ASTM C1287-18
(Test Method)Standard Test Method for Determination of Impurities in Nuclear Grade Uranium Compounds by Inductively Coupled Plasma Mass Spectrometry
Standard Test Method for Determination of Impurities in Nuclear Grade Uranium Compounds by Inductively Coupled Plasma Mass Spectrometry
SIGNIFICANCE AND USE
5.1 This test method is capable of measuring the elements listed in Table 1, some of which are required by Specifications C753, C776, C787, C788, C967 and C996.
SCOPE
1.1 This test method covers the determination of 67 elements in uranium dioxide samples and nuclear grade uranium compounds and solutions without matrix separation by inductively coupled plasma mass spectrometry (ICP-MS). The elements are listed in Table 1. These elements can also be determined in uranyl nitrate hexahydrate (UNH), uranium hexafluoride (UF6), triuranium octoxide (U3O8) and uranium trioxide (UO3) if these compounds are treated and converted to the same uranium concentration solution.
1.2 The elements boron, sodium, silicon, phosphorus, potassium, calcium and iron can be determined using different techniques. The analyst's instrumentation will determine which procedure is chosen for the analysis.
1.3 The test method for technetium-99 is given in Annex A1.
1.4 The values stated in SI units are to be regarded as standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Warning—The ICP-MS is a source of intense ultra-violet radiation from the radio frequency induced plasma. Protection from radio frequency radiation and UV radiation is provided by the instrument under normal operation.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Dec-2017
- Technical Committee
- C26 - Nuclear Fuel Cycle
- Drafting Committee
- C26.05 - Methods of Test
Relations
- Effective Date
- 01-Jan-2018
- Replaces
ASTM C1219-05(2009) - Standard Test Methods for Arsenic in Uranium Hexafluoride (Withdrawn 2015) - Effective Date
- 09-Jun-2015
- Effective Date
- 01-Jan-2024
- Effective Date
- 01-Mar-2020
- Effective Date
- 01-Mar-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Feb-2016
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Jul-2015
- Effective Date
- 15-Jun-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Jan-2014
- Effective Date
- 01-Jun-2013
- Effective Date
- 01-May-2013
Overview
ASTM C1287-18 is the internationally recognized standard test method for determining impurities in nuclear grade uranium compounds using inductively coupled plasma mass spectrometry (ICP-MS). Developed by ASTM Committee C26 on Nuclear Fuel Cycle, this method enables the measurement of up to 67 elements in uranium dioxide samples, as well as other nuclear-grade uranium compounds and solutions. It applies without the need for matrix separation and supports high-purity uranium products used throughout the nuclear industry.
Key Topics
- Comprehensive Impurity Detection: Measures 67 trace and major impurity elements in uranium compounds, including uranium dioxide (UO₂), triuranium octoxide (U₃O₈), uranium trioxide (UO₃), uranyl nitrate hexahydrate (UNH), and uranium hexafluoride (UF₆).
- ICP-MS Technology: Utilizes the sensitivity and multi-element capability of ICP-MS, allowing simultaneous detection and quantitation of a broad range of elements at very low concentrations.
- Sample Flexibility: Accommodates solid uranium oxides, uranium solutions, and other compounds after appropriate sample preparation to match uranium concentrations.
- Alternate Techniques: For certain elements-such as boron, sodium, silicon, phosphorus, potassium, calcium, and iron-analysts may select alternative approved methodologies based on instrumentation.
- Regulatory Compliance: Supports quality and specification requirements outlined in related ASTM specifications for nuclear-grade uranium products.
- International Standardization: Developed in accordance with World Trade Organization Technical Barriers to Trade (WTO TBT) Committee principles.
Applications
ASTM C1287-18 is vital for ensuring the quality and safety of materials used in the nuclear fuel cycle. Key applications include:
- Quality Control: Nuclear fuel manufacturers use this method for routine quality assurance of uranium dioxide powders and sintered pellets to meet stringent impurity limits.
- Certification of Uranium Compounds: Helps laboratories certify nuclear-grade uranyl nitrate, uranium hexafluoride, and other uranium compounds according to international and ASTM specifications.
- Regulatory Reporting: Supports compliance for suppliers and processors regarding impurity content in nuclear material shipments.
- Research and Development: Enables precise impurity profiling to support new process development or material performance evaluations in nuclear applications.
- Environmental and Safety Monitoring: Assists in monitoring trace element contamination and validating purification processes in uranium processing facilities.
Related Standards
The methodology in ASTM C1287-18 is directly relevant to and referenced by several key nuclear industry standards and specifications, including:
- ASTM C753 - Specification for Nuclear-Grade, Sinterable Uranium Dioxide Powder
- ASTM C776 - Specification for Sintered Uranium Dioxide Pellets for Light Water Reactors
- ASTM C787 - Specification for Uranium Hexafluoride for Enrichment
- ASTM C788 - Specification for Nuclear-Grade Uranyl Nitrate Solution or Crystals
- ASTM C967 - Specification for Uranium Ore Concentrate
- ASTM C996 - Specification for Uranium Hexafluoride Enriched to Less than 5% 235U
- ASTM D1193 - Specification for Reagent Water
- ASTM C859 - Terminology Relating to Nuclear Materials
Practical Value
- Assurance of Purity: Critical for preventing reactor performance issues and regulatory non-compliance caused by impurities.
- Trace-Level Sensitivity: ICP-MS delivers detection limits suitable for current and emerging nuclear industry demands.
- Versatile Application: Suitable for a range of uranium forms and compounds across the fuel cycle-from conversion to fabrication.
- Support for International Trade: Ensures uranium products meet global standards, facilitating international commerce and cooperation in the nuclear sector.
Keywords: ASTM C1287-18, determination of impurities, uranium compounds, nuclear grade, ICP-MS, uranium dioxide, uranium hexafluoride, uranyl nitrate, trace elements, nuclear fuel, quality control, nuclear standards, impurity analysis.
Buy Documents
ASTM C1287-18 - Standard Test Method for Determination of Impurities in Nuclear Grade Uranium Compounds by Inductively Coupled Plasma Mass Spectrometry
REDLINE ASTM C1287-18 - Standard Test Method for Determination of Impurities in Nuclear Grade Uranium Compounds by Inductively Coupled Plasma Mass Spectrometry
Get Certified
Connect with accredited certification bodies for this standard

DNV
DNV is an independent assurance and risk management provider.

Lloyd's Register
Lloyd's Register is a global professional services organisation specialising in engineering and technology.

DNV Energy Systems
Energy and renewable energy certification.
Sponsored listings
Frequently Asked Questions
ASTM C1287-18 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Impurities in Nuclear Grade Uranium Compounds by Inductively Coupled Plasma Mass Spectrometry". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is capable of measuring the elements listed in Table 1, some of which are required by Specifications C753, C776, C787, C788, C967 and C996. SCOPE 1.1 This test method covers the determination of 67 elements in uranium dioxide samples and nuclear grade uranium compounds and solutions without matrix separation by inductively coupled plasma mass spectrometry (ICP-MS). The elements are listed in Table 1. These elements can also be determined in uranyl nitrate hexahydrate (UNH), uranium hexafluoride (UF6), triuranium octoxide (U3O8) and uranium trioxide (UO3) if these compounds are treated and converted to the same uranium concentration solution. 1.2 The elements boron, sodium, silicon, phosphorus, potassium, calcium and iron can be determined using different techniques. The analyst's instrumentation will determine which procedure is chosen for the analysis. 1.3 The test method for technetium-99 is given in Annex A1. 1.4 The values stated in SI units are to be regarded as standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Warning—The ICP-MS is a source of intense ultra-violet radiation from the radio frequency induced plasma. Protection from radio frequency radiation and UV radiation is provided by the instrument under normal operation. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is capable of measuring the elements listed in Table 1, some of which are required by Specifications C753, C776, C787, C788, C967 and C996. SCOPE 1.1 This test method covers the determination of 67 elements in uranium dioxide samples and nuclear grade uranium compounds and solutions without matrix separation by inductively coupled plasma mass spectrometry (ICP-MS). The elements are listed in Table 1. These elements can also be determined in uranyl nitrate hexahydrate (UNH), uranium hexafluoride (UF6), triuranium octoxide (U3O8) and uranium trioxide (UO3) if these compounds are treated and converted to the same uranium concentration solution. 1.2 The elements boron, sodium, silicon, phosphorus, potassium, calcium and iron can be determined using different techniques. The analyst's instrumentation will determine which procedure is chosen for the analysis. 1.3 The test method for technetium-99 is given in Annex A1. 1.4 The values stated in SI units are to be regarded as standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Warning—The ICP-MS is a source of intense ultra-violet radiation from the radio frequency induced plasma. Protection from radio frequency radiation and UV radiation is provided by the instrument under normal operation. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM C1287-18 is classified under the following ICS (International Classification for Standards) categories: 27.120.30 - Fissile materials and nuclear fuel technology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C1287-18 has the following relationships with other standards: It is inter standard links to ASTM C1287-10, ASTM C1219-05(2009), ASTM C859-24, ASTM C787-20, ASTM C996-20, ASTM C967-20, ASTM C1346-19, ASTM C753-16, ASTM C996-15, ASTM C787-15, ASTM C859-14a, ASTM C859-14, ASTM C1346-08(2014), ASTM C859-13a, ASTM C859-13. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C1287-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: C1287 − 18
Standard Test Method for
Determination of Impurities in Nuclear Grade Uranium
Compounds by Inductively Coupled Plasma Mass
Spectrometry
This standard is issued under the fixed designation C1287; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This test method covers the determination of 67 ele-
2.1 ASTM Standards:
ments in uranium dioxide samples and nuclear grade uranium
C753Specification for Nuclear-Grade, Sinterable Uranium
compounds and solutions without matrix separation by induc-
Dioxide Powder
tively coupled plasma mass spectrometry (ICP-MS). The
C776SpecificationforSinteredUraniumDioxidePelletsfor
elements are listed in Table1. These elements can also be
Light Water Reactors
determined in uranyl nitrate hexahydrate (UNH), uranium
C787Specification for Uranium Hexafluoride for Enrich-
hexafluoride (UF ), triuranium octoxide (U O ) and uranium
6 3 8
ment
trioxide(UO )ifthesecompoundsaretreatedandconvertedto
C788Specification for Nuclear-Grade Uranyl Nitrate Solu-
the same uranium concentration solution.
tion or Crystals
1.2 The elements boron, sodium, silicon, phosphorus,
C859Terminology Relating to Nuclear Materials
potassium, calcium and iron can be determined using different
C967Specification for Uranium Ore Concentrate
techniques. The analyst’s instrumentation will determine
C996Specification for Uranium Hexafluoride Enriched to
which procedure is chosen for the analysis.
Less Than 5% U
C1346Practice for Dissolution of UF from P-10 Tubes
1.3 The test method for technetium-99 is given in Annex
A1. C1347Practice for Preparation and Dissolution of Uranium
Materials for Analysis
1.4 The values stated in SI units are to be regarded as
D1193Specification for Reagent Water
standard.
1.5 This standard does not purport to address all of the
3. Terminology
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
3.1 Definitions:
priate safety, health, and environmental practices and deter-
3.1.1 For definitions of terms relating to the nuclear fuel
mine the applicability of regulatory limitations prior to use.
cycle, refer to Terminology C859.
Warning—The ICP-MS is a source of intense ultra-violet
radiation from the radio frequency induced plasma. Protection
4. Summary of Test Method
fromradiofrequencyradiationandUVradiationisprovidedby
4.1 The sample is dissolved in acid if it is not already a
the instrument under normal operation.
solution. A fixed quantity of internal standard is added to
1.6 This international standard was developed in accor-
dance with internationally recognized principles on standard- monitor and correct for signal instability. The level of impuri-
ization established in the Decision on Principles for the ties in the solution is measured by ICP-MS. Customized
Development of International Standards, Guides and Recom- software calculates the concentration of each element.
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
ThistestmethodisunderthejurisdictionofASTMCommitteeC26onNuclear
Fuel Cycle and is the direct responsibility of Subcommittee C26.05 on Methods of
Test. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Jan. 1, 2018. Published January 2018. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1994. Last previous edition approved in 2010 as C1287–10. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/C1287-18. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1287 − 18
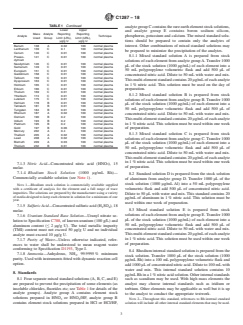
TABLE 1 Reporting Limits of Impurity Elements
4.2 Uranium-concentration-matched standard solutions are
used to calibrate the ICP-MS instrument. The calibration is
NOTE 1—The impurity elements were determined in 0.2% uranium
3,4
linearuptoatleast0.2µg/mL(100µg/gU)foreachanalyte.
solutions, prepared following Section 9.
4.3 Microwave dissolution may be used as an alternate
NOTE 2—Acquisition time=10 s/isotope using peak jump mode.
dissolution method.
NOTE 3—103 Rh was used as an internal standard. For the elements
where the technique is identified as Perkin Elmer DRCII scandium was
5. Significance and Use
usedasinternalstandardforboron,sodiumandphosphorus.Rhodiumwas
used as the internal standard for potassium, calcium and iron in Reaction
5.1 This test method is capable of measuring the elements
Cell mode.
listed in Table 1, some of which are required by Specifications
C753, C776, C787, C788, C967 and C996.
NOTE4—TheLRLisbasedonthewithinrunstandarddeviation(S)of
b
20 uranium-matched blank determinations for each analyte. This limit
6. Apparatus equals 4× S , rounded up to a preferred value in the series 1, 1.5, 2, 3, 4,
b
6, multiplied or divided by the appropriate integer power of ten.
6.1 ICP-MS, controlled by computer and fitted with the
NOTE 5—The upper reporting limit can be increased by extending the
associated software and peripherals. May be fitted with cold
calibrationto10µg/mL(5000µg/gU)iftheICP-MSusedhasanextended
plasma option. Current instrumentation is available with dy-
dynamic range (EDR) accessory.
namic reaction cell or collision cell options.
NOTE 6—For the elements where the technique is listed as P-E DRCII,
6.2 Autosampler, with tube racks and disposable plastic
the instrumentation may be specific to those elements.Alternatively cold
sample tubes compatible with 6.1 (optional).
plasma technique may be used and it is up to the analyst to perform
testwork using spikes and reference materials and to determine the lower
6.3 Variable Micropipettes:
reporting levels. The impurity elements were determined in 0.16%
6.3.1 10 µL to 100 µL capacity.
uranium solutions, prepared following Section 9. The dwell times are
6.3.2 100 µL to 1000 µL capacity.
listed in 9.4.1.1.
6.3.3 1000 µL to 10.00 mL capacity.
NOTE 7—Some of the elements are not included in the material
specifications and have been included only as a research record for the
6.4 Volumetric Flasks:
reader’s interest.
6.4.1 50 mL capacity—polypropylene.
Lower Upper
6.4.2 100 mL capacity—polypropylene.
Mass Analyte Reporting Reporting
Analyte Technique
6.4.3 1 L capacity—glass.
Used Group Limit (LRL), Limit (URL),
µg/g U µg/g U
6.5 Platinum Dish—100 mL capacity.
Lithium 7 A 0.01 100 normal plasma
6.6 Silica Beaker—250 mL capacity. Beryllium 9 A 0.04 100 normal plasma
Boron 11 E 0.3 100 DRCII
6.7 Watch Glasses—75 mm diameter.
Sodium 23 E 0.3 100 DRCII
Magnesium 24 A 4 100 normal plasma
6.8 Polypropylene Tubes—50 mL, with graduation marks
Aluminum 27 D 2 1000 normal plasma
and with caps. Phosphorus 31 E 1 100 DRCII
Potassium 39 E 2.0 100 DRCII
Calcium 40 E 3 100 DRCII
7. Reagents
Scandium 45 A 4 100 normal plasma
Titanium 48 B 0.2 100 normal plasma
7.1 ThesensitivityoftheICP-MStechniquerequirestheuse
Vanadium 51 B 0.04 100 normal plasma
of ultra high purity reagents in order to be able to obtain the
Chromium 52 B 0.1 100 normal plasma
low levels of detection. All the reagents below are ultra high
Manganese 55 A 0.1 100 normal plasma
Iron 56 A 0.2 100 DRCII
purity grade unless otherwise stated:
Cobalt 59 A 0.02 100 normal plasma
7.1.1 Element stock standards at 1000 µg/mL for all the
Nickel 60 A 0.4 100 normal plasma
elements in Table 1.
Copper 65 A 0.2 100 normal plasma
Zinc 66 A 0.3 100 normal plasma
7.1.2 Hydrofluoric Acid (HF), (40 g/100 g), 23 molar.
Gallium 69 A 0.04 100 normal plasma
7.1.2.1 Warning—Hydrofluoric acid is a highly corrosive
Germanium 74 A 0.2 100 normal plasma
acidthatcanseverelyburnskin,eyes,andmucousmembranes.
Arsenic 75 A 0.2 100 normal plasma
Selenium 82 A 3 100 normal plasma
Hydrofluoric acid differs from other acids because the fluoride
Rubidium 85 A 0.06 100 normal plasma
ion readily penetrates the skin, causing destruction of deep
Strontium 88 A 0.06 100 normal plasma
tissue layers. Unlike other acids that are rapidly neutralized,
Yttrium 89 A 0.04 100 normal plasma
Zirconium 90 B 0.02 100 normal plasma
hydrofluoricacidreactionswithtissuemaycontinuefordaysif
Niobium 93 B 0.01 100 normal plasma
left untreated. Familiarization and compliance with the Safety
Molybde- 95 B 0.04 100 normal plasma
Data Sheet is essential.
num
Ruthenium 102 B 0.02 100 normal plasma
Palladium 106 B 0.2 100 normal plasma
Silver 107 A 0.1 100 normal plasma
“ICP-MSVersus Conventional Methods for theAnalysis ofTrace Impurities in
Cadmium 111 A 0.03 100 normal plasma
Nuclear Fuel,” by Allenby, P., Clarkson, A. S., Makinson, P. R., presented at 2nd
Indium 115 A 0.04 100 normal plasma
Surrey Conference on Plasma Source Mass Spectrometry, Guildford, UK, July
Tin 116 B 0.04 100 normal plasma
1987.
Antimony 121 B 0.02 100 normal plasma
“Trace Metals in NBL Uranium Standard CRM 124 Using ICP-MS,” by
Tellurium 130 B 0.4 100 normal plasma
Aldridge, A. J., Clarkson, A. S., Makinson, P. R., Dawson, K. W., presented at 1st
Caesium 133 A 0.06 100 normal plasma
Durham International Conference on Plasma Source Mass Spectrometry, Durham,
UK, September 1988.
C1287 − 18
TABLE 1 Continued
analytegroupCcontainstherareearthelementstocksolutions,
Lower Upper
and analyte group E contains boron sodium silicon,
Mass Analyte Reporting Reporting
Analyte Technique
phosphorus, potassium and calcium. The mixed standard solu-
Used Group Limit (LRL), Limit (URL),
µg/g U µg/g U tions should be prepared to contain only the analytes of
Barium 138 A 0.02 100 normal plasma interest. Other combinations of mixed standard solutions may
Lanthanum 139 C 0.1 100 normal plasma
be prepared to minimize the precipitation of the analytes.
Cerium 140 C 0.01 100 normal plasma
8.1.1 Mixed standard solution A is prepared from stock
Praseo- 141 C 0.01 100 normal plasma
dymium
solutions of each element from analyte groupA.Transfer 1000
Neodymium 146 C 0.01 100 normal plasma
µL of the stock solution (1000 µg/mL) of each element into a
Samarium 149 C 0.01 100 normal plasma
50 mL polypropylene volumetric flask and add 500 µL of
Europium 151 C 0.01 100 normal plasma
Gadolinium 158 C 0.01 100 normal plasma
concentrated nitric acid. Dilute to 50 mL with water and mix.
Terbium 159 C 0.01 100 normal plasma
Thismulti-elementstandardcontains20µg/mLofeachanalyte
Dysprosium 163 C 0.01 100 normal plasma
Holmium 165 C 0.01 100 normal plasma in 1% nitric acid. This solution must be used on the day of
Erbium 166 C 0.01 100 normal plasma
preparation.
Thulium 169 C 0.01 100 normal plasma
8.1.2 Mixed standard solution B is prepared from stock
Ytterbium 174 C 0.01 100 normal plasma
Lutetium 175 C 0.01 100 normal plasma
solutions of each element from analyte group B.Transfer 1000
Hafnium 178 B 0.01 100 normal plasma
µL of the stock solution (1000 µg/mL) of each element into a
Tantalum 181 B 0.01 100 normal plasma
50 mL polypropylene volumetric flask and add 500 µL of
Tungsten 184 B 0.01 100 normal plasma
Rhenium 187 A 0.02 100 normal plasma
concentrated nitric acid. Dilute to 50 mL with water and mix.
Osmium 190 B 0.2 100 normal plasma
Thismulti-elementstandardcontains20µg/mLofeachanalyte
Iridium 193 B 0.2 100 normal plasma
in 1% nitric acid.This solution must be used within one week
Platinum 195 B 0.2 100 normal plasma
Gold 197 B 0.06 100 normal plasma
of preparation.
Mercury 202 A 0.4 100 normal plasma
8.1.3 Mixed standard solution C is prepared from stock
Thallium 205 A 0.02 100 normal plasma
Lead 208 A 0.02 100 normal plasma solutions of each element from analyte group C.Transfer 1000
Bismuth 209 A 0.03 100 normal plasma
µL of the stock solution (1000 µg/mL) of each element into a
Thorium 232 B 0.01 100 normal plasma
50 mL polypropylene volumetric flask and add 500 µL of
concentrated nitric acid. Dilute to 50 mL with water and mix.
Thismulti-elementstandardcontains20µg/mLofeachanalyte
in 1% nitric acid.This solution must be used within one week
7.1.3 Nitric Acid—Concentrated nitric acid (HNO ), 15
of preparation.
molar.
7.1.4 Rhodium Stock Solution (1000 µg/mL Rh)—
8.2 Standard solution D is prepared from the stock solution
Commercially available solution (see Note 1).
of aluminum from analyte group D. Transfer 1000 µL of the
stock solution (1000 µg/mL Al) into a 50 mL polypropylene
NOTE 1—Rhodium stock solution is commercially available supplied
with a certificate of analysis for the element and a full range of trace volumetric flask and add 500 µL of concentrated nitric acid.
impurities.Thesolutionsarepreparedbythemanufacturerusingavariety
Dilute to 50 µLwith water and mix. This standard contains 20
ofmediadesignedtokeepeachelementinsolutionforaminimumofone
µg/mL of aluminum in 1% nitric acid. This solution must be
year.
used within one week of preparation.
7.1.5 SulfuricAcid—Concentratedsulfuricacid(H SO ),18
2 4
8.3 Mixed standard solution E is prepared from stock
molar.
solutions of each element from analyte group E.Transfer 1000
7.1.6 Uranium Standard Base Solution—Uranyl nitrate so-
µL of the stock solution (1000 µg/mL) of each element into a
lution to Specification C788, of known uranium (100 g/L) and
50 mL polypropylene volumetric flask and add 500 µL of
aluminum content (≤ 2 µg/g U). The total metallic impurity
concentrated nitric acid. Dilute to 50 mL with water and mix.
(TMI) content must not exceed 50 µg/g U and no individual
Thismulti-elementstandardcontains20µg/mLofeachanalyte
analyte must exceed 10 µg/g U.
in 1% nitric acid.This solution must be used within one week
7.1.7 Purity of Water—Unless otherwise indicated, refer-
of preparation.
ences to water shall be understood to mean reagent water
conforming to Specification D1193, Type I.
8.4 Rhodiuminternalstandardsolutionispreparedfromthe
7.1.8 Ammonia—Anhydrous, NH , 99.9995% minimum
stock solution. Transfer 1000 µL of the stock solution (1000
purity. Used with instruments fitted with dynamic reaction cell
µg/mL Rh) into a 100 mL polypropylene volumetric flask and
option.
add1000µLofconcentratednitricacid.Diluteto100mLwith
water and mix. This internal standard solution contains 10
8. Standards
µg/mLRhina1%nitricacidsolution.Otherinternalstandards
8.1 Fourseparatemixedstandardsolutions(A,B,C,andE)
such as scandium may be used. With high mass elements the
are prepared to prevent the precipitation of some elements (as
analyst may choose internal standards such as iridium or
insoluble chlorides, fluorides etc; see Table 1 for details of the
terbium. Other elements may be applicable as well but it is up
analyte groups). Analyte group A contains element stock
to the analyst to conduct the appropriate testwork.
solutions prepared in HNO or HNO /HF, analyte group B
3 3 NOTE 2—Throughout this standard, references to Rh internal standard
contains element stock solutions prepared in HCl or HCl/HF, solutionwillincludeallotherinternalstandardelementsthatmaybeused.
C1287 − 18
9. Procedure 9.2.6 Analyze these solutions as in 9.4 using the calibration
solutions prepared in 9.3. The solutions must be analyzed
NOTE 3—Auranium-free reagent blank is used to eliminate bias due to
the analyte concentrations in the uranium standard base solution. A
within8hof preparation to minimize the effects of analyte
uranium-matched reagent blank is necessary to provide a constant acid
precipitation.
concentration in the nebulized solution.
9.3 Preparation of Blanks and Calibration Standard Solu-
9.1 Sample Preparation for the Determination of All Ele-
tions:
ments Except Boron:
9.3.1 For the Determination of All Elements Except Boron:
9.1.1 Weigh a portion of uranium oxide containing between
9.3.1.1 Uranium-free Reagent Blank—Transfer 12.5 mL of
2.45 and 2.55 g of uranium into a platinum dish. Record the
concentrated nitric acid and 2.5 mL of hydrofluoric acid (40
weight to the nearest 0.001 g. For uranyl fluoride solutions
g/100 g) into a 50 mL polypropylene volumetric flask. Con-
prepared using Practice C1346 and uranyl nitrate solutions,
tinue as instructed from 9.1.5 onwards.
aliquot between 2.45 and 2.55 g of uranium into a platinum
9.3.1.2 Uranium-matched Calibration Blank—Transfer2.00
dish. Use a variable volume plastic pipet for the transfer of
mL of the uranium standard base solution (see 7.1.6; this is
uranyl fluoride solutions. Record the weight to the nearest
equivalent to 0.20 g of uranium) into a 100 mLpolypropylene
0.001 g.
volumetric flask.Add 1000 µLof concentrated nitric acid, 200
µL of hydrofluoric acid (40 g/100 g) and 1000 µL of rhodium
9.1.2 Add 10 mL of water and 12.5 mL of concentrated
internal standard solution (see 8.4). Dilute to 100 mL with
nitric acid. Heat on a hotplate to assist dissolution.
water and mix. This solution contains2gof uranium per litre
9.1.3 Add 2.5 mL of hydrofluoric acid (40 g/100 g) and
and 0.1 µg/mL Rh in 1% nitric acid/0.2% hydrofluoric acid.
warm at about 80°C for 5 min.
9.3.1.3 Uranium-matched Calibration Standard—Transfer
9.1.4 Allow the solution to cool and transfer quantitatively
2.00 mL of the uranium standard base solution (see 7.1.6; this
to a 50 mL polypropylene volumetric flask. Dilute to 50 mL
is equivalent to 0.20 g of uranium) into a 100 mLpolypropyl-
withwaterandmix.Thissolutioncontains50gofuraniumper
ene volumetric flask.Add 1000 µLof concentrated nitric acid,
litre in 25% nitric acid/5% hydrofluoric acid.
200 µL of hydrofluoric acid (40 g/100 g), 1000 µL of each
9.1.5 Transfer 4.00 mLof the solution in 9.1.4 and 1.00 mL
mixed standard solution (see 8.1.1, 8.1.2 and 8.1.3) and 1000
of the rhodium internal standard solution (see 8.4) into a 100
µL of rhodium internal standard solution (see 8.4). Dilute to
mL polypropylene volumetric flask. Dilute to 100 mL with
100 mL with water and mix. This solution contains2gof
water and mix. This solution contains2gof uranium per litre
uraniumperlitre,0.2µg/mLofeachanalyte(equivalentto100
and 0.1 µg/mL Rh in 1% nitric acid/0.2% hydrofluoric acid.
µg/g U) and 0.1 µg/mL Rh in 1% nitric acid/0.2% hydroflu-
9.1.6 Auranium-freereagentblank(see9.3.1)andacontrol
oric acid.
or recovery sample must be prepared with every run of
9.3.2 For the Determination of Boron:
samples.
9.3.2.1 Uranium-matched Reagent/Calibration Blank—
Transfer 2.00 mL of the uranium standard base solution (see
9.1.7 Analyze these solutions as in 9.4 using the calibration
solutions prepared in 9.3. The solutions must be analyzed 7.1.6; this is equivalent to 0.20 g of uranium) into a 100 mL
polypropylene volumetric flask. Add 2.5 mL of concentrated
within8hof preparation to minimize the effects of analyte
precipitation. nitric acid, 200 µLof hydrofluoric acid (40 g/100 g), and 1000
µL of scandium internal standard solution (see 8.4). Dilute to
9.2 Sample Preparation for the Determination of Boron:
100 mL with water and mix. This solution contains2gof
9.2.1 Weighaportionofuraniumoxide,containingbetween
uranium per litre and 0.1 µg/mL Sc in 2.5% nitric acid/0.2%
0.095 and 0.105 g of uranium into a graduated 50 mL
hydrofluoric acid.
polypropylene tube (or alternative). The accuracy of the
9.3.2.2 Uranium-matched Calibration Standard—Transfer
graduations on the tube must be verified. Record the weight to
2.00 mL of the uranium standard base solution (see 7.1.7; this
the nearest 0.001 g. For uranyl fluoride solutions prepared
is equivalent to 0.20 g of uranium) into a 100 mLpolypropyl-
using Practice C1346 and uranyl nitrate solutions, aliquot
ene volumetric flask. Add 2.5 mL of concentrated nitric acid,
between 0.095 and 0.105 g of uranium using variable volume
200 µL of hydrofluoric acid (40 g/100 g), 1000 µL of mixed
plastic pipets. Record the weight to the nearest 0.001 g.
standard solution (see 8.1 and 8.3), and 1000 µL of scandium
9.2.2 Add1mLofwaterand1.25mLofconcentratednitric
internal standard solution (see 8.4). Dilute to 100 mL with
acid. Cap. Heat in a hot water bath at about 80°C to assist
water and mix. This solution contains2gof uranium per litre,
dissolution. Heat until all the sample is dissolved.
0.2 µg/mL of each analyte (equivalent to 100 µg/g U) and 0.1
9.2.3 Cooltoroomtemperature.Add0.1mLofhydrofluoric µg/mL Sc in 2.5% nitric acid/0.2% hydrofluoric acid.
acid (40 g/100 g) and cap. Heat in a hot water bath at about
9.4 Measurement of Elements by ICP-MS:
80°C for 5 min.
9.4.1 Toavoidcontaminationproblemswhennebulizingthe
9.2.4 Allow the solution to cool. Add 0.5 mL of scandium
samples,whichcontainhydrofluoricacid,thenebulizersystem
internalstandardsolution(see8.4).Diluteto50mLwithwater
(that is, spray chamber and nebulizer) must be made from
andmix.Thissolutioncontains2gofuraniumperlitreand0.1
fluorinatedplasticmaterials(forexample,TFE-fluorocarbonor
µg/mL Sc in 2.5% nitric acid/0.2% hydrofluoric acid.
polychlorotrifluoroethylene).
9.2.5 A uranium-free reagent blank and a control or recov- 9.4.1.1 Set up the ICP-MS for the analysis using the
ery sample must be prepared with every run of samples. parameters given in the manufacturer’s operating manual.
C1287 − 18
TABLE 2 Precision Data Derived from PCS and CRM Samples
Nebulize the uranium-matched reagent/calibration blank solu-
tion to optimize conditions using the 103 Rh internal standard.
NOTE 1—Acquisition time=10 s/isotope using peak jump mode.
Acquisition time is 2.5 s / isotope for B, Na, P, K, Ca, Fe.
Example Instrument Operating Conditions
Solution Pumping Rate Sample solution IN: 1.25 mL/min
NOTE 2—Table 2 is a list of “between-run” standard deviations for a
ICP Incident Power 1400 watts
single determination based on the analysis of in-house primary control
ICP Reflected Power <10 watts
samples (PCS series), NBLCertified Reference Material CRM 124-2 and
Plasma Argon Coolant 14 L/min at 70 psig
CRM 98-2.
Plasma Argon Auxiliar
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: C1287 − 10 C1287 − 18
Standard Test Method for
Determination of Impurities in Nuclear Grade Uranium
Compounds by Inductively Coupled Plasma Mass
Spectrometry
This standard is issued under the fixed designation C1287; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the determination of 67 elements in uranium dioxide samples and nuclear grade uranium
compounds and solutions without matrix separation by inductively coupled plasma mass spectrometry (ICP-MS). The elements are
listed in Table 1. These elements can also be determined in uranyl nitrate hexahydrate (UNH), uranium hexafluoride (UF ),
triuranium octoxide (U O ) and uranium trioxide (UO ) if these compounds are treated and converted to the same uranium
3 8 3
concentration solution.
1.2 The elements boron, sodium, silicon, phosphorus, potassium, calcium and iron can be determined using different techniques.
The analyst’s instrumentation will determine which procedure is chosen for the analysis.
1.3 The test method for technetium-99 is given in Annex A1.
1.4 The values stated in SI units are to be regarded as standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. Warning—The ICP-MS is a source of intense ultra-violet radiation from the
radio frequency induced plasma. Protection from radio frequency radiation and UV radiation is provided by the instrument under
normal operation.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
C753 Specification for Nuclear-Grade, Sinterable Uranium Dioxide Powder
C776 Specification for Sintered Uranium Dioxide Pellets for Light Water Reactors
C787 Specification for Uranium Hexafluoride for Enrichment
C788 Specification for Nuclear-Grade Uranyl Nitrate Solution or Crystals
C859 Terminology Relating to Nuclear Materials
C967 Specification for Uranium Ore Concentrate
C996 Specification for Uranium Hexafluoride Enriched to Less Than 5 % U
C1346 Practice for Dissolution of UF from P-10 Tubes
C1347 Practice for Preparation and Dissolution of Uranium Materials for Analysis
D1193 Specification for Reagent Water
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms relating to the nuclear fuel cycle, refer to Terminology C859.
This test method is under the jurisdiction of ASTM Committee C26 on Nuclear Fuel Cycle and is the direct responsibility of Subcommittee C26.05 on Methods of Test.
Current edition approved June 1, 2010Jan. 1, 2018. Published July 2010January 2018. Originally approved in 1994. Last previous edition approved in 20032010 as
C1287 – 03.C1287 – 10. DOI: 10.1520/C1287-10.10.1520/C1287-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1287 − 18
4. Summary of Test Method
4.1 The sample is dissolved in acid if it is not already a solution. A fixed quantity of internal standard is added to monitor and
correct for signal instability. The level of impurities in the solution is measured by ICP-MS. Customized software calculates the
concentration of each element.
4.2 Uranium-concentration-matched standard solutions are used to calibrate the ICP-MS instrument. The calibration is linear up
3,4
to at least 0.2 μg/mlμg/mL (100 μg/g U) for each analyte.
4.3 Microwave dissolution may be used as an alternate dissolution method.
5. Significance and Use
5.1 This test method is capable of measuring the elements listed in Table 1, some of which are required by Specifications C753,
C776, C787, C788, C967 and C996.
6. Apparatus
6.1 ICP-MS, controlled by computer and fitted with the associated software and peripherals. May be fitted with cold plasma
option. Current instrumentation is available with dynamic reaction cell or collision cell options.
6.2 Autosampler, with tube racks and disposable plastic sample tubes compatible with 5.16.1 (optional).
6.3 Variable Micropipettes:
6.3.1 10 μL to 100 μL capacity.
6.3.2 100 μL to 1000 μL capacity.
6.3.3 1000 μL to 10.00 mL capacity.
6.4 Volumetric Flasks:
6.4.1 50 mL capacity—polypropylene.
6.4.2 100 mL capacity—polypropylene.
6.4.3 1 L capacity—glass.
6.5 Platinum Dish—100 mL capacity.
6.6 Silica Beaker—250 mL capacity.
6.7 Watch Glasses—75 mm diameter.
6.8 Polypropylene Tubes—50 mL, with graduation marks and with caps.
7. Reagents
7.1 The sensitivity of the ICP-MS technique requires the use of ultra high purity reagents in order to be able to obtain the low
levels of detection. All the reagents below are ultra high purity grade unless otherwise stated:
7.1.1 Element stock standards at 1000 μg/mL for all the elements in Table 1.
7.1.2 Hydrofluoric Acid (HF), (40 g/100 g), 23 molar.
7.1.2.1 Warning—Hydrofluoric acid is a highly corrosive acid that can severely burn skin, eyes, and mucous membranes.
Hydrofluoric acid is similar to other acids in that the initial extent of a burn depends on the concentration, the temperature, and
the duration of contact with the acid. Hydrofluoric acid differs from other acids because the fluoride ion readily penetrates the skin,
causing destruction of deep tissue layers. Unlike other acids that are rapidly neutralized, hydrofluoric acid reactions with tissue may
continue for days if left untreated. Due to the serious consequences of hydrofluoric acid burns, prevention of exposure or injury
of personnel is the primary goal. Utilization of appropriate laboratory controls (hoods) and wearing adequate personnel protective
equipment to protect from skin and eye contact is essential. Acute exposure to HF can cause painful and severe burns upon skin
contact that require special medical attention. Chronic or prolonged exposure to low levels on the skin may cause
fluorosis.Familiarization and compliance with the Safety Data Sheet is essential.
7.1.3 Nitric Acid—Concentrated nitric acid (HNO ), 15 molar.
7.1.4 Rhodium Stock Solution (1000 μg/mL Rh)—Commercially available solution (see Note 1).
NOTE 1—Rhodium stock solution is commercially available supplied with a certificate of analysis for the element and a full range of trace impurities.
The solutions are prepared by the manufacturer using a variety of media designed to keep each element in solution for a minimum of one year.
7.1.5 Sulfuric Acid—Concentrated sulfuric acid (H SO ), 18 molar.
2 4
“ICP-MS Versus Conventional Methods for the Analysis of Trace Impurities in Nuclear Fuel,” by Allenby, P., Clarkson, A. S., Makinson, P. R., presented at 2nd Surrey
Conference on Plasma Source Mass Spectrometry, Guildford, UK, July 1987.
“Trace Metals in NBL Uranium Standard CRM 124 Using ICP-MS,” by Aldridge, A. J., Clarkson, A. S., Makinson, P. R., Dawson, K. W., presented at 1st Durham
International Conference on Plasma Source Mass Spectrometry, Durham, UK, September 1988.
C1287 − 18
TABLE 1 Reporting Limits of Impurity Elements
NOTE 1—The impurity elements were determined in 0.2 % uranium
solutions, prepared following Section 89.
NOTE 2—Acquisition time = 10 s/isotope using peak jump mode.
NOTE 3—103 Rh was used as an internal standard. For the elements
where the technique is identified as Perkin Elmer DRCII scandium was
used as internal standard for boron, sodium and phosphorus. Rhodium was
used as the internal standard for potassium, calcium and iron in Reaction
Cell mode.
NOTE 4—The LRL is based on the within run standard deviation (S ) of
b
20 uranium-matched blank determinations for each analyte. This limit
equals 4 × S , rounded up to a preferred value in the series 1, 1.5, 2, 3, 4,
b
6, multiplied or divided by the appropriate integer power of ten.
NOTE 5—The upper reporting limit can be increased by extending the
calibration to 10 μg/mL (5000 μg/g U) if the ICP-MS used has an extended
dynamic range (EDR) accessory.
NOTE 6—For the elements where the technique is listed as P-E DRCII,
the instrumentation may be specific to those elements. Alternatively cold
plasma technique may be used and it is up to the analyst to perform
testwork using spikes and reference materials and to determine the lower
reporting levels. The impurity elements were determined in 0.16 %
uranium solutions, prepared following Section 89. The dwell times are
listed in 8.4.1.19.4.1.1.
NOTE 7—Some of the elements are not included in the material
specifications and have been included only as a research record for the
reader’s interest.
Lower Upper
Mass Analyte Reporting Reporting
Analyte Technique
Used Group Limit (LRL), Limit (URL),
μg/g U μg/g U
Lithium 7 A 0.01 100 normal plasma
Beryllium 9 A 0.04 100 normal plasma
Boron 11 E 0.3 100 DRCII
Sodium 23 E 0.3 100 DRCII
Magnesium 24 A 4 100 normal plasma
Aluminum 27 D 2 1000 normal plasma
Phosphorus 31 E 1 100 DRCII
Potassium 39 E 2.0 100 DRCII
Calcium 40 E 3 100 DRCII
Scandium 45 A 4 100 normal plasma
Titanium 48 B 0.2 100 normal plasma
Vanadium 51 B 0.04 100 normal plasma
Chromium 52 B 0.1 100 normal plasma
Manganese 55 A 0.1 100 normal plasma
Iron 56 A 0.2 100 DRCII
Cobalt 59 A 0.02 100 normal plasma
Nickel 60 A 0.4 100 normal plasma
Copper 65 A 0.2 100 normal plasma
Zinc 66 A 0.3 100 normal plasma
Gallium 69 A 0.04 100 normal plasma
Germanium 74 A 0.2 100 normal plasma
Arsenic 75 A 0.2 100 normal plasma
Selenium 82 A 3 100 normal plasma
Rubidium 85 A 0.06 100 normal plasma
Strontium 88 A 0.06 100 normal plasma
Yttrium 89 A 0.04 100 normal plasma
Zirconium 90 B 0.02 100 normal plasma
Niobium 93 B 0.01 100 normal plasma
Molybde- 95 B 0.04 100 normal plasma
num
Ruthenium 102 B 0.02 100 normal plasma
Palladium 106 B 0.2 100 normal plasma
Silver 107 A 0.1 100 normal plasma
Cadmium 111 A 0.03 100 normal plasma
Indium 115 A 0.04 100 normal plasma
Tin 116 B 0.04 100 normal plasma
Antimony 121 B 0.02 100 normal plasma
Tellurium 130 B 0.4 100 normal plasma
Caesium 133 A 0.06 100 normal plasma
C1287 − 18
TABLE 1 Continued
Lower Upper
Mass Analyte Reporting Reporting
Analyte Technique
Used Group Limit (LRL), Limit (URL),
μg/g U μg/g U
Barium 138 A 0.02 100 normal plasma
Lanthanum 139 C 0.1 100 normal plasma
Cerium 140 C 0.01 100 normal plasma
Praseo- 141 C 0.01 100 normal plasma
dymium
Neodymium 146 C 0.01 100 normal plasma
Samarium 149 C 0.01 100 normal plasma
Europium 151 C 0.01 100 normal plasma
Gadolinium 158 C 0.01 100 normal plasma
Terbium 159 C 0.01 100 normal plasma
Dysprosium 163 C 0.01 100 normal plasma
Holmium 165 C 0.01 100 normal plasma
Erbium 166 C 0.01 100 normal plasma
Thulium 169 C 0.01 100 normal plasma
Ytterbium 174 C 0.01 100 normal plasma
Lutetium 175 C 0.01 100 normal plasma
Hafnium 178 B 0.01 100 normal plasma
Tantalum 181 B 0.01 100 normal plasma
Tungsten 184 B 0.01 100 normal plasma
Rhenium 187 A 0.02 100 normal plasma
Osmium 190 B 0.2 100 normal plasma
Iridium 193 B 0.2 100 normal plasma
Platinum 195 B 0.2 100 normal plasma
Gold 197 B 0.06 100 normal plasma
Mercury 202 A 0.4 100 normal plasma
Thallium 205 A 0.02 100 normal plasma
Lead 208 A 0.02 100 normal plasma
Bismuth 209 A 0.03 100 normal plasma
Thorium 232 B 0.01 100 normal plasma
7.1.6 Uranium Standard Base Solution—Uranyl nitrate solution to Specification C788, of known uranium (100 g/L) and
aluminum content (≤ 2 μg/g U). The total metallic impurity (TMI) content must not exceed 50 μg/g U and no individual analyte
must exceed 10 μg/g U.
7.1.7 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water conforming
to Specification D1193, Type I.
7.1.8 Ammonia—Anhydrous, NH , 99.9995 % 99.9995 % minimum purity. Used with instruments fitted with dynamic reaction
cell option.
8. Standards
8.1 Four separate mixed standard solutions (A, B, C, and E) are prepared to prevent the precipitation of some elements (as
insoluble chlorides, fluorides etc; see Table 1 for details of the analyte groups). Analyte group A contains element stock solutions
prepared in HNO or HNO /HF, analyte group B contains element stock solutions prepared in HCl or HCl/HF, analyte group C
3 3
contains the rare earth element stock solutions, and analyte group E contains boron sodium silicon, phosphorus, potassium and
calcium. The mixed standard solutions should be prepared to contain only the analytes of interest. Other combinations of mixed
standard solutions may be prepared to minimize the precipitation of the analytes.
8.1.1 Mixed standard solution A is prepared from stock solutions of each element from analyte group A. Transfer 1000 μL of
the stock solution (1000 μg/mL) of each element into a 50 mL polypropylene volumetric flask and add 500 μL of concentrated nitric
acid. Dilute to 50 mL with water and mix. This multi-element standard contains 20 μg/mL of each analyte in 1 % nitric acid. This
solution must be used on the day of preparation.
8.1.2 Mixed standard solution B is prepared from stock solutions of each element from analyte group B. Transfer 1000 μL of
the stock solution (1000 μg/mL) of each element into a 50 mL polypropylene volumetric flask and add 500 μL of concentrated nitric
acid. Dilute to 50 mL with water and mix. This multi-element standard contains 20 μg/mL of each analyte in 1 % nitric acid. This
solution must be used within one week of preparation.
8.1.3 Mixed standard solution C is prepared from stock solutions of each element from analyte group C. Transfer 1000 μL of
the stock solution (1000 μg/mL) of each element into a 50 mL polypropylene volumetric flask and add 500 μL of concentrated nitric
acid. Dilute to 50 mL with water and mix. This multi-element standard contains 20 μg/mL of each analyte in 1 % nitric acid. This
solution must be used within one week of preparation.
8.2 Standard solution D is prepared from the stock solution of aluminum from analyte group D. Transfer 1000 μL of the stock
solution (1000 μg/mL Al) into a 50 mL polypropylene volumetric flask and add 500 μL of concentrated nitric acid. Dilute to 50
μL with water and mix. This standard contains 20 μg/mL of aluminum in 1 % nitric acid. This solution must be used within one
week of preparation.
C1287 − 18
8.3 Mixed standard solution E is prepared from stock solutions of each element from analyte group E. Transfer 1000 μL of the
stock solution (1000 μg/mL) of each element into a 50 mL polypropylene volumetric flask and add 500 μL of concentrated nitric
acid. Dilute to 50 mL with water and mix. This multi-element standard contains 20 μg/mL of each analyte in 1 % nitric acid. This
solution must be used within one week of preparation.
8.4 Rhodium internal standard solution is prepared from the stock solution. Transfer 1000 μL of the stock solution (1000 μg/mL
Rh) into a 100 mL polypropylene volumetric flask and add 1000 μL of concentrated nitric acid. Dilute to 100 mL with water and
mix. This internal standard solution contains 10 μg/mL Rh in a 1 % nitric acid solution. Other internal standards such as scandium
may be used. With high mass elements the analyst may choose internal standards such as iridium or terbium. Other elements may
be applicable as well but it is up to the analyst to conduct the appropriate testwork.
NOTE 2—Throughout this standard, references to Rh internal standard solution will include all other internal standard elements that may be used.
9. Procedure
NOTE 3—A uranium-free reagent blank is used to eliminate bias due to the analyte concentrations in the uranium standard base solution. A
uranium-matched reagent blank is necessary to provide a constant acid concentration in the nebulized solution.
9.1 Sample Preparation for the Determination of All Elements Except Boron:
9.1.1 Weigh a portion of uranium oxide containing between 2.45 and 2.55 g of uranium into a platinum dish. Record the weight
to the nearest 0.001 g. For uranyl fluoride solutions prepared using Practice C1346 and uranyl nitrate solutions, aliquot between
2.45 and 2.55 g of uranium into a platinum dish. Use a variable volume plastic pipet for the transfer of uranyl fluoride solutions.
Record the weight to the nearest 0.001 g.
9.1.2 Add 10 mL of water and 12.5 mL of concentrated nitric acid. Heat on a hotplate to assist dissolution.
9.1.3 Add 2.5 mL of hydrofluoric acid (40 g/100 g) and warm at about 80°C for 5 min.
9.1.4 Allow the solution to cool and transfer quantitatively to a 50 mL polypropylene volumetric flask. Dilute to 50 mL with
water and mix. This solution contains 50 g of uranium per litre in 25 % nitric acid/5 % hydrofluoric acid.
9.1.5 Transfer 4.00 mL of the solution in 8.1.49.1.4 and 1.00 mL of the rhodium internal standard solution (see 7.48.4) into a
100 mL polypropylene volumetric flask. Dilute to 100 mL with water and mix. This solution contains 2 g of uranium per litre and
0.1 μg/mL Rh in 1 % nitric acid/0.2 % hydrofluoric acid.
9.1.6 A uranium-free reagent blank (see 8.3.19.3.1) and a control or recovery sample must be prepared with every run of
samples.
9.1.7 Analyze these solutions as in 8.49.4 using the calibration solutions prepared in 8.39.3. The solutions must be analyzed
within 8 h of preparation to minimize the effects of analyte precipitation.
9.2 Sample Preparation for the Determination of Boron:
9.2.1 Weigh a portion of uranium oxide, containing between 0.095 and 0.105 g of uranium into a graduated 50 mL
polypropylene tube (or alternative). The accuracy of the graduations on the tube must be verified. Record the weight to the nearest
0.001 g. For uranyl fluoride solutions prepared using Practice C1346 and uranyl nitrate solutions, aliquot between 0.095 and 0.105
g of uranium using variable volume plastic pipets. Record the weight to the nearest 0.001 g.
9.2.2 Add 1 mL of water and 1.25 mL of concentrated nitric acid. Cap. Heat in a hot water bath at about 80°C to assist
dissolution. Heat until all the sample is dissolved.
9.2.3 Cool to room temperature. Add 0.1 mL of hydrofluoric acid (40 g/100 g) and cap. Heat in a hot water bath at about 80°C
for 5 min.
9.2.4 Allow the solution to cool. Add 0.5 mL of scandium internal standard solution (see 7.48.4). Dilute to 50 mL with water
and mix. This solution contains 2 g of uranium per litre and 0.1 μg/mL Sc in 2.5 % nitric acid/0.2 % hydrofluoric acid.
9.2.5 A uranium-free reagent blank and a control or recovery sample must be prepared with every run of samples.
9.2.6 Analyze these solutions as in 8.49.4 using the calibration solutions prepared in 8.39.3. The solutions must be analyzed
within 8 h of preparation to minimize the effects of analyte precipitation.
9.3 Preparation of Blanks and Calibration Standard Solutions:
9.3.1 For the Determination of All Elements Except Boron:
9.3.1.1 Uranium-free Reagent Blank—Transfer 12.5 mL of concentrated nitric acid and 2.5 mL of hydrofluoric acid (40 g/100
g) into a 50 mL polypropylene volumetric flask. Continue as instructed from 8.1.59.1.5 onwards.
9.3.1.2 Uranium-matched Calibration Blank—Transfer 2.00 mL of the uranium standard base solution (see 6.1.67.1.6; this is
equivalent to 0.20 g of uranium) into a 100 mL polypropylene volumetric flask. Add 1000 μL of concentrated nitric acid, 200 μL
of hydrofluoric acid (40 g/100 g) and 1000 μL of rhodium internal standard solution (see 7.48.4). Dilute to 100 mL with water and
mix. This solution contains 2 g of uranium per litre and 0.1 μg/mL Rh in 1 % nitric acid/0.2 % hydrofluoric acid.
9.3.1.3 Uranium-matched Calibration Standard—Transfer 2.00 mL of the uranium standard base solution (see 6.1.67.1.6; this
is equivalent to 0.20 g of uranium) into a 100 mL polypropylene volumetric flask. Add 1000 μL of concentrated nitric acid, 200
μL of hydrofluoric acid (40 g/100 g), 1000 μL of each mixed standard solution (see 7.1.18.1.1, 7.1.28.1.2 and 7.1.38.1.3) and 1000
μL of rhodium internal standard solution (see 7.48.4). Dilute to 100 mL with water and mix. This solution contains 2 g of uranium
per litre, 0.2 μg/mL of each analyte (equivalent to 100 μg/g U) and 0.1 μg/mL Rh in 1 % nitric acid/0.2 % hydrofluoric acid.
9.3.2 For the Determination of Boron:
C1287 − 18
9.3.2.1 Uranium-matched Reagent/Calibration Blank—Transfer 2.00 mL of the uranium standard base solution (see 6.1.67.1.6;
this is equivalent to 0.20 g of uranium) into a 100 mL polypropylene volumetric flask. Add 2.5 mL of concentrated nitric acid, 200
μL of hydrofluoric acid (40 g/100 g), and 1000 μL of scandium internal standard solution (see 7.48.4). Dilute to 100 mL with water
and mix. This solution contains 2 g of uranium per litre and 0.1 μg/mL Sc in 2.5 % nitric acid/0.2 % hydrofluoric acid.
9.3.2.2 Uranium-matched Calibration Standard—Transfer 2.00 mL of the uranium standard base solution (see 6.1.77.1.7; this
is equivalent to 0.20 g of uranium) into a 100 mL polypropylene volumetric flask. Add 2.5 mL of concentrated nitric acid, 200 μL
of hydrofluoric acid (40 g/100 g), 1000 μL of mixed standard solution (see 7.18.1 and 7.38.3), and 1000 μL of scandium internal
standard solution (see 7.48.4). Dilute to 100 mL with water and mix. This solution contains 2 g of uranium per litre, 0.2 μg/mL
of each analyte (equivalent to 100 μg/g U) and 0.1 μg/mL Sc in 2.5 % nitric acid/0.2 % hydrofluoric acid.
9.4 Measurement of Elements by ICP-MS:
9.4.1 To avoid contamination problems when nebulizing the samples, which contain hydrofluoric acid, the nebulizer system
(that is, spray chamber and nebulizer) must be made from fluorinated plastic materials (for example, TFE-fluorocarbon or
polychlorotrifluoroethylene).
9.4.1.1 Set up the ICP-MS for the analysis using the parameters given in the manufacturer’s operating manual. Nebulize the
uranium-matched reagent/calibration blank solution to optimize conditions using the 103 Rh internal standard.
Example Instrument Operating Conditions
Solution Pumping Rate Sample solution IN: 1.25 mL/min
ICP Incident Power 1400 watts
ICP Reflected Power <10 watts
Plasma Argon Coolant 14 L/min at 70 psig
Plasma A
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...