ASTM F2714-08(2021)
(Test Method)Standard Test Method for Oxygen Headspace Analysis of Packages Using Fluorescent Decay
Standard Test Method for Oxygen Headspace Analysis of Packages Using Fluorescent Decay
SIGNIFICANCE AND USE
3.1 The oxygen content of a package’s headspace is an important determinant of the packaging protection afforded by barrier materials. The package under test is typically MAP (modified atmosphere packaging) packaged.
3.2 Oxygen content is a key contributor to off-flavors and spoilage of various products, such as chemicals, food and pharmaceuticals.
3.3 The method determines the oxygen in a closed package headspace. This ability has application in:
3.3.1 Package Permeability Studies—The change of headspace composition over a known length of time allows the calculation of permeation. Since the headspace oxygen is measured as a percentage, the volume of the container’s headspace must be known to allow conversion into a quantity such as millilitres (ml) of oxygen. The use of this approach to measure permeation generally applies to empty package systems only as oxygen uptake or outgassing of contained products could affect results.
3.3.2 Leak Detection—If the headspace contains more oxygen than expected or is increasing faster than expected, a leak can be suspected. A wide variety of techniques can be employed to verify that a leak is present and to identify its location. If necessary or of interest, a leak rate may be calculated with known headspace volume and measured oxygen concentration change over time.
3.3.3 Efficacy of the MAP Packaging Process—If the headspace oxygen concentration is found to be higher than expected soon after packaging, the gas flushing process may not be working as well as expected. Various techniques can evaluate whether the MAP system is functioning properly.
3.3.4 Storage Studies—As the method is non-destructive, the headspace can be monitored over time on individual samples to insure that results of storage studies such as shelf life testing are correctly interpreted.
SCOPE
1.1 This test method covers a procedure for determination of the oxygen concentration in the headspace within a sealed package without opening or compromising the integrity of the package.
1.2 This test method requires that chemically coated components be placed on the inside surface of the package before closing.
1.3 The package must be either transparent, translucent, or a transparent window must be affixed to the package surface without affecting the package’s integrity.
1.4 As this test method determines the oxygen headspace over time, the oxygen permeability can easily be calculated as ingress per unit time as long as the volume of the container is known.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Jan-2021
- Technical Committee
- F02 - Primary Barrier Packaging
- Drafting Committee
- F02.40 - Package Integrity
Overview
ASTM F2714-08(2021) is the Standard Test Method for Oxygen Headspace Analysis of Packages Using Fluorescent Decay, published by ASTM International. This standard provides a non-destructive procedure for measuring the oxygen concentration in the headspace of sealed packages, particularly those employing modified atmosphere packaging (MAP). Measuring headspace oxygen is critical for evaluating the protective performance of barrier materials in packaging and minimizing product spoilage, off-flavors, and quality concerns in packaged foods, pharmaceuticals, and chemicals.
Key Topics
- Non-destructive oxygen measurement: The method allows determination of oxygen levels inside sealed, transparent or translucent packages without breaking the seal or compromising the package integrity.
- Chemically coated sensor dots: Special fluorescent dye-coated components are affixed to the inside surface of the package prior to sealing, enabling sensitive oxygen detection.
- Fluorescent decay principle: The analysis is based on the fluorescence quenching effect - the presence of oxygen reduces the fluorescent lifetime of the coated dye (such as ruthenium complexes), which is measured using a pulsed light source and detector system.
- Quantitative results: The system compares measured decay times to those obtained from known oxygen concentrations, reporting results in percent oxygen, partial pressure, or parts per million (ppm).
- Calibration and environmental control: Proper calibration with certified oxygen concentrations, as well as careful control of temperature and humidity, are essential for accurate and repeatable test results.
Applications
ASTM F2714-08(2021) has broad applications in package testing and quality assurance for industries seeking reliable oxygen headspace analysis, including:
- Barrier performance studies: Evaluating the oxygen permeability of packaging films, containers, and closures by monitoring headspace oxygen changes over time.
- Leak detection: Identifying suspect packages with oxygen levels higher than expected or increasing more rapidly than normal, which may indicate package leaks.
- Modified Atmosphere Packaging (MAP) validation: Assessing the efficacy of MAP processes by verifying that oxygen levels meet the desired specifications immediately after packaging.
- Shelf-life and storage studies: Non-destructive monitoring of individual samples throughout storage, ensuring that package integrity is maintained and results of shelf-life testing are accurate.
- Pharmaceutical and chemical packaging: Ensuring that sensitive products are protected from oxygen ingress that could compromise stability and efficacy.
Related Standards
Those implementing ASTM F2714-08(2021) for oxygen headspace analysis may also find the following standards relevant:
- ASTM D3985: Standard Test Method for Oxygen Gas Transmission Rate Through Plastic Film and Sheeting Using a Coulometric Sensor.
- ASTM F1307: Standard Test Method for Oxygen Transmission Rate Through Dry Packages Using a Coulometric Sensor.
- ASTM F1927: Standard Test Method for Determination of Oxygen Gas Transmission Rate, Permeability and Permeance at Controlled Relative Humidity Through Barrier Materials Using a Coulometric Detector.
- ISO 15105-2: Plastics - Film and sheeting - Determination of gas-transmission rate - Part 2: Equal-pressure method.
Practical Value
Implementing ASTM F2714-08(2021) offers packaging professionals and quality managers a robust tool to:
- Ensure packaging materials and closure systems deliver required oxygen barrier performance.
- Detect leaks and process deficiencies that could compromise product shelf life or quality.
- Conduct ongoing, non-destructive monitoring of package integrity in storage and distribution.
- Demonstrate compliance with international best practices for package headspace testing.
Keywords: oxygen headspace analysis, modified atmosphere packaging, MAP, oxygen permeation, fluorescent decay, package integrity, oxygen transmission rate, leak detection, non-destructive testing, packaging quality assurance.
Buy Documents
ASTM F2714-08(2021) - Standard Test Method for Oxygen Headspace Analysis of Packages Using Fluorescent Decay
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM F2714-08(2021) is a standard published by ASTM International. Its full title is "Standard Test Method for Oxygen Headspace Analysis of Packages Using Fluorescent Decay". This standard covers: SIGNIFICANCE AND USE 3.1 The oxygen content of a package’s headspace is an important determinant of the packaging protection afforded by barrier materials. The package under test is typically MAP (modified atmosphere packaging) packaged. 3.2 Oxygen content is a key contributor to off-flavors and spoilage of various products, such as chemicals, food and pharmaceuticals. 3.3 The method determines the oxygen in a closed package headspace. This ability has application in: 3.3.1 Package Permeability Studies—The change of headspace composition over a known length of time allows the calculation of permeation. Since the headspace oxygen is measured as a percentage, the volume of the container’s headspace must be known to allow conversion into a quantity such as millilitres (ml) of oxygen. The use of this approach to measure permeation generally applies to empty package systems only as oxygen uptake or outgassing of contained products could affect results. 3.3.2 Leak Detection—If the headspace contains more oxygen than expected or is increasing faster than expected, a leak can be suspected. A wide variety of techniques can be employed to verify that a leak is present and to identify its location. If necessary or of interest, a leak rate may be calculated with known headspace volume and measured oxygen concentration change over time. 3.3.3 Efficacy of the MAP Packaging Process—If the headspace oxygen concentration is found to be higher than expected soon after packaging, the gas flushing process may not be working as well as expected. Various techniques can evaluate whether the MAP system is functioning properly. 3.3.4 Storage Studies—As the method is non-destructive, the headspace can be monitored over time on individual samples to insure that results of storage studies such as shelf life testing are correctly interpreted. SCOPE 1.1 This test method covers a procedure for determination of the oxygen concentration in the headspace within a sealed package without opening or compromising the integrity of the package. 1.2 This test method requires that chemically coated components be placed on the inside surface of the package before closing. 1.3 The package must be either transparent, translucent, or a transparent window must be affixed to the package surface without affecting the package’s integrity. 1.4 As this test method determines the oxygen headspace over time, the oxygen permeability can easily be calculated as ingress per unit time as long as the volume of the container is known. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 3.1 The oxygen content of a package’s headspace is an important determinant of the packaging protection afforded by barrier materials. The package under test is typically MAP (modified atmosphere packaging) packaged. 3.2 Oxygen content is a key contributor to off-flavors and spoilage of various products, such as chemicals, food and pharmaceuticals. 3.3 The method determines the oxygen in a closed package headspace. This ability has application in: 3.3.1 Package Permeability Studies—The change of headspace composition over a known length of time allows the calculation of permeation. Since the headspace oxygen is measured as a percentage, the volume of the container’s headspace must be known to allow conversion into a quantity such as millilitres (ml) of oxygen. The use of this approach to measure permeation generally applies to empty package systems only as oxygen uptake or outgassing of contained products could affect results. 3.3.2 Leak Detection—If the headspace contains more oxygen than expected or is increasing faster than expected, a leak can be suspected. A wide variety of techniques can be employed to verify that a leak is present and to identify its location. If necessary or of interest, a leak rate may be calculated with known headspace volume and measured oxygen concentration change over time. 3.3.3 Efficacy of the MAP Packaging Process—If the headspace oxygen concentration is found to be higher than expected soon after packaging, the gas flushing process may not be working as well as expected. Various techniques can evaluate whether the MAP system is functioning properly. 3.3.4 Storage Studies—As the method is non-destructive, the headspace can be monitored over time on individual samples to insure that results of storage studies such as shelf life testing are correctly interpreted. SCOPE 1.1 This test method covers a procedure for determination of the oxygen concentration in the headspace within a sealed package without opening or compromising the integrity of the package. 1.2 This test method requires that chemically coated components be placed on the inside surface of the package before closing. 1.3 The package must be either transparent, translucent, or a transparent window must be affixed to the package surface without affecting the package’s integrity. 1.4 As this test method determines the oxygen headspace over time, the oxygen permeability can easily be calculated as ingress per unit time as long as the volume of the container is known. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2714-08(2021) is classified under the following ICS (International Classification for Standards) categories: 71.040.40 - Chemical analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2714-08(2021) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2714 − 08 (Reapproved 2021)
Standard Test Method for
Oxygen Headspace Analysis of Packages Using Fluorescent
Decay
This standard is issued under the fixed designation F2714; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2.2 The package is gas flushed to a reduced level of oxygen
either manually or by subjecting the package to a filling
1.1 This test method covers a procedure for determination
operation.
of the oxygen concentration in the headspace within a sealed
2.3 Apulsing light source is directed through the package at
package without opening or compromising the integrity of the
package. the chemically treated dot (the package must be transparent,
translucent or contain a window through which the light can
1.2 This test method requires that chemically coated com-
pass).
ponents be placed on the inside surface of the package before
2.4 The fluorescent response from the dot is monitored and
closing.
the decay rate determined.
1.3 The package must be either transparent, translucent, or a
2.5 The internal oxygen content of the package is deter-
transparent window must be affixed to the package surface
mined by comparing the measured decay rate to the decay rate
without affecting the package’s integrity.
observed with known oxygen concentrations.
1.4 As this test method determines the oxygen headspace
over time, the oxygen permeability can easily be calculated as
3. Significance and Use
ingress per unit time as long as the volume of the container is
3.1 The oxygen content of a package’s headspace is an
known.
important determinant of the packaging protection afforded by
1.5 The values stated in SI units are to be regarded as
barrier materials. The package under test is typically MAP
standard. No other units of measurement are included in this
(modified atmosphere packaging) packaged.
standard.
3.2 Oxygen content is a key contributor to off-flavors and
1.6 This standard does not purport to address all of the
spoilage of various products, such as chemicals, food and
safety concerns, if any, associated with its use. It is the
pharmaceuticals.
responsibility of the user of this standard to establish appro-
3.3 The method determines the oxygen in a closed package
priate safety, health, and environmental practices and deter-
headspace. This ability has application in:
mine the applicability of regulatory limitations prior to use.
3.3.1 Package Permeability Studies—The change of head-
1.7 This international standard was developed in accor-
space composition over a known length of time allows the
dance with internationally recognized principles on standard-
calculation of permeation. Since the headspace oxygen is
ization established in the Decision on Principles for the
measured as a percentage, the volume of the container’s
Development of International Standards, Guides and Recom-
headspace must be known to allow conversion into a quantity
mendations issued by the World Trade Organization Technical
such as millilitres (ml) of oxygen. The use of this approach to
Barriers to Trade (TBT) Committee.
measure permeation generally applies to empty package sys-
tems only as oxygen uptake or outgassing of contained
2. Summary of Test Method
products could affect results.
2.1 Chemically coated components (dots) are affixed to the
3.3.2 Leak Detection—If the headspace contains more oxy-
inside surface of the package to be tested.
gen than expected or is increasing faster than expected, a leak
can be suspected. A wide variety of techniques can be
employed to verify that a leak is present and to identify its
location. If necessary or of interest, a leak rate may be
This test method is under the jurisdiction ofASTM Committee F02 on Primary
Barrier Packaging and is the direct responsibility of Subcommittee F02.40 on
calculated with known headspace volume and measured oxy-
Package Integrity.
gen concentration change over time.
Current edition approved Jan. 15, 2021. Published February 2021. Originally
3.3.3 Effıcacy of the MAP Packaging Process—If the head-
approved in 2008. Last previous edition approved in 2013 as F2714 – 08 (2013).
DOI: 10.1520/F2714-08R21. spaceoxygenconcentrationisfoundtobehigherthanexpected
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2714 − 08 (2021)
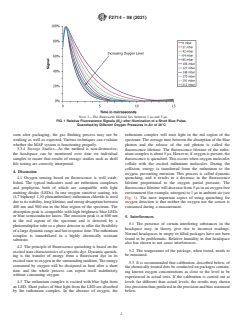
NOTE 1—The fluorescent lifetime lies between 1 µs and 5 µs.
FIG. 1 Relative Fluorescence Signals (I/I ) after Illumination of a Short Blue Pulse,
Quenched by Different Oxygen Pressures in Air of 20°C
soon after packaging, the gas flushing process may not be ruthenium complex will emit light in the red region of the
working as well as expected. Various techniques can evaluate spectrum. The average time between the absorption of the blue
whether the MAP system is functioning properly. photon and the release of the red photon is called the
3.3.4 Storage Studies—As the method is non-destructive,
fluorescence lifetime. The fluorescence lifetime of the ruthe-
the headspace can be monitored over time on individual nium complex is about 5 µs. However, if oxygen is present, the
samples to insure that results of storage studies such as shelf
fluorescence is quenched. This occurs when oxygen molecules
life testing are correctly interpreted. collide with the excited ruthenium molecules. During the
collision, energy is transferred from the ruthenium to the
4. Discussion
oxygen, preventing emission. This process is called dynamic
4.1 Oxygen sensing based on fluorescence is well estab- quenching, and it results in a decrease in the fluorescence
lished. The typical indicators used are ruthenium complexes
lifetime proportional to the oxygen partial pressure. The
and porphyrins both of which are compatible with light fluorescence lifetime will decrease from 5 µs in an oxygen free
emitting diodes (LEDs). In one oxygen sensitive coating, tris
environment (for example, nitrogen) to 1 µs in ambient air (see
(4,7 biphenyl 1,10 phenanthroline) ruthenium chloride is used Fig. 1). The most important aspect of using quenching for
duetoitsstability,longlifetime,andstrongabsorptionbetween
oxygen detection is that neither the oxygen nor the sensor is
400 nm and 500 nm in the blue region of the spectrum. The consumed during a measurement.
absorption peak is compatible with high brightness blue LEDs
or blue semiconductor lasers. The emission peak is at 600 nm
5. Interferences
in the red region of the spectrum and is detected by a
5.1 The presence of certain interfering substances in the
photomultiplier tube or a photo detector to offer the flexibility
headspace may, in theory, give rise to incorrect readings.
ofalargedynamicrangeandfastresponsetime.Theruthenium
Normal headspaces in empty or filled packages have not been
complex is immobilized in a highly chemically resistant
found to be problematic. Relative humidity in that headspace
substrate.
also has shown to not cause interferences.
4.2 The principle of fluorescence quenching is based on the
5.2 The temperature of the package, when tested, needs to
excitedstatecharacteristicsofaspecificdye.Dynamicquench-
be measured.
ing is the transfer of energy from a fluorescent dye in its
excited state to oxygen in the surrounding medium.The energy
5.3 It is recommended that calibration, described below, of
consumed by oxygen will be dissipated as heat after a short
the chemically treated dots be conducted on packages contain-
time and the whole process can repeat itself indefinitely
ing known oxygen concentrations as close to the level to be
without consuming oxygen.
experienced in actual tests. If the calibration is carried out at
4.3 The ruthenium complex is excited with blue light from levels far different than actual levels, the results may shown
an LED. Short pulses of blue light from the LED are absorbed lessprecisionthanpredictedintheprecisionandbiasstatement
below.
by the ruthenium complex. In the absence of oxygen, the
F2714 − 08 (2021)
6. Apparatus 12. Procedure
6.1 Chemically Treated Components (aka “dots”)—Coated 12.1 Calibration—Prior to testing, the fluorescent dots and
substrates of glass or flexible clear plastic have been found to the complete measuring system are to be calibrated.
be satisfactory. A fluorescent dye polymer is deposited on one 12.1.1 Fluorescent dots are affixed to the inside of a rigid
side of the substrate. package, flexible package, or a calibration fixture using adhe-
sives described above. The package or fixture is sealed and
6.2 Adhesive is
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...