BO , and heat the solution for
3 3 from a previously prepared calibration curve.
20 min to complex the fluoride.
42.3 Determination of Phosphorus:
42.1.5 Forphosphorusanalysisonly,transferthesolutionto
42.3.1 Preparation of Calibration Curve—Using a TFE-
a 100-mL borosilicate beaker and evaporate to 20 mL (see
fluorocarbon beaker, dissolve sufficient uranium oxide
42.3).
(phosphorus-free) containing 20 g of uranium in HNO . Dilute
42.2 Determination of Silicon:
itto200mLinaplasticbottle.Toseparate10-mLaliquots,add
42.2.1 Preparation of Calibration Curve: 0, 5, 10, 25, and 50 µg of phosphorus. Analyze by the
42.2.1.1 Pipet standard aliquots containing 0, 2.5, 5.0, 7.5, procedure described below. Plot the absorbances corrected for
10.0, and 12.5 µg of silicon into plastic beakers. theblanksagainsttheknownquantitiesofphosphorustoobtain
a calibration curve.
42.2.1.2 Add silicon-free uranium, 0.1 g as uranyl nitrate
solution, to each beaker. Prepare the uranyl nitrate solution by 42.3.2 Analysis:
42.3.2.1 Neutralizethesamplealiquotcontaining1to50µg
dissolving silicon-free uranium oxide in nitric acid in a
TFE-fluorocarbon beaker. of phosphorus in a volume of 20 mL or less in a 100-mL
borosilicate beaker with NH OH until a precipitate begins to
42.2.1.3 Add from 1.0 to 1.5 mL of 18 N H SO to each
2 4
form.
beaker and dilute to 25 mL. Then proceed with the analysis
starting with 42.2.2.4. Plot the absorbances corrected for the 42.3.2.2 Add sufficient 6 N HCl to make the sample 1 N in
acid.
blank against the known quantities of silicon taken to obtain a
calibration curve. In a typical case, 10 µg of silicon gave a 42.3.2.3 Transfer the sample to a 100-mL volumetric flask
correctedabsorbanceofabout0.285ina5-cmcell.Uptoabout and dilute to about 50 mL with 1 N HCl.
150 µg can be handled using a 1-cm absorbance cell and an 42.3.2.4 Add 5 mL of (NH ) Mo O solution.
4 6 7 24
appropriate calibration curve. 42.3.2.5 Heat the sample to 90 to 95°C in a water bath.
42.2.2 Analysis: 42.3.2.6 Allow the sample to cool to room temperature in a
42.2.2.1 Ablank containing all the reagents in the amounts waterbath;add2mLofreducingmixtothesolutionandswirl.
42.3.2.7 Dilute the solution in the 100-mL volumetric flask
used in the sample aliquot must be analyzed with the samples.
Normally10mLof1 NNaOHsolutiongivesanabsorbanceof to volume with 1 N HCl.
0.030 to 0.050 in this procedure. Most other reagents were 42.3.2.8 After1h,measuretheabsorbanceofthesolutionin
found to be nearly silicon-free. a 5-cm cell at 710 nm against a reagent blank.
42.2.2.2 Dilutethealiquotofthesampleina100-mLplastic
NOTE 8—Since the uranyl ion absorbs slightly at this wavelength, a
beaker to 25 mL with water.
correction must be made for the quantity of uranium present. This can be
42.2.2.3 Add from 1 to 1.5 mL of 18 N H SO . donebymeasuringtheabsorbanceofanadditionaluntreatedaliquotofthe
2 4
C761−18
original sample made to a 50-mL volume or by applying a previously
document. The Spectrophotometric determination has been
determined standard correction when the amounts of uranium in the
discontinued (see C761–96).
aliquot are known (typically, each gram of uranium in a 50-mL volume
gives an absorbance reading of 0.038 at 710 mm).
DETERMINATION OF THORIUM AND RARE
42.3.2.9 Determine the quantity of phosphorus present in
EARTHS
the aliquot from a previously prepared calibration curve. Then
calculatethephosphoruscontentoftheoriginalmaterialonthe
49. Scope
basis desired.
49.1 ThoriumandrareearthscanbedeterminedbyICP-MS
(see Test Method C1287). The Spectrophotometric determina-
43. Reliability
ε1
tion has been discontinued (see C761–04 ).
43.1 At the 10-µg level of either element, the precision at
DETERMINATION OF
the 95% confidence level is 610%.
MOLYBDENUM
DETERMINATION OF BORON
AND SILICON
50. Scope
50.1 Molybdenum can be determined by ICP-MS (see Test
44. Scope
Method C1287) or by ICP-AES as described later in this
44.1 Boron and Silicon can be determined by ICP-MS (see
document. The Spectrophotometric determination using the
Test Method C1287). They can also be analyzed by FTIR on
thiocyanate complex has been discontinued (see C761–96).
gaseous UF (see Test Method C1441). The test method using
DC-arc spectrographic emission has been discontinued (see
ATOMIC ABSORPTION DETERMINATION OF
C761–96).
METALLIC IMPURITIES
DETERMINATION OF RUTHENIUM
51. Scope
51.1 A test method is presented for the analysis of 14
45. Scope
metallicelementsinuraniumcompoundsbyatomicabsorption
45.1 Ruthenium can be determined by ICP-MS (see Test
spectroscopy (7, 8). The test method has been shown to be
Method C1287). The Atomic Absorption test method using
applicable to the anal
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: C761 − 11 C761 − 18
Standard Test Methods for
Chemical, Mass Spectrometric, Spectrochemical, Nuclear,
and Radiochemical Analysis of Uranium Hexafluoride
This standard is issued under the fixed designation C761; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 These test methods cover or give reference to procedures for subsampling and for chemical, mass spectrometric,
spectrochemical, nuclear, and radiochemical analysis of uranium hexafluoride (UF ). Most of these test methods are in routine use
to determine conformance to UF specifications in the Enrichment and Conversion Facilities.
1.2 The analytical procedures in this document appear in the following order:
NOTE 1—Subcommittee C26.05 will confer with C26.02 concerning the renumbered section in Test Methods C761 to determine how concerns with
renumbering these sections are best addressed in subsequent publications as analytical methods are replaced with stand-alone analytical methods.
Sections
Subsampling of Uranium Hexafluoride 7
Subsampling of Uranium Hexafluoride 8
Gravimetric Determination of Uranium 8 – 16
Gravimetric Determination of Uranium 9 – 17
Titrimetric Determination of Uranium 17
Titrimetric Determination of Uranium 18
Preparation of High-Purity U O 18
3 8
Preparation of High-Purity U O 19
3 8
Isotopic Analysis 19
Isotopic Analysis 20
Determination of Hydrocarbons, Chlorocarbons, and Partially 20–26
Substituted Halohydrocarbons
Determination of Hydrocarbons, Chlorocarbons, and Partially 21 – 27
Substituted Halohydrocarbons
Determination of Antimony 27
Determination of Antimony 28
Determination of Bromine 28
Determination of Bromine 29
Determination of Chlorine 29 – 35
Determination of Chlorine 30 – 36
Determination of Silicon and Phosphorus 36– 42
Determination of Silicon and Phosphorus 37 – 43
Determination of Boron and Silicon 43
Determination of Boron and Silicon 44
Determination of Ruthenium 44
Determination of Ruthenium 45
Determination of Titanium and Vanadium 45
Determination of Titanium and Vanadium 46
Spectrographic Determination of Metallic Impurities 46
Spectrographic Determination of Metallic Impurities 47
Determination of Tungsten 47
Determination of Tungsten 48
Determination of Thorium and Rare Earths 48
Determination of Thorium and Rare Earths 49
Determination of Molybdenum 49
Determination of Molybdenum 50
Atomic Absorption Determination of Metallic Impurities 50 – 55
Atomic Absorption Determination of Metallic Impurities 51 – 56
Impurity Determination by Spark-Source Mass Spectrography 56
Impurity Determination by Spark-Source Mass Spectrography 57
These test methods are under the jurisdiction of ASTM Committee C26 on Nuclear Fuel Cycle and are the direct responsibility of Subcommittee C26.05 on Methods
of Test.
Current edition approved May 15, 2011Feb. 1, 2018. Published July 2011February 2018. Originally approved in 1973. Last previous edition approved in 20042011 as
ε1
C761 – 04C761 – 11. . DOI: 10.1520/C0761-11.10.1520/C0761-18.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C761 − 18
Determination of Boron-Equivalent Neutron Cross Section 57
Determination of Boron-Equivalent Neutron Cross Section 58
Determination of Uranium-233 Abundance by Thermal Ionization 58
Mass Spectrometry
Determination of Uranium-233 Abundance by Thermal Ionization 59
Mass Spectrometry
Determination of Uranium-232 by Alpha Spectrometry 59 – 65
Determination of Uranium-232 by Alpha Spectrometry 60 – 66
Determination of Fission Product Activity 66
Determination of Fission Product Activity 67
Determination of Plutonium by Ion Exchange and Alpha 67 – 71
Counting
Determination of Plutonium by Ion Exchange and Alpha 68 – 72
Counting
Determination of Plutonium by Extraction and Alpha Counting 72 – 79
Determination of Plutonium by Extraction and Alpha Counting 73 – 80
Determination of Neptunium by Extraction and Alpha Counting 80 – 87
Determination of Neptunium by Extraction and Alpha Counting 81 – 88
Atomic Absorption Determination of Chromium Soluble In 88 – 94
Uranium Hexafluoride
Atomic Absorption Determination of Chromium Soluble In 89 – 95
Uranium Hexafluoride
Atomic Absorption Determination of Chromium Insoluble In 95 – 101
Uranium Hexafluoride
Atomic Absorption Determination of Chromium Insoluble In 101 – 102
Uranium Hexafluoride
Determination of Technetium-99 In Uranium Hexafluoride 102 – 110
Determination of Technetium-99 In Uranium Hexafluoride 103 – 110
Method for the Determiation of Gamma-Energy Emission Rate 111
from Fission Products in Uranium Hexafluoride
Method for the Determination of Gamma-Energy Emission Rate 112
from Fission Products in Uranium Hexafluoride
Determination of Metallic Impurities by ICP-AES 112 – 121
Determination of Metallic Impurities by ICP-AES 113 – 122
Determination of Molybdenum, Niobium, Tantalum, Titanium, 122 – 131
and Tungsten by ICP-AES
Determination of Molybdenum, Niobium, Tantalum, Titanium, 123 – 132
and Tungsten by ICP-AES
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. (For specific safeguard and safety consideration statements, see Section 67.)
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 The following documents of the issue in effect on date of material procurement form a part of this specification to the extent
referenced herein:
2.2 ASTM Standards:
C787 Specification for Uranium Hexafluoride for Enrichment
C799 Test Methods for Chemical, Mass Spectrometric, Spectrochemical, Nuclear, and Radiochemical Analysis of Nuclear-
Grade Uranyl Nitrate Solutions
C859 Terminology Relating to Nuclear Materials
C996 Specification for Uranium Hexafluoride Enriched to Less Than 5 % U
C1128 Guide for Preparation of Working Reference Materials for Use in Analysis of Nuclear Fuel Cycle Materials
C1219 Test Methods for Arsenic in Uranium Hexafluoride (Withdrawn 2015)
C1233 Practice for Determining Equivalent Boron Contents of Nuclear Materials
C1267 Test Method for Uranium by Iron (II) Reduction in Phosphoric Acid Followed by Chromium (VI) Titration in the
Presence of Vanadium
C1287 Test Method for Determination of Impurities in Nuclear Grade Uranium Compounds by Inductively Coupled Plasma
Mass Spectrometry
C1295 Test Method for Gamma Energy Emission from Fission and Decay Products in Uranium Hexafluoride and Uranyl Nitrate
Solution
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
C761 − 18
C1344 Test Method for Isotopic Analysis of Uranium Hexafluoride by Single-Standard Gas Source Mass Spectrometer Method
C1346 Practice for Dissolution of UF from P-10 Tubes
C1380 Test Method for the Determination of Uranium Content and Isotopic Composition by Isotope Dilution Mass
Spectrometry
C1413 Test Method for Isotopic Analysis of Hydrolyzed Uranium Hexafluoride and Uranyl Nitrate Solutions by Thermal
Ionization Mass Spectrometry
C1428 Test Method for Isotopic Analysis of Uranium Hexafluoride by Single–Standard Gas Source Multiple Collector Mass
Spectrometer Method
C1429 Test Method for Isotopic Analysis of Uranium Hexafluoride by Double-Standard Multi-Collector Gas Mass Spectrometer
C1441 Test Method for The Analysis of Refrigerant 114, Plus Other Carbon-Containing and Fluorine-Containing Compounds
in Uranium Hexafluoride via Fourier-Transform Infrared (FTIR) Spectroscopy
C1474 Test Method for Analysis of Isotopic Composition of Uranium in Nuclear-Grade Fuel Material by Quadrupole
Inductively Coupled Plasma-Mass Spectrometry
C1477 Test Method for Isotopic Abundance Analysis of Uranium Hexafluoride and Uranyl Nitrate Solutions by Multi-Collector,
Inductively Coupled Plasma-Mass Spectrometry
C1508 Test Method for Determination of Bromine and Chlorine in UF and Uranyl Nitrate by X-Ray Fluorescence (XRF)
Spectroscopy
C1539 Test Method for Determination of Technetium-99 in Uranium Hexafluoride by Liquid Scintillation Counting
C1561 Guide for Determination of Plutonium and Neptunium in Uranium Hexafluoride and U-Rich Matrix by Alpha
Spectrometry
C1636 Guide for the Determination of Uranium-232 in Uranium Hexafluoride
C1689 Practice for Subsampling of Uranium Hexafluoride
C1742 Test Method for Isotopic Analysis of Uranium Hexafluoride by Double Standard Single-Collector Gas Mass
Spectrometer Method
D1193 Specification for Reagent Water
D3084 Practice for Alpha-Particle Spectrometry of Water
E60 Practice for Analysis of Metals, Ores, and Related Materials by Spectrophotometry
2.3 American Chemical Society Specification:
Reagent Chemicals
2.4 Other Specifications:
Uranium HexafluorideHexafluoride: : Base Charges, Use Charges, Special Charges, Table of Enriching Services, Specifications,
and Packaging
USEC 651 Good Handling and Practices for UF
2.5 ANSI Standards:
ANSI N 14.1 Nuclear Material-Uranium Hexafluoride-Packaging for Transport
2.6 ISO Standards:
ISO 7195 Nuclear Energy-Packaging of Uranium Hexafluoride (UF ) for Transport
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms relating to the nuclear fuel cycle, refer to Terminology C859.
4. Significance and Use
4.1 Uranium hexafluoride is a basic material used to prepare nuclear reactor fuel. To be suitable for this purpose the material
must meet criteria for uranium content, isotopic composition, metallic impurities, hydrocarbon and halohydrocarbon content.
These test methods are designed to determine whether the material meets the requirements described in Specifications C787 and
C996.
5. Reagents
5.1 Purity of Reagents—Reagent grade chemicals shall be used in all procedures. Unless otherwise indicated, all reagents shall
conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where such
specifications are available. Other grades may be used, provided that it is first established that the reagent to be used is of
sufficiently high purity to permit its use without lessening the accuracy of the determination.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K. and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
United States Department of Energy, Oak Ridge, TN 37830.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036.
C761 − 18
5.2 Purity of Water—Unless otherwise indicated, references to water shall mean reagent water conforming to Specification
D1193.
6. Rejection
6.1 Rejection or acceptance criteria are described in Specifications C787 and C996.
7. Safety Considerations
7.1 Since UF is radioactive, toxic, and highly reactive, especially with reducing substances and moisture (see Uranium
Hexafluoride: Handling Procedures and Container Criteria, sections 2.42.4 through 2.62.6),), appropriate facilities and practices for
sampling and analysis must be provided.
7.2 Hydrofluoric acid is a highly corrosive acid that can severely burn skin, eyes, and mucous membranes. Hydrofluoric acid
is similar to other acids in that the initial extent of the burn depends on the concentration, the temperature, and the duration of the
contact with the acid. Hydrofluoric acid differs from other acids because the fluoride ion readily penetrates the skin, causing
destruction of deep issuetissue layers. Unlike other acids that are rapidly neutralized, hydrofluoric acid reactions with tissue may
continue for days if left untreated. Due to the serious consequences of hydrofluoric acid burns, prevention of exposure or injury
of personnel is the primary goal. Utilization of appropriate laboratory controls (hoods) and wearing adequate personal protective
equipment to protect from skin and eye contact Familiarization and compliance with the Safety Data Sheet is essential.
7.3 Committee C-26C26 Safeguards Statement:
7.3.1 The material (uranium hexafluoride) to which these test methods apply, is subject to nuclear safeguards regulations
governing its possession and use. The following analytical procedures in these test methods have been designated as technically
acceptable for generating safeguards accountability measurement data: Gravimetric Determination of Uranium; Titrimetric
Determination of Uranium; All Isotopic Analyses.
7.3.2 When used in conjunction with appropriate certified Reference Materials (CRMs), these procedures can demonstrate
traceability to the national measurement base. However, adherence to these procedures does not automatically guarantee regulatory
acceptance of the resulting safeguards measurements. It remains the sole responsibility of the user of these test methods to assure
that its application to safeguards has the approval of the proper regulatory authorities.
SUBSAMPLING OF URANIUM HEXAFLUORIDE
7. Scope
ε1
7.1 This test method has been discontinued (see C 761–04 ). The subsampling of UF from bulk sample containers into smaller
containers suitable for laboratory analyses has been published as a separate Practice, C1689.
8. Scope
ε1
8.1 This test method has been discontinued (see C761 – 04 ). The subsampling of UF from bulk sample containers into
smaller containers suitable for laboratory analyses has been published as a separate Practice C1689.
GRAVIMETRIC DETERMINATION OF URANIUM
9. Scope
9.1 Practice C1346 is applicable to the hydrolysis of uranium hexafluoride in polychlorotrifluoroethylene (P10) tubes. The
following test method is then applicable to the direct gravimetric determination of uranium.
10. Summary of Test Method
10.1 A sample of uranium hexafluoride is weighed, cooled in liquid nitrogen, and hydrolyzed with water. The uranyl fluoride
solution produced is evaporated to dryness and converted to uranic oxide by pyrohydrolysis. The uranium content is determined
from the weight of the uranium oxide after correcting for stoichiometry based on isotopic content, ignition conditions, and
nonvolatile impurities. Ref. (1-4).
11. Interferences
11.1 Nonvolatile impurities affect the accuracy of the method and must be measured by spectrographic analysis with corrections
applied.
Type 1 and 2 water have been found to be suitable.
The boldface numbers in parentheses refer to a list of references at the end of these test methods.
C761 − 18
12. Apparatus
12.1 Polytrifluorochloroethylene (PTFCE) Sample Tube, TFCE Gasket, Flare Nut, and Plug, see Fig. 1.
12.2 Platinum Boat and Cover—The cover should be platinum gauze (52 mesh) and shaped to cover the boat (Fig. 2).
12.3 Muffle Furnace, must be capable of operating continuously at 875°C and maintain this temperature within 625°C. The
furnace shall be equipped with a steam supply that is passed through a tube furnace to preheat the steam to 875°C.
12.4 Tube Furnace, must be capable of operating continuously at 875°C and maintain this temperature within 25°C.
12.5 Infrared Heat Lamps, 250 watts.
12.6 Analytical Balance.
12.7 Vacuum Oven.
12.8 Dewar Flask, stainless steel.
12.9 Spatula, platinum.
12.10 PTFCE Rod, 120 mm long and 1.6 mm in diameter.
12.11 Forceps, platinum tipped.
12.12 Jig, suitable for holding the TFCE sample tube so that it can be opened with a wrench.
12.13 Box Wrench, to fit sample tube plug.
12.14 Beaker, stainless steel, 125 mL capacity.
13. Reagents
13.1 Liquid Nitrogen.
13.2 Nitric Acid (sp gr 1.42)—concentrated nitric acid (HNO ).
13.3 Nitric Acid (4M)—Mix 500 mL of concentrated HNO with 1500 mL of distilled water.
13.4 Detergent.
14. Sampling
14.1 A UF sample is taken as described in Practice C1689.
FIG. 1 Example of a Polychlorotrifluoroethylene P-10 Tube
C761 − 18
FIG. 2 Platinum Boat and Cover
15. Procedure
15.1 Inspect the PTFCE sample tube for leaks.
NOTE 2—An indication of a leak is a yellow-green residue on the flare nut and cap or a yellow discoloration in the tube. Discard the sample if a leak
is indicated.
15.2 Allow the sample tube to stand overnight in the laboratory.
15.3 Wipe the sample tube with a lint-free tissue to remove any moisture or foreign material that might be adhering.
15.4 Weigh the sample tube to the nearest 0.1 mg.
15.5 Heat the platinum boat and screen in the pyrohydrolysis furnace at 875°C for 20 min.
15.6 Cool the platinum boat and store in a desiccator for 40 min. Weigh the boat and screen to the nearest 0.1 mg.
15.7 Freeze the sample by immersing the sample tube in liquid nitrogen for 10 min.
15.8 Add enough chilled water to the tared platinum boat to immerse the sample tube (about 50 mL).
15.9 Place the sample tube in the jig and loosen the plug with the box wrench.
15.10 Remove the sample tube from the jig and unscrew the plug while holding the sample tube in an upright position.
15.11 Remove the flare nut from the sample tube and immerse the tube and gasket in the chilled water in the tared platinum
boat.
15.12 Let the gasket remain in the chilled water about 30 min.
15.13 Remove the gasket with the forceps and rinse well with deionized water into the boat.
15.14 Place the plug-nut assembly and gasket into a stainless steel beaker for drying.
15.15 Allow the tube to remain in the water until the UF has been hydrolyzed (2 to 4 h).
C761 − 18
15.16 Remove the tube from the sample solution by inserting the TFCE rod or platinum spatula into the tube and lifting directly
above the boat.
15.17 Rinse the sample tube with deionized water into the boat using extreme care to prevent splashing.
15.18 Cover the sample boat containing the UO F solution with the matching cover shown in Fig. 2. Place under the infrared
2 2
head lamps and evaporate to dryness for 16 h.
15.19 Shake the excess water from the sample tube and place in the stainless beaker containing the plug-nut assembly and
gasket.
15.20 Dry the sample tube parts in the vacuum oven at 80°C.
15.21 Allow the unassembled parts to sit in the room overnight.
15.22 Assemble the empty sample tube and weigh to the nearest 0.1 mg.
15.23 Disassemble the sample tube and soak the tube and gasket in 4M HNO at 75° to 80°C for 1 h.
15.24 Rinse with deionized water and place in the stainless steel beaker.
15.25 Clean the metal parts with detergent and rinse with deionized water and acetone.
15.26 Place the metal parts to the stainless steel beaker and dry all parts in the vacuum oven at 80°C overnight.
15.27 Reassemble the sample tube for the next sample.
15.28 Set the temperatures of the furnace and tube furnace at 875°C.
15.29 Establish a steam flow to the furnace equal to 1 L of water per hour.
15.30 Place the boat into the furnace with the platinum cover on the boat and pyrohydrolyze the sample for 1 h.
15.31 Remove the boat from the furnace, cool, and place in a desiccator while still warm.
15.32 Desiccate the sample for 1 h and weigh quickly to the nearest 0.1 mg.
15.33 Transfer a portion of the U O residue to a vial and submit for spectrographic analysis to determine the weight of
3 8
nonvolatile impurities.
15.34 Place the platinum boat in hot 4M HNO for 3 to 4 h and rinse with deionized water acetone.
16. Calculation
16.1 Calculate the weight fraction of uranium in the sample as follows:
gU/g UF 5 ~A 2 ~AB!!~Gravimetric Factor!/W (1)
where:
Gravimetric Factor = gU/g U O which varies with isotopic composition. Theoretical stoichiomtry for U O cannot be
3 8 3 8
assumed and the actual gU/g U O must be established by potentiometric titration (1–4). (Tri-diffusion
3 8
plant committee with DOE approval has established 0.8479 g U/g U O by titration as the factor for
3 8
natural uranium,
Gravimetric Factor = gU/g U O which varies with isotopic composition. Theoretical stoichiometry for U O cannot be
3 8 3 8
assumed and the actual gU/g U O must be established by potentiometric titration (1-4). (Tri-diffusion
3 8
plant committee with DOE approval has established 0.8479 gU/g U O by titration as the factor for
3 8
natural uranium,
A = grams of U O from the pyrohydrolysis of UO F ,
3 8 2 2
B = grams of impurity metal oxides per gram of U O ,
3 8
W = corrected sample weight in grams.
The correction is for the combined effects of cover gas trapped over the UF in the sample tube and the air buoyancy correction
(5). The following equation has been determined for the sample tube in Fig. 1 and the subsampling conditions described in Practice
C1689. The correction equation is applicable for sample weights in the range of 7 to 13 g.
W 5 ~1.00047!x 2 0.0058 (2)
where:
x = observed UF sample weight, g.
17. Precision and Bias
17.1 Precision—The precision within a laboratory and between laboratories was established by analyzing 15 samples at each
laboratory. The sampling scheme is shown in Table 1. Within a laboratory, based on 15 measurements made on separate days the
relative standard deviation is 0.021 %. The results from all the laboratories are shown in Table 2.
C761 − 18
TABLE 2 Results of Interlaboratory Study—U in UF
Analysis Site %U in UF
GAT ORGDP PGDP
Subsampled at GAT:
67.600 67.619 67.589
67.601 67.574 67.575
67.583 67.607 67.612
67.611 67.600 67.612
67.618 67.606 Sample Lost
Subsampled at ORGDP:
67.614 67.580 67.611
67.611 67.621 67.598
67.587 67.600 67.501
67.599 67.606 67.610
67.617 67.596 67.624
Subsampled at PGDP:
67.616 67.588 67.591
67.586 67.602 67.620
67.573 67.612 67.612
67.614 67.606 67.612
67.607 67.586
Mean and Standard Deviation:
67.602 ± 0.014 67.601 ± 0.013 67.603 ± 0.014
17.2 Bias—To establish an estimate of bias for the gravimetric method, a series of comparative analyses of UF control batches
were made using the gravimetric and potentiometric titration methods. The potentiometric titration was used as the reference
method because the uranium was measured directly using NIST potassium dichromate. The results are shown in Table 3.
TABLE 3 Determination of Uranium in Uranium Hexafluoride—Comparison of Gravimetric and Potentiometric Titration Methods
%Uranium
Control UF Number of Bias
Method
A B
Date Measurements Estimate
Mean SD
9/78 Gravimetric 24 67.610 0.009 −0.001
Potentiometric Titration 9 67.611 0.015
5/82–8/82 Gravimetric 30 67.596 0.010 −0.009
Potentiometric Titration 8 67.605 0.011
7/83–9/83 Gravimetric 25 67.610 0.006 + 0.005
7/83–9/83 Gravimetric 25 67.610 0.006 +0.005
Potentiometric Titration 8 67.605 0.010
A
Control UF used in 9/78 was a different batch of material from that used in 1982 and 1983.
B
Potentiometric titration results are used as the reference values for the bias estimates.
Standard reference material, now available as NIST SRM 136e.
C761 − 18
TITRIMETRIC DETERMINATION OF
URANIUM
17. Scope
17.1 A sample of the U O produced by the hydrolysis of the UF and ignition of the resulting UO F is analyzed according
3 8 6 2 2
to Test Method C1267.
18. Scope
18.1 A sample of the U O produced by the hydrolysis of the UF and ignition of the resulting UO F is analyzed according
3 8 6 2 2
to Test Method C1267.
PREPARATION OF HIGH-PURITY U O
3 8
18. Scope
18.1 High purity U O can be prepared according to Preparation C1128. High purity uranium is needed for a blank matrix for
3 8
analyses using ICP-MS, ICP-AES, AA, XRF, and MS equipment.
19. Scope
19.1 High purity U O can be prepared according to Preparation C1128. High purity uranium is needed for a blank matrix for
3 8
analyses using ICP-MS, ICP-AES, AA, XRF, and MS equipment.
ISOTOPIC ANALYSIS
19. Scope
19.1 The isotopic composition can be determined on either gaseous UF or on hydrolyzed UF .
6 6
19.2 For gaseous UF , using single collector mass spectrometer instruments, Test Methods C1344 and C1742 have been
developed and can be used for single or double standard method respectively. For multi-collector instruments, Test Methods C1428
and C1429, using single or double standard can be used.
19.3 For hydrolyzed UF , methods using Thermal Ionization Mass Spectrometry (TIMS) have been developed and can be used:
Test Methods C1413 and C1380. Methods using ICP-MS can also be used: Test Methods C1474 and C1477.
20. Scope
20.1 The isotopic composition can be determined on either gaseous UF or on hydrolyzed UF .
6 6
20.2 For gaseous UF , using single collector mass spectrometer instruments, Test Methods C1344 and C1742 have been
developed and can be used for single or double standard method respectively. For multi-collector instruments, Test Methods C1428
and C1429, using single or double standard can be used.
20.3 For hydrolyzed UF , methods using Thermal Ionization Mass Spectrometry (TIMS) have been developed and can be used:
Test Methods C1413 and C1380. Methods using ICP-MS can also be used: Test Methods C1474 and C1477.
DETERMINATION OF HYDROCARBONS, CHLOROCARBONS, AND HALOHYDROCARBONS
20. Scope
20.1 The determination of some forms of hydrocarbons, chlorocarbons, and halohydrocarbons in UF vapor can be performed
using Test Method C1441. As an alternative, a mass spectrometry technique may be used and is detailed below. Although this test
method is only semiquantitative, it is adequate for certifying that the subject impurities do not exceed 0.01 mol % of the UF .
21. Scope
21.1 The determination of some forms of hydrocarbons, chlorocarbons, and halohydrocarbons in UF vapor can be performed
using Test Method C1441. As an alternative, a mass spectrometry technique may be used and is detailed below. Although this test
method is only semiquantitative, it is adequate for certifying that the subject impurities do not exceed 0.01 mol % of the UF .
22. Summary of Test Method
22.1 UF is admitted to a mass spectrometer through a gas sample leak, and magnetic scanning is employed to record a spectrum
of peaks. A representative group of recorded peaks is compared to the same peaks in a pure UF standard scan to determine whether
appreciable ion fragments from subject impurities are present.
C761 − 18
23. Interferences
23.1 If detectable impurities are present, a complete mass scan of the range from 12 to 400 is performed. All impurities are then
identified from their cracking patterns, and calculations are performed using ionization efficiency factors for the compounds
present. Since cracking patterns vary with ionization potential and ionization efficiencies vary with focus conditions, this
measurement can only be performed by one proficient in analytical mass spectrometery.
24. Apparatus
24.1 A mass spectrometer with resolution adequate to distinguish between adjacent peaks at m/e = 400 is required. For
example, a 152-mm radius, 60-deg, Nier-type spectrometer modified for spectrum recording (6) is suitable. The sample inlet
system should be of nickel or Monel, equipped with an adjustable viscous-flow or molecular leak for delivering the sample to the
ion source.
24.2 The ion source must be fabricated from nonmagnetic material such as Nichrome V, and must be designed so it can be
disassembled for cleaning. The magnetic field of the analyzer magnet must be continuously variable from about 200 to 6500 gauss.
A single ion collector electrode is suitable, and a vibrating-reed electrometer and 304-mm strip chart recorder are optimum for
amplifying and recording ion signals.
24.3 It is quite possible that quadrupole or time-of-flight instruments could be adapted to this measurement.
25. Procedure
25.1 UF Standard Measurements:
25.1.1 Select a standard material that has been given repetitive flash purifications to rid it of all volatile impurities. Isotopic UF
standards usually fall in this category.
25.1.2 With the electrometer sensitivity set at ⁄100 of the most sensitive usable operating range, adjust the gas flow to the ion
+
source to record a mass (Note 3) 333 peak (UF ) approximately 80 % of full scale (80 divisions).
NOTE 3—The term “mass” in this procedure alludes to m/e, the mass-to-charge ratio (see also Terminology C859).
+ ++
25.1.3 Measure the ratio of mass 333 (UF ) to 147.5 (UF ). Mass 333 is measured on a sensitivity range one-hundredth that
5 3
of mass 147.5. Depending on focus conditions, a ratio of the order of 10 is obtained. Measure this ratio only once per day and
use for calculating results of all samples analyzed that day.
25.1.4 With the electrometer sensitivity set at ⁄100 of the most sensitive usable operating range, increase the gas flow to provide
an output signal of approximately 80 divisions at the 147.5 mass position. This gives a detection limit of the order of 2 ppm per
+ ++
chart division: 50 to 100 due to ratio between UF and UF , 100 due to sensitivity shunts, and approximately 80 on the recorder
5 3
chart.
25.1.5 Record a scan of mass range from 12 to 150 using the most sensitive usable operating shunt, and use this scan as a
background for all samples analyzed that day.
25.2 UF Sample Measurement:
25.2.1 Introduce the sample to the spectrometer source such that an output intensity of approximately 80 chart divisions is
++
obtained at the 147.5 mass number (UF ), using ⁄100 the most sensitive usable operating range.
25.2.2 Record a scan of mass range from 12 to 150 using the most sensitive usable operating range.
25.2.3 Repeat 24.2.125.2.1 and 24.2.225.2.2 for each sample to be analyzed that day.
26. Calculation
26.1 Due to mass spectrometer cracking patterns, low-mass ion fragments are produced from all compounds, even the
high-mass ones.
26.2 It is practical to look for a representative group of such ion fragments at specific masses. Thus, initially monitor the
following masses for purposes of this procedure:
Mass Number Positively Charged Ion
Fragment
15 CH
26 C H
2 2
27 C H
2 3
31 CF
43 C H
3 7
47 CCl
49 CCl
69 CF
26.3 Read sample intensities for the representative ion fragments from the recorder chart.
26.4 Subtract the background intensities observed on the pure standard from respective sample intensities.
C761 − 18
26.5 Examine the net intensity at each of the eight mass numbers. (It will be recalled that one recorder chart division of net
intensity is equivalent to about 2 ppm on a UF basis; however, ionization efficiencies of compounds differ, and a specific ion
fragment may result from many different compounds. Thus, the net intensity at a specific mass number is only qualitative and not
a quantitative measurement of impurity.)
26.5.1 If the net ion intensity does not exceed 2 ppm at any of the mass positions, report the sample as containing less than 0.01
mol % of the subject impurities.
26.5.2 Where detectable impurities are apparent, perform a complete mass scan of the range from 12 to 400, identify impurities,
and perform calculations using ionization efficiency factors for the compounds present.
27. Reliability
27.1 This simplified procedure was designed specifically to certify that a UF sample contains less than 0.01 mol %
hydrocarbons, chlorocarbons, and partially substituted halohydrocarbons. Thus, the procedure is qualitative rather than quantitative
in cases where the impurity level is below 100 ppm.
27.2 The detectability limit for any ion fragment is about 2 ppm. The detectability limit for the parent compound could be
greater or less than 2 ppm depending on ionization efficiency and cracking pattern. If one of the subject compounds were present
to 10 ppm or more, it would be evident from monitoring the eight masses. Thus, a quoted result of less than 0.01 mol % is
conservative.
27.3 The results are considered quantitative when the impurity being determined is present to a level greater than 100 ppm. In
such instances the impurity is identified and measured. The 95 % symmetrical confidence interval for such a measurement is
650 % of the quoted impurity.
DETERMINATION OF ANTIMONY
27. Scope
27.1 The Atomic Absorption test method has been discontinued (see C761–96). Antimony can be determined by ICP-MS. Test
Method C1287 can be used.
28. Scope
28.1 The Atomic Absorption test method has been discontinued (see C761–96). Antimony can be determined by ICP-MS. Test
Method C1287 can be used.
DETERMINATION OF BROMINE
28. Scope
28.1 The Spectrophotometric test method has been discontinued (see C761–96). Bromine can be determined by X-Ray
spectroscopy. Test Method C1508 can be used.
29. Scope
29.1 The Spectrophotometric test method has been discontinued (see C761–96). Bromine can be determined by X-Ray
spectroscopy. Test Method C1508 can be used.
DETERMINATION OF CHLORINE
29. Scope
29.1 Chlorine can be determined by X-Ray Spectroscopy. Test Method C1508 can be used. Chlorine can also be determined by
titrimetry. This test method is described below. It is applicable over a range from 10 to 100 ppm chlorine; however, higher
concentrations can be measured by appropriate sample dilution.
30. Scope
30.1 Chlorine can be determined by X-Ray Spectroscopy. Test Method C1508 can be used. Chlorine can also be determined by
titrimetry. This test method is described below. It is applicable over a range from 10 to 100 ppm chlorine; however, higher
concentrations can be measured by appropriate sample dilution.
31. Summary of Test Method
31.1 The test method consists of treating a hydrolyzed sample of UF with ferrous sulfate in sulfuric acid solution to reduce
chlorates, and then with potassium permanganate to liberate free chlorine. The chlorine gas is carried by a nitrogen stream into
a potassium iodide solution, and the liberated iodine is titrated with sodium thiosulfate. Bromine, if present, is determined
separately, and a correction is applied to the chlorine result.
C761 − 18
31.2 It is recommended that the potassium iodide-sodium acetate solution be made up fresh once each week. Any color change
signals the need for a new solution. As the solution ages, the blank result increases; therefore, the same potassium iodide solution
is used for both sample and blank.
31.3 If the sample solution is allowed to boil too vigorously when chlorine gas is being released, liquid droplets may be carried
by the nitrogen stream into the potassium iodide solution, resulting in sample bias.
32. Apparatus
32.1 Distillation Apparatus, shown in Fig. 3.
33. Reagents
33.1 Boric Acid (H BO ), reagent grade, crystal or powder.
3 3
33.2 Ferrous Sulfate Solution—5 g FeSO × 7H O dissolved in 500 mL of 3.6 M sulfuric acid.
4 2
33.3 Potassium Iodide (KI), reagent grade.
33.4 Potassium Iodide-Sodium Acetate Solution—Dissolve 100 g of KI and 100 g of NaC H O × 3H O in distilled water and
2 3 2 2
dilute to 2 L.
33.5 Potassium Permanganate Solution (1 %)—Prepare a 1 % solution of KMnO in water.
33.6 Sodium Acetate (NaC H O ), reagent grade.
2 3 2
33.7 Sodium Thiosulfate Solution (0.025 N)—Prepare a 0.025 N solution of Na S O in water.
2 2 3
33.8 Starch Indicator Solution, pH 7.
33.9 Sulfuric Acid (sp gr 1.84)—Concentrated sulfuric acid (H SO ).
2 4
34. Procedure
34.1 Sample Preparation:
34.1.1 Hydrolyze the sample of UF with distilled water. Approximately 250 g of UF from a nickel knockout container may
6 6
be hydrolyzed to provide a sample for various chemical measurements, or about 20 g from two polychlorotrifluoroethylene sample
tubes may be hydrolyzed to provide the chlorine sample. The procedure for hydrolyzing the contents of the polychlorotrifluoro-
ethylene tubes is described here.
34.1.1.1 Immerse the tubes in liquid nitrogen and cool for 10 min.
34.1.1.2 Remove the top flare plugs and collars, and place the tubes into a platinum dish or a polychlorotrifluoroethylene beaker
containing 100 mL of chilled distilled water.
34.1.1.3 After hydrolysis of the UF , remove the polychlorotrifluoroethylene tubes and rinse with distilled water. Add the rinse
solution to the UO F solution.
2 2
34.2 Analysis:
34.2.1 Fill the graduated cylinder in Fig. 2 to the 150-mL level with KI-NaC H O solution. Then connect the delivery tube so
2 3 2
its tip is near the bottom of the solution in the receiving graduate.
FIG. 3 Apparatus for Distillation of Chlorine
C761 − 18
34.2.2 Dispense 20 g of H BO into the 1-L round-bottom flask.
3 3
34.2.3 Transfer the sample solution containing UO F from approximately 20 g of UF in 100 mL of solution to the flask.
2 2 6
34.2.4 Add 10 mL of concentrated H SO (sp gr 1.84) to the flask, and swirl the contents for mixing.
2 4
34.2.5 Add 10 mL of the FeSO solution, rinse the mouth of the flask, and connect the flask immediately to the apparatus as
in Fig. 3.
34.2.6 Initiate nitrogen flow through the solution at a rate of 2 to 3 bubbles per second and start the water flow through the
condenser.
34.2.7 Heat the contents of the flask until boiling and allow to boil for 30 s.
34.2.8 Remove the heat, add 10 mL of 1 % KMnO solution through the sidearm, and close the sidearm by clamping the rubber
tube that is attached to the end of the sidearm.
34.2.9 Reapply heat and allow the contents of the flask to simmer for 5 min.
34.2.10 Remove the heat, but continue the nitrogen purge for an additional 5 min.
34.2.11 Rinse the delivery tube into the receiving graduate and transfer the contents of the graduate to a 300-mL Erlenmeyer
flask. Add 1 mL of starch solution and titrate the iodine with 0.025 N Na S O solution to the starch end point. (The iodine may
2 2 3
be measured spectrophotometrically rather than titrimetrically.)
34.2.12 Perform a blank analysis by carrying 100 mL of distilled water through procedural steps, 33.2.134.2.1 through
33.2.1134.2.11, and subtracting from the sample titration.
35. Calculation
35.1 Calculate the concentration of chlorine in ppm chlorine on a uranium basis as follows:
Cl, ppm 5 V 2 V N 0.03545 310 /S
~ ! ~ ! ~ !
1 2
where:
V = millilitres of thiosulfate for sample,
V = millilitres of thiosulfate for blank,
N = normality of Na S O solution, and
2 2 3
N = normality of Na S O solution,
2 2 3
0.03545 = grams of chlorine per milliequivalents, and
S = grams of uranium.
36. Reliability
36.1 The precision at the 95 % confidence level is 610 % at the 100-ppm level.
DETERMINATION OF SILICON AND PHOSPHORUS
36. Scope
36.1 Phosphorus and Silicon can be determined by ICP-MS (see Test Method C1287).
36.2 Silicon can be analyzed by Atomic Absorption without matrix separation.
36.3 Phosphorus and Silicon can be analyzed by spectrophotometry (as described below). With these procedures about 0.5 μg
silicon or phosphorus per gram of uranium can be detected.
37. Scope
37.1 Phosphorus and Silicon can be determined by ICP-MS (see Test Method C1287).
37.2 Silicon can be analyzed by Atomic Absorption without matrix separation.
37.3 Phosphorus and Silicon can be analyzed by spectrophotometry (as described below). With these procedures about 0.5 μg
silicon or phosphorus per gram of uranium can be detected.
38. Summary of Test Method
38.1 The test methods are based on the development of the color known as molybdenum blue obtained by the reduction of
silico- or phosphomolybdate ions.
38.2 Reduction of the silico- or phosphomolybdate ions with a combination 1-amino-2-naphthol-4-sulfonic acid, sodium sulfite,
sodium pyrosulfite solution produces the same molybdenum blue complex that is measured spectrophotometrically, directly in the
uranium solution at 710 nm. One gram of uranium absorbs slightly at this wavelength, but its absorbance is easily corrected by
the use of an additional aliquot to which no reducing agent is added as the blank.
C761 − 18
39. Interferences
39.1 Phosphate interference in the silicon determination is eliminated by the addition of oxalic acid to decompose any
phospho-molybdate formed. Silicon in small amounts does not interfere in the phosphorus analysis since silicomolybdate does not
form at the acid concentration at which the phosphomolybdic acid is formed.
39.2 Fluoride, which would be a serious interference in the silicon analysis, is complexed with boric acid. A high concentration
of silicon which could interfere in the phosphorus analysis is prevented by handling hydrolyzed UF samples in platinum or plastic.
Other potential interferences rarely present in significant amounts are arsenic and tungsten.
40. Apparatus
40.1 Polyethylene Bottles, 100 and 500 mL.
40.2 Polyethylene Beakers, 100 mL.
40.3 Polyethylene Pipets; 1, 2, 5, and 10 mL.
40.4 Spectrophotometer, equipped with 1 and 5 cm cells as described in Practice E60.
41. Reagents
41.1 Ammonium Hydroxide Silicon-Free—Distill 500 mL of saturated NH OH through plastic tubing into 300 mL of distilled
water.
41.2 Ammonium Molybdate Solution (10 %)—Dissolve 100 g of reagent grade (NH ) Mo O in water, and dilute the solution
4 6 7 24
to 1 L with distilled water.
NOTE 4—Not all commercially available (NH ) Mo O is suitable. Material supplied by J. T. Baker Chemical Co. or Baker and Adamson Products,
4 6 7 24
however, has been found to be satisfactory consistently.
41.3 Boric Acid Solution (5 %)—Dissolve 25 g of reagent grade H BO in water, and dilute the solution to 500 mL.
3 3
41.4 Oxalic Acid Solution (5 %)—Dissolve 25 g of reagent grade H C O in water, and dilute the solution to 500 mL. (This
2 2 4
solution is not required for determination of phosphorus.)
41.5 Phosphorus Standard Solution (25 μg P/mL)—Dissolve 4.6422 g of ammonium dihydrogen phosphate [(NH )H PO ] in
4 2 4
distilled water and dilute the solution to 1 L with distilled water. Transfer 20 mL of this solution to a 1-L volumetric flask and dilute
to 1 volume with distilled water to obtain a solution containing 2.0 μg P/mL.
41.6 Reducing Mix—Dissolve 0.1 g of 1-amino-2-naphthol-4-sulfonic acid, 1.0 g of sodium sulfite (Na SO ), and 10.0 g of
2 3
sodium pyrosulfite (Na S O ) in water; then dilute the solution to 100 mL.
2 2 5
41.7 Silicon Standard Solution (2.5 μg Si/mL)—Dissolve 10.6 mg of precipitated silica (SiO ) and 0.5 g of sodium hydroxide
(NaOH) in a platinum dish. Transfer the solution to a 2-L plastic bottle and dilute to volume.
NOTE 5—Silicon in solution as sodium silicate is not stable when stored in polyethylene bottles. New standard solutions should be prepared monthly.
41.8 Sulfuric Acid—Boric Acid Solution (10 % H SO —4 % H BO )—Dissolve 20 g of reagent grade H BO in water. Add 50
2 4 3 3 3 3
mL of concentrated H SO (sp gr 1.84) and dilute the solution to 500 mL.
2 4
41.9 Uranium Oxide (U O , UO , or UO ), phosphorus- and silicon-free.
3 8 2 3
NOTE 6—All standard solutions should be made and stored in plastic containers to prevent silicon contamination from glassware.
42. Procedure
42.1 Sample Preparation:
42.1.1 Hydrolyze a weighed portion of 6 to 10 g of UF in a platinum boat in 80 mL of distilled water as described in 14.715.7
through 14.1715.17.
42.1.2 Transfer the solution to a 100-mL plastic bottle and dilute to 100 mL.
42.1.3 Transfer an aliquot equivalent to 1 g of UF to a 100-mL TFE-fluorocarbon beaker, and add 1 mL of 6 N H SO .
6 2 4
(Phosphorus aliquots should contain 1 to 50
...



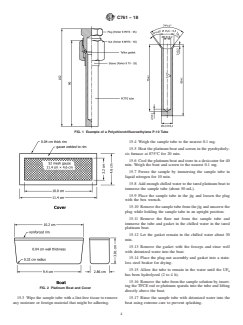




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...