ASTM F564-17
(Specification)Standard Specification and Test Methods for Metallic Bone Staples
Standard Specification and Test Methods for Metallic Bone Staples
ABSTRACT
This specification covers characterization of the design and mechanical function of metallic staples used in the internal fixation of the muscular skeletal system. It is not the intention of this specification to describe or specify specific designs for metallic bone staples. Different test methods shall be performed in order to determine the following mechanical properties of metallic bone staples: bending fatigue, pull-out fixation strength, soft tissue fixation strength, and elastic static bending.
SIGNIFICANCE AND USE
A1.3 Significance and Use
A1.3.1 This test method is used to determine the fatigue resistance of metallic bone staples when subjected to repetitive loading for large numbers of cycles. This information may also be useful for comparing the effect of variations in staple material, geometry, surface condition, or placement under certain circumstances.
A1.3.2 It is essential that uniform fatigue practices be established in order that such basic fatigue data be comparable and reproducible and can be correlated among laboratories.
A1.3.3 The results of fatigue tests are suitable for direct application to design only when the service conditions parallel the test conditions exactly. This test method may not be appropriate for all types of bone staple applications. The user is cautioned to consider the appropriateness of the test method in view of the materials being tested and their potential application.
SCOPE
1.1 This specification covers characterization of the design and mechanical function of metallic staples used in the internal fixation of the musculoskeletal system. It is not the intention of this specification to describe or specify specific designs for metallic bone staples.
1.2 This specification includes the following four test methods for measuring mechanical properties of metallic bone staples:
1.2.1 Test Method for Constant Amplitude Bending Fatigue Tests of Metallic Bone Staples—Annex A1.
1.2.2 Test Method for Pull-Out Fixation Strength of Metallic Bone Staples—Annex A2.
1.2.3 Test Method for Soft Tissue Fixation Strength of Metallic Bone Staples—Annex A3.
1.2.4 Test Method for Elastic Static Bending of Metallic Bone Staples—Annex A4.
1.3 The values stated in SI units are to be regarded as standard. Any other units of measurement included in this standard are shown for reference only.
1.4 Multiple test methods are included in this standard. However, the user is not necessarily obligated to test using all of the described methods. Instead, the user should only select, with justification, test methods that are appropriate for a particular device design. This may be only a subset of the herein described test methods.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2017
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.21 - Osteosynthesis
Relations
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Apr-2023
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Dec-2015
- Effective Date
- 01-Nov-2014
- Effective Date
- 01-Jun-2014
- Refers
ASTM F601-13 - Standard Practice for Fluorescent Penetrant Inspection of Metallic Surgical Implants - Effective Date
- 01-Dec-2013
- Refers
ASTM F565-04(2013) - Standard Practice for Care and Handling of Orthopedic Implants and Instruments - Effective Date
- 01-Oct-2013
- Effective Date
- 01-Jun-2013
- Refers
ASTM F86-12a - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 01-Dec-2012
- Effective Date
- 15-May-2012
- Refers
ASTM F86-12 - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 15-May-2012
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Aug-2011
Overview
ASTM F564-17: Standard Specification and Test Methods for Metallic Bone Staples provides guidelines for the characterization, testing, and evaluation of metallic staples intended for the internal fixation of the musculoskeletal system. Developed by ASTM International, this standard ensures consistent methods to assess the design and mechanical properties of these orthopedic devices, promoting safety and reliability in surgical implant applications. The standard focuses on test methods rather than prescribing specific staple designs, enabling flexibility across various devices and clinical needs.
Key Topics
Scope and Purpose
- Coverage includes evaluation of design and mechanical function of metallic bone staples used for internal bone fixation.
- The standard is not intended to dictate specific geometric or structural designs.
- It emphasizes mechanical testing methods for reproducibility and comparability among laboratories.
Core Mechanical Properties Assessed
- Bending Fatigue Resistance: Assesses the staple's durability under repetitive, cyclic loading, simulating long-term service conditions in vivo.
- Pull-Out Fixation Strength: Measures the force required to dislodge the staple from hard tissue (bone), critical for assessing stable fixation.
- Soft Tissue Fixation Strength: Evaluates the staple’s ability to secure soft tissue attachments without failure.
- Elastic Static Bending: Determines the staple’s elastic response to bending loads, indicating structural integrity under load.
Test Methodology
- Procedures designed according to internationally recognized practices.
- Methods include mechanical setups, specimen preparation guidelines, and failure criteria.
- Emphasis on selecting only those test methods relevant to the specific device design and intended clinical application.
Safety and Compliance
- The user must establish appropriate safety, health, and environmental measures.
- Alignment with World Trade Organization (WTO) principles for international standards facilitates global recognition and trade.
Applications
Device Design and Development:
- Provides manufacturers, engineers, and researchers with standardized mechanical tests for product development, quality control, and regulatory submissions.
- Enables comparative analysis of different staple materials, geometries, and surface treatments to optimize device performance.
Clinical Evaluation and Risk Assessment:
- Offered test data assists clinicians and regulatory agencies in assessing a staple's suitability for specific surgical applications, including bone fracture fixation and soft tissue attachment.
- Reliable determination of fixation strength helps minimize implant failure risks during post-surgical recovery.
Laboratory Testing & Product Benchmarking:
- Laboratories use these guidelines to ensure reproducibility and comparability of results, both within and between organizations.
- Facilitates benchmarking of new or modified designs against established standards, enhancing competitiveness and innovation.
Regulatory Compliance:
- Demonstrates adherence to recognized international mechanical testing standards, assisting with device approvals in global markets.
Related Standards
For comprehensive testing and evaluation, ASTM F564-17 should be used in conjunction with other relevant ASTM standards, including:
- ASTM E4: Practices for Force Verification of Testing Machines
- ASTM F75: Specification for Cobalt-28 Chromium-6 Molybdenum Alloy Castings for Surgical Implants
- ASTM F86: Practice for Surface Preparation and Marking of Metallic Surgical Implants
- ASTM F565: Practice for Care and Handling of Orthopedic Implants and Instruments
- ASTM F601: Practice for Fluorescent Penetrant Inspection of Metallic Surgical Implants
- ASTM F629: Practice for Radiography of Cast Metallic Surgical Implants
- ASTM F382: Specification and Test Method for Metallic Bone Plates
These related standards support complete device evaluation, covering material properties, manufacturing quality, and in-use performance, further ensuring metallic bone staples’ safety and effectiveness.
Keywords: ASTM F564-17, metallic bone staples, orthopedic implants, mechanical testing, bending fatigue, pull-out strength, soft tissue fixation, elastic static bending, surgical implants, standard specification, internal fixation, device evaluation, orthopedic device standard.
Buy Documents
ASTM F564-17 - Standard Specification and Test Methods for Metallic Bone Staples
REDLINE ASTM F564-17 - Standard Specification and Test Methods for Metallic Bone Staples
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F564-17 is a technical specification published by ASTM International. Its full title is "Standard Specification and Test Methods for Metallic Bone Staples". This standard covers: ABSTRACT This specification covers characterization of the design and mechanical function of metallic staples used in the internal fixation of the muscular skeletal system. It is not the intention of this specification to describe or specify specific designs for metallic bone staples. Different test methods shall be performed in order to determine the following mechanical properties of metallic bone staples: bending fatigue, pull-out fixation strength, soft tissue fixation strength, and elastic static bending. SIGNIFICANCE AND USE A1.3 Significance and Use A1.3.1 This test method is used to determine the fatigue resistance of metallic bone staples when subjected to repetitive loading for large numbers of cycles. This information may also be useful for comparing the effect of variations in staple material, geometry, surface condition, or placement under certain circumstances. A1.3.2 It is essential that uniform fatigue practices be established in order that such basic fatigue data be comparable and reproducible and can be correlated among laboratories. A1.3.3 The results of fatigue tests are suitable for direct application to design only when the service conditions parallel the test conditions exactly. This test method may not be appropriate for all types of bone staple applications. The user is cautioned to consider the appropriateness of the test method in view of the materials being tested and their potential application. SCOPE 1.1 This specification covers characterization of the design and mechanical function of metallic staples used in the internal fixation of the musculoskeletal system. It is not the intention of this specification to describe or specify specific designs for metallic bone staples. 1.2 This specification includes the following four test methods for measuring mechanical properties of metallic bone staples: 1.2.1 Test Method for Constant Amplitude Bending Fatigue Tests of Metallic Bone Staples—Annex A1. 1.2.2 Test Method for Pull-Out Fixation Strength of Metallic Bone Staples—Annex A2. 1.2.3 Test Method for Soft Tissue Fixation Strength of Metallic Bone Staples—Annex A3. 1.2.4 Test Method for Elastic Static Bending of Metallic Bone Staples—Annex A4. 1.3 The values stated in SI units are to be regarded as standard. Any other units of measurement included in this standard are shown for reference only. 1.4 Multiple test methods are included in this standard. However, the user is not necessarily obligated to test using all of the described methods. Instead, the user should only select, with justification, test methods that are appropriate for a particular device design. This may be only a subset of the herein described test methods. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This specification covers characterization of the design and mechanical function of metallic staples used in the internal fixation of the muscular skeletal system. It is not the intention of this specification to describe or specify specific designs for metallic bone staples. Different test methods shall be performed in order to determine the following mechanical properties of metallic bone staples: bending fatigue, pull-out fixation strength, soft tissue fixation strength, and elastic static bending. SIGNIFICANCE AND USE A1.3 Significance and Use A1.3.1 This test method is used to determine the fatigue resistance of metallic bone staples when subjected to repetitive loading for large numbers of cycles. This information may also be useful for comparing the effect of variations in staple material, geometry, surface condition, or placement under certain circumstances. A1.3.2 It is essential that uniform fatigue practices be established in order that such basic fatigue data be comparable and reproducible and can be correlated among laboratories. A1.3.3 The results of fatigue tests are suitable for direct application to design only when the service conditions parallel the test conditions exactly. This test method may not be appropriate for all types of bone staple applications. The user is cautioned to consider the appropriateness of the test method in view of the materials being tested and their potential application. SCOPE 1.1 This specification covers characterization of the design and mechanical function of metallic staples used in the internal fixation of the musculoskeletal system. It is not the intention of this specification to describe or specify specific designs for metallic bone staples. 1.2 This specification includes the following four test methods for measuring mechanical properties of metallic bone staples: 1.2.1 Test Method for Constant Amplitude Bending Fatigue Tests of Metallic Bone Staples—Annex A1. 1.2.2 Test Method for Pull-Out Fixation Strength of Metallic Bone Staples—Annex A2. 1.2.3 Test Method for Soft Tissue Fixation Strength of Metallic Bone Staples—Annex A3. 1.2.4 Test Method for Elastic Static Bending of Metallic Bone Staples—Annex A4. 1.3 The values stated in SI units are to be regarded as standard. Any other units of measurement included in this standard are shown for reference only. 1.4 Multiple test methods are included in this standard. However, the user is not necessarily obligated to test using all of the described methods. Instead, the user should only select, with justification, test methods that are appropriate for a particular device design. This may be only a subset of the herein described test methods. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F564-17 is classified under the following ICS (International Classification for Standards) categories: 11.040.30 - Surgical instruments and materials. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F564-17 has the following relationships with other standards: It is inter standard links to ASTM F564-10(2015), ASTM F2503-23e1, ASTM F629-20, ASTM F629-15, ASTM F382-14, ASTM E4-14, ASTM F601-13, ASTM F565-04(2013), ASTM F2503-13, ASTM F86-12a, ASTM F75-12, ASTM F86-12, ASTM F629-11, ASTM E467-08e1, ASTM E122-09e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F564-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:F564 −17
Standard Specification and Test Methods for
Metallic Bone Staples
ThisstandardisissuedunderthefixeddesignationF564;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
1.1 This specification covers characterization of the design
and mechanical function of metallic staples used in the internal
2. Referenced Documents
fixation of the musculoskeletal system. It is not the intention of
2.1 ASTM Standards:
this specification to describe or specify specific designs for
E4 Practices for Force Verification of Testing Machines
metallic bone staples.
E122 Practice for Calculating Sample Size to Estimate,With
1.2 This specification includes the following four test meth-
Specified Precision, the Average for a Characteristic of a
ods for measuring mechanical properties of metallic bone
Lot or Process
staples:
E467 Practice for Verification of Constant Amplitude Dy-
1.2.1 Test Method for ConstantAmplitude Bending Fatigue
namic Forces in an Axial Fatigue Testing System
Tests of Metallic Bone Staples—Annex A1.
F75 Specification for Cobalt-28 Chromium-6 Molybdenum
1.2.2 Test Method for Pull-Out Fixation Strength of Metal-
Alloy Castings and Casting Alloy for Surgical Implants
lic Bone Staples—Annex A2.
(UNS R30075)
1.2.3 Test Method for Soft Tissue Fixation Strength of
F86 Practice for Surface Preparation and Marking of Metal-
Metallic Bone Staples—Annex A3.
lic Surgical Implants
1.2.4 Test Method for Elastic Static Bending of Metallic
F382 SpecificationandTestMethodforMetallicBonePlates
Bone Staples—Annex A4.
F565 Practice for Care and Handling of Orthopedic Implants
and Instruments
1.3 The values stated in SI units are to be regarded as
standard. Any other units of measurement included in this F601 Practice for Fluorescent Penetrant Inspection of Me-
tallic Surgical Implants
standard are shown for reference only.
F629 Practice for Radiography of Cast Metallic Surgical
1.4 Multiple test methods are included in this standard.
Implants
However, the user is not necessarily obligated to test using all
F2503 Practice for Marking Medical Devices and Other
of the described methods. Instead, the user should only select,
Items for Safety in the Magnetic Resonance Environment
with justification, test methods that are appropriate for a
particular device design. This may be only a subset of the
3. Finish
herein described test methods.
3.1 Staples conforming to this specification shall be finished
1.5 This standard does not purport to address all of the
and identified in accordance with Practice F86, as appropriate.
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
4. Inspection Practices
priate safety, health and environmental practices and deter-
4.1 Staples made in accordance with Specification F75
mine the applicability of regulatory limitations prior to use.
should be inspected in accordance with Practice F601 or
1.6 This international standard was developed in accor-
X-rayed in accordance with Practice F629.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
5. Care and Handling
Development of International Standards, Guides and Recom-
5.1 Staples should be cared for and handled in accordance
with Practice F565, as appropriate.
This specification is under the jurisdiction of ASTM Committee F04 on
Medical and Surgical Materials and Devices and is the direct responsibility of
Subcommittee F04.21 on Osteosynthesis. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Sept. 1, 2017. Published October 2017. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1985. Last previous edition approved in 2015 as F564 – 10 (2015). Standards volume information, refer to the standard’s Document Summary page on
DOI: 10.1520/F0564-17. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F564−17
5.2 Consider Practice F2503 to identify potential hazards Materials should be chosen based on the design requirements
produced by interactions between the device and the MR of the particular device. ASTM subcommittee F04.12 main-
environment and for terms that may be used to label the device tains a number of specifications for materials that are suitable
for safety in the MR environment for surgical implant applications.
6. Materials 7. Keywords
6.1 Bone staples shall be fabricated from a metallic material 7.1 bendingtest;bonefixation;fatiguetest;fixationdevices;
intended for surgical implant applications. In addition, the metallic bone staples; orthopaedic medical devices; pullout
materials shall be biocompatible for the intended application. test; soft tissue fixation; surgical implants
ANNEXES
(Mandatory Information)
A1. TEST METHOD FOR CONSTANT AMPLITUDE BENDING FATIGUE TESTS OF METALLIC BONE STAPLES
A1.1. Scope A1.3.2 It is essential that uniform fatigue practices be
established in order that such basic fatigue data be comparable
A1.1.1 This test method covers procedures for the perfor-
and reproducible and can be correlated among laboratories.
mance of constant amplitude fatigue testing of metallic staples
used in internal fixation of the musculoskeletal system. This A1.3.3 The results of fatigue tests are suitable for direct
test method may be used when testing in air at ambient application to design only when the service conditions parallel
temperature or in an aqueous or physiological solution. the test conditions exactly. This test method may not be
appropriate for all types of bone staple applications. The user
A1.1.2 The values stated in SI units are to be regarded as
is cautioned to consider the appropriateness of the test method
standard. Any other units of measurement included in this
in view of the materials being tested and their potential
standard are shown for reference only.
application.
A1.1.3 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
A1.4. Apparatus
responsibility of the user of this standard to establish appro-
A1.4.1 Testing Machines,conformingtotherequirementsof
priate safety, health and environmental practices and deter-
Practices E4 and E467. The loads used for determining
mine the applicability of regulatory limitations prior to use.
strengths shall be within the loading range of the testing
A1.1.4 This international standard was developed in accor-
machine as defined in Practices E4 and E467.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the A1.4.2 Gripping Devices:
Development of International Standards, Guides and Recom-
A1.4.2.1 Staple Extensions—Pairs of specially designed
mendations issued by the World Trade Organization Technical
metal blocks that permit the holding of individual staples for
Barriers to Trade (TBT) Committee.
theapplicationofbendingfatigueloads.Thelegsofeachstaple
are fitted into fixation holes in each block with minimal
A1.2. Summary of Test Method
clearance to restrict bending of the staple within the hole. The
staple is fixed securely in the block using a moldable filling or
A1.2.1 Metallic bone staples are tested under bending loads
grouting agent. The extension design should minimize the
until the specimen fails or a predetermined number of cycles
weight to reduce the influence on the staple while maintaining
has been applied to it. Bending tests may be performed in one
sufficient stiffness to transfer the load to the staple without
of two modes: either pure, in-plane bending; or tension (or
undesirable deflection. Holes for pin and clevis fixation are
compression) combined with in-plane bending. Tests using
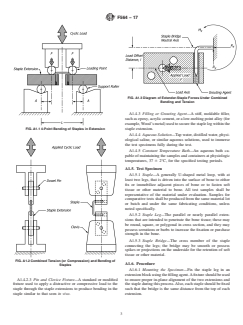
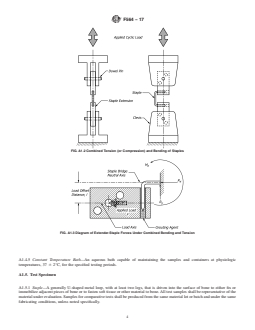
optional (see Figs. A1.1-A1.3).
either of these methods may be conducted at ambient condi-
tions or in aqueous or physiological solutions (at either room
NOTE A1.1—Variations in fixation hole configuration may be required
temperature or 37°C). for staple legs with noncircular cross sections. Also, it is necessary to
provide a gap between the underside of the staple bridge and edge of the
staple extender in most cases. This is necessary to eliminate contact
A1.3. Significance and Use
between the staple bridge (or other bridge features such as tissue spikes)
and the staple extender. However, this gap should be standardized within
A1.3.1 This test method is used to determine the fatigue
any test group as required.
resistance of metallic bone staples when subjected to repetitive
loading for large numbers of cycles.This information may also A1.4.2.2 4-Point Bend Fixture—A standard or modified
be useful for comparing the effect of variations in staple bending fixture that produces pure bending in the staple
material, geometry, surface condition, or placement under withoutappreciableshearortorsionwhenusedtoapplyloadto
certain circumstances. the staple through the staple extensions.
F564−17
FIG. A1.3Diagram of Extender-Staple Forces Under Combined
Bending and Tension
A1.4.3 Filling or Grouting Agent—A stiff, moldable filler,
suchasepoxy,acryliccement,oralow-meltingpointalloy(for
example,Wood’smetal)usedtosecurethestaplelegwithinthe
staple extension.
FIG. A1.14-Point Bending of Staples in Extension
A1.4.4 Aqueous Solution—Tap water, distilled water, physi-
ological saline, or similar aqueous solutions, used to immerse
the test specimens fully during the test.
A1.4.5 Constant Temperature Bath—An aqueous bath ca-
pable of maintaining the samples and containers at physiologic
temperatures, 37 6 2°C, for the specified testing periods.
A1.5. Test Specimen
A1.5.1 Staple—A generally U-shaped metal loop, with at
least two legs, that is driven into the surface of bone to either
fix or immobilize adjacent pieces of bone or to fasten soft
tissue or other material to bone. All test samples shall be
representative of the material under evaluation. Samples for
comparative tests shall be produced from the same material lot
or batch and under the same fabricating conditions, unless
noted specifically.
A1.5.2 Staple Leg—The parallel or nearly parallel exten-
sions that are intended to penetrate the bone tissue; these may
be round, square, or polygonal in cross section, and they may
possess serrations or barbs to increase the fixation or purchase
strength in the bone.
A1.5.3 Staple Bridge—The cross member of the staple
connecting the legs; the bridge may be smooth or possess
spikes or projections on the underside for the retention of soft
tissue or other material.
FIG. A1.2Combined Tension (or Compression) and Bending of
A1.6. Procedure
Staples
A1.6.1 Mounting the Specimen—Fix the staple leg in an
extensionblockusingthefillingagent.Afixtureshouldbeused
A1.4.2.3 Pin and Clevice Fixture—A standard or modified to ensure proper in-plane alignment of the two extensions and
fixture used to apply a distractive or compressive load to the the staple during this process.Also, each staple should be fixed
staple through the staple extensions to produce bending in the such that the bridge is the same distance from the top of each
staple similar to that seen in vivo. extension.
F564−17
NOTE A1.2—This distance is at the discretion of the operator, but it
induced in the specimen. This is accomplished most easily on
determines the portion of the staple subjected to the bending loads.
thestaplebridge,butitmaybepossibletoperformonaportion
A1.6.2 4-Point Bend Testing: of the staple leg or at the leg-bridge junction under certain
A1.6.2.1 Place the staple and attached staple extensions in circumstances and with certain staple designs. The recom-
mended technique is to strain gage the actual fatigue test
the 4-point bending fixture such that the loading point and
support rollers contact the staple extensions on either side of specimens, if possible, provided that the installation of the
the staple; direct contact of the rollers with the staple shall not strain gage will not influence the test results.
be permitted during the test. Alignment of the loading point
rollers shall be symmetric on the centerline between the
A1.7 Test Termination
support rollers (see Fig. A1.1).
A1.7.1 Continue the tests until the specimen fails or a
A1.6.2.2 Apply cyclic loads (sinusoidal, sawtooth, and so
predetermined number of cycles have been applied to the
forth), generating bending moments in the staple without
specimen. Failure should be defined as complete separation, a
permanent deformation. Appropriate starting loads should be
crack visible at a specified magnification, a crack of certain
50 to 75 % of the static bending strength, unless indicated
dimensions, or by some other criterion. State the criterion
otherwise.
selected for defining failure when reporting the results.
NOTE A1.3—It may be necessary to provide a low-friction means of
A1.7.2 Atest shall be considered invalid if loosening of the
maintaining the position of the staple and attached extensions. Also, the
staple occurs in the embedding medium, if noticeable yielding
fixtures should be designed so that loads are applied equally at the loading
points during each deflection throughout the test. occurs, or permanent deformation occurs in the specimen.
A1.6.2.3 Compute the bending moment, M, by the follow-
A1.8. Report
ing formula, where F = force applied at each loading point and
A = distance between the loading point and support roller:
A1.8.1 Report the following information:
M 5 FA (A1.1)
A1.8.1.1 Staple Description—Type, size, special features
bending
(barbs, spikes, and so forth), manufacturer, material, batch or
A1.6.2.4 Continue the test until failure of the staple, the
lot number, and dimensions (including leg length, bridge
fixation, or a predetermined number of load cycles have been
width, and length), as appropriate.
applied.
A1.8.1.2 Test Type—4-point or combined tension (or com-
A1.6.3 Combined Tension or (Compression) and Bending:
pression) and bending.
A1.6.3.1 Place the staple and attached staple extensions in
A1.8.1.3 Fixation Geometry—Load point separation dis-
the axial bending fixture. The pins and clevices should permit
tances (4-point bending), load offset distance (combined ten-
free rotation of the staple extensions, with minimal friction,
sion and bending), staple bridge-extension distance, and so
whilemaintainingalignmentofthestaplelegs(andextensions)
forth.
in the same plane (see Fig. A1.2).
A1.8.1.4 Minimum and maximum cycle loads, test fre-
A1.6.3.2 Apply cyclic loads (sinusoidal, sawtooth, and so
quency(forexample,cycles/s),andforcingfunctiontype(sine,
forth), generating bending moments in the staple without
ramp, saw tooth, and so forth).
permanent deformation. Appropriate starting loads should be
A1.8.1.5 Bending moment, M (N-m).
50 to 75 % of the static yield strength, unless indicated
A1.8.1.6 Load ratio, R, where R = minimum load/maximum
otherwise.
load.
NOTE A1.4—It may be necessary to maintain a minimum tensile or
A1.8.1.7 Test Environment—Ambient air or physiological
compressive load on the specimen throughout the test, since operating at
solution.
or near zero load may result in either loss of machine control due to
discontinuity in the load feedback loop or undesirable transient loading of
A1.8.1.8 Number of cycles at failure or test termination
the staple.
(runout).
A1.6.3.3 Compute the bending moment in the staple bridge,
A1.8.1.9 Location of fatigue fracture (if applicable).
M, by the following formula, where F = force applied at each
A1.8.1.10 Reason for test termination (that is, staple failure,
center of each pin and L = distance between the load applica-
fixation failure, runout to specified cycle limit, and so forth).
tion axis (that is, the pin center) and the neutral axis of the
staple bridge:
A1.9. Precision
M 5 FL ~see Fig. A1.3! (A1.2)
bending
A1.9.1 Intralaboratory and interlaboratory reproducibility
NOTE A1.5—The application of this test method produces bending,
have not been determined systematically.
tensile(orcompressive),andshearstressesinthestaple.Thedirectionand
magnitudes of these stresses should be analyzed using superposition
theory or other suitable methods.
A1.10 Rationale (Nonmandatory Information)
A1.6.3.4 Continue the test until failure of the staple or the
A1.10.1 Thistestmethodisintendedtoaidincharacterizing
fixation or a predetermined number of load cycles have been
the fatigue behavior of metallic bone staples used for the
applied.
fixation of bone to bone or soft tissue to bone. The data
A1.6.4 Stress Verification—It is recommended that strain obtained using this test method may be used to compare staple
gages(orextensometry)beusedtomeasurethebendingstrains materials, designs, or placements under certain circumstances.
F564−17
A1.10.2 Thistestmethodisintendedtosimulatepotential in
vivo staple loading conditions. However, in vitro testing of
these devices may not duplicate their in vivo behavior
adequately.
A2. TEST METHOD FOR PULL-OUT FIXATION STRENGTH OF METALLIC BONE STAPLES
A2.1. Scope A2.4.2 Gripping Devices:
A2.4.2.1 Hard Tissue Grips—Grips designed specifically
A2.1.1 This test method covers testing of the hard tissue
for secure grasping of bones, bone segments, or bone substi-
pull-outfixationstrengthofmetallicstaplesusedintheinternal
tutes with minimal material effects at the staple fixation site
fixation of the musculoskeletal system. This test method may
during the application of tension to the metal or fabric strap.
be used with physiologic bone or a synthetic substitute. It may
These grips shall allow for variation in bone geometry and
also be used when testing in an aqueous or physiological
permit proper orientation and alignment of the bone (or
solution.
substitute) during the test.
A2.1.2 The values stated in SI units are to be regarded as
A2.4.2.2 Metal or Fabric Strap Grips—Grips designed for
standard. Any other units of measurement included in this
secure grasping of the metal or fabric strap with minimal
standard are shown for reference only.
damage during the application of tension.Typical grips may be
A2.1.3 This standard does not purport to address all of the
actuated mechanically, pneumatically, or hydraulically to com-
safety concerns, if any, associated with its use. It is the press the strap between two flat grip faces parallel to the line of
responsibility of the user of this standard to establish appro-
load application. The grip faces may be smooth or serrated.
priate safety, health and environmental practices and deter- A2.4.2.3 Constant Temperature Bath—An aqueous bath
mine the applicability of regulatory limitations prior to use.
capable of maintaining the samples and containers at physi-
A2.1.4 This international standard was developed in accor- ologic temperatures, 37 6 2°C, for the specified testing
dance with internationally recognized principles on standard-
periods.
ization established in the Decision on Principles for the
A2.5. Test Specimen
Development of International Standards, Guides and Recom-
A2.5.1 Staple—A generally U-shaped metal loop, with at
mendations issued by the World Trade Organization Technical
least two legs, that is driven into the surface of bone to either
Barriers to Trade (TBT) Committee.
fix or immobilize adjacent pieces of bone or to fasten soft
A2.2. Summary of Test Method
tissue or other material to bone. All test samples shall be
representative of the material under evaluation. Samples for
A2.2.1 A thin, flat, flexible metal or fabric strap is fixed to
comparative tests shall be produced from the same material lot
bone (or a substitute) using a single staple. Tension is applied
or batch and under the same fabricating conditions, unless
equally to both ends of the strap in a direction parallel to the
noted specifically.
staple legs until loss of staple fixation occurs. Tests using this
test method may be conducted at ambient conditions or in
A2.5.2 Staple Leg—The parallel or nearly parallel exten-
aqueous or physiological solutions at either room temperature
sions that are intended to penetrate the bone tissue; these may
or 37°C.
be round, square, or polygonal in cross section, and they may
possess serrations or barbs to increase the fixation or purchase
A2.3. Significance and Use
strength in the bone.
A2.3.1 This test method is intended to help assess the axial
A2.5.3 Staple Bridge—The cross member of the staple
holding strength of various metallic bone staples used to fix
connecting the legs; the bridge may be smooth or possess
either soft or hard tissue to bone. This information may be
spikes or projections on the underside for the retention of soft
useful for comparisons of staple materials, designs, or place-
tissue or other material.
ment under certain circumstances.
A2.5.4 Bone—Fresh, fresh frozen, or preserved bone with
A2.3.2 Thistestmethodmaynotbeappropriateforalltypes
material properties representative of the staple placement site;
of bone staple applications. The user is cautioned to consider
either cadaveric or animal bone may be suitable.
the appropriateness of the test method in view of the materials
A2.5.5 Bone Substitute—Amaterial with known, consistent
being tested and their potential application.
properties,similarinstrength,density,orrigiditytofresh,fresh
A2.4. Apparatus
frozen, or preserved bone. Substitutes such as natural wood,
solid plastics, composites, or rigid foams may be suitable,
A2.4.1 Testing Machines,conformingtotherequirementsof
depending on type, material properties, and configuration.
Practices E4. The loads used for determining strengths shall be
within the loading range of the testing machine as defined in A2.5.6 Metal or Fabric Tension Strap—Athin, flat, flexible,
Practices E4. highly conformable metal strip or fabric (woven or braided)
F564−17
NOTEA2.1—In order to either reduce the inherent variability of manual
that is used to apply a balanced lifting or pull-out force under
staple insertion techniques or investigate the effects of deliberate variation
the staple bridge. The strap should have a width sufficient to
of insertion parameters, it is permissible to devise special instrumentation
support 90 % of the staple bridge and have sufficient tensile
in addition to that provided by the manufacturer. These additional
properties so that it does not deform plastically or rupture
instrumentsmaybeusedtolimitstapleangulation,controlinsertiondepth,
under the applied loads.
or standardize insertion forces. However, any use of nonstandard surgical
instrumentation for staple insertion shall be described and reported fully.
A2.5.7 Aqueous Solution—Tap water, distilled water, physi-
ological saline, or similar aqueous solutions, used to either
A2.6.3 Record the insertion method (manual or
maintain hydration of the soft tissue and bone used in the test
mechanical), staple orientation, and position from known
or bathe or immerse the test specimens fully during the test.
landmarks (if applicable).
A2.6. Procedure
A2.6.4 Fix the bone (or substitute) in the machine grips and
align so that tension can be applied to the strap ends parallel to
A2.6.1 Prepare the staple fixation site(s) on the bone (or
the staple legs. The bone should be gripped at a sufficient
substitute); this may require predrilling to accommodate the
distance from the staple fixation site so that the fixation site is
staple legs without damaging or fracturing the bone (or
left undamaged and undisturbed (see the schematic of staple
substitute).
pull-out testing in Fig. A2.1).
A2.6.2 Insert and seat the staple in the bone (or substitute)
in a standardized, repeatable manner using all appropriate
A2.6.5 Grasp the ends of the strap, place them together, and
surgical instrumentation specified by the manufacturer. The
securethembetweentheflatgripfacesunderequaltension(see
staple should be positioned over the tension strap, interposing
Fig. A2.1).
the metal or fabric between the staple bridge and bone surface,
NOTE A2.2—It is recommended that self-alignment devices such as
leaving equal lengths of the strap on either side of the staple
universaljoints,pinpivots,andsoforthbeplacedintheloadtraininorder
bridge. The strap should also be centered under the staple
to minimize the eccentric loading of either the tension strap or the staple.
bridge, and the strap bulk should permit the staple to be seated
A2.6.6 Apply the load to the strap at a constant
against the bone fully. The staple bridge should be perpendicu-
lar to the long axis of the strap. displacement, load, or strain rate.
FIG. A2.1Schematic of Staple Pull-Out Testing
F564−17
A2.6.7 Monitor the load-displacement continuously until A2.8.2.5 Gage Length—Theminimumdistancebetweenthe
one of the test termination criteria is reached. top of the staple bridge and the lower end of the grip faces.
A2.8.2.6 Peak pull-out strength (average and standard de-
NOTE A2.3—Elastic deformation in the strap may require a separate
viation) or other fixation criteria (for example, load at the onset
method of monitoring staple movement during the test.
of defined staple movement), as deemed appropriate.
A2.7 Test Termination
A2.8.2.7 Failure mode (pull-out of staple, rupture of strap,
bone fracture, and so forth).
A2.7.1 Testing of the samples shall be terminated when one
or more of the
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F564 − 10 (Reapproved 2015) F564 − 17
Standard Specification and Test Methods for
Metallic Bone Staples
This standard is issued under the fixed designation F564; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This specification covers characterization of the design and mechanical function of metallic staples used in the internal
fixation of the muscular skeletal musculoskeletal system. It is not the intention of this specification to describe or specify specific
designs for metallic bone staples.
1.2 This specification includes the following four test methods for measuring mechanical properties of metallic bone staples:
1.2.1 Test Method for Constant Amplitude Bending Fatigue Tests of Metallic Bone Staples—Annex A1.
1.2.2 Test Method for Pull-Out Fixation Strength of Metallic Bone Staples—Annex A2.
1.2.3 Test Method for Soft Tissue Fixation Strength of Metallic Bone Staples—Annex A3.
1.2.4 Test Method for Elastic Static Bending of Metallic Bone Staples—Annex A4.
1.3 The values stated in SI units are to be regarded as standard. NoAny other units of measurement are included in this
standard.standard are shown for reference only.
1.4 Multiple test methods are included in this standard. However, the user is not necessarily obligated to test using all of the
described methods. Instead, the user should only select, with justification, test methods that are appropriate for a particular device
design. This may be only a subset of the herein described test methods.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Verification of Testing Machines
E122 Practice for Calculating Sample Size to Estimate, With Specified Precision, the Average for a Characteristic of a Lot or
Process
E467 Practice for Verification of Constant Amplitude Dynamic Forces in an Axial Fatigue Testing System
F75 Specification for Cobalt-28 Chromium-6 Molybdenum Alloy Castings and Casting Alloy for Surgical Implants (UNS
R30075)
F86 Practice for Surface Preparation and Marking of Metallic Surgical Implants
F382 Specification and Test Method for Metallic Bone Plates
F565 Practice for Care and Handling of Orthopedic Implants and Instruments
F601 Practice for Fluorescent Penetrant Inspection of Metallic Surgical Implants
F629 Practice for Radiography of Cast Metallic Surgical Implants
F2503 Practice for Marking Medical Devices and Other Items for Safety in the Magnetic Resonance Environment
3. Finish
3.1 Staples conforming to this specification shall be finished and identified in accordance with Practice F86, as appropriate.
This specification is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.21 on Osteosynthesis.
Current edition approved March 1, 2015Sept. 1, 2017. Published April 2015October 2017. Originally approved in 1985. Last previous edition approved in 20102015 as
F564 – 10.F564 – 10 (2015). DOI: 10.1520/F0564-10R15.10.1520/F0564-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F564 − 17
4. Inspection Practices
4.1 Staples made in accordance with Specification F75 should be inspected in accordance with Practice F601 or X-rayed in
accordance with Practice F629.
5. Care and Handling
5.1 Staples should be cared for and handled in accordance with Practice F565, as appropriate.
5.2 Consider Practice F2503 to identify potential hazards produced by interactions between the device and the MR environment
and for terms that may be used to label the device for safety in the MR environment
6. Materials
6.1 Bone staples shall be fabricated from a metallic material intended for surgical implant applications. In addition, the materials
shall be biocompatible for the intended application. Materials should be chosen based on the design requirements of the particular
device. ASTM subcommittee F04.12 maintains a number of specifications for materials that are suitable for surgical implant
applications.
7. Keywords
7.1 bending test; bone fixation; fatigue test; fixation devices; metallic bone staples; orthopaedic medical devices; pullout test;
soft tissue fixation; surgical implants
ANNEXES
(Mandatory Information)
A1. TEST METHOD FOR CONSTANT AMPLITUDE BENDING FATIGUE TESTS OF METALLIC BONE STAPLES
A1.1. Scope
A1.1.1 This test method covers procedures for the performance of constant amplitude fatigue testing of metallic staples used in
internal fixation of the musculoskeletal system. This test method may be used when testing in air at ambient temperature or in an
aqueous or physiological solution.
A1.1.2 The values stated in SI units are to be regarded as standard. NoAny other units of measurement are included in this
standard.standard are shown for reference only.
A1.1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
A1.1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
A1.2. Summary of Test Method
A1.2.1 Metallic bone staples are tested under bending loads until the specimen fails or a predetermined number of cycles has been
applied to it. Bending tests may be performed in one of two modes: either pure, in-plane bending; or tension (or compression)
combined with in-plane bending. Tests using either of these methods may be conducted at ambient conditions or in aqueous or
physiological solutions (at either room temperature or 37°C).
A1.3. Significance and Use
A1.3.1 This test method is used to determine the fatigue resistance of metallic bone staples when subjected to repetitive loading
for large numbers of cycles. This information may also be useful for comparing the effect of variations in staple material, geometry,
surface condition, or placement under certain circumstances.
A1.3.2 It is essential that uniform fatigue practices be established in order that such basic fatigue data be comparable and
reproducible and can be correlated among laboratories.
F564 − 17
A1.3.3 The results of fatigue tests are suitable for direct application to design only when the service conditions parallel the test
conditions exactly. This test method may not be appropriate for all types of bone staple applications. The user is cautioned to
consider the appropriateness of the test method in view of the materials being tested and their potential application.
A1.4. Apparatus
A1.4.1 Testing Machines, conforming to the requirements of Practices E4 and E467. The loads used for determining strengths shall
be within the loading range of the testing machine as defined in Practices E4 and E467.
A1.4.2 Gripping Devices:
A1.4.2.1 Staple Extensions—Pairs of specially designed metal blocks that permit the holding of individual staples for the
application of bending fatigue loads. The legs of each staple are fitted into fixation holes in each block with minimal clearance to
restrict bending of the staple within the hole. The staple is fixed securely in the block using a moldable filling or grouting agent.
The extension design should minimize the weight to reduce the influence on the staple while maintaining sufficient stiffness to
transfer the load to the staple without undesirable deflection. Holes for pin and clevis fixation are optional (see Figs. A1.1-A1.3).
NOTE A1.1—Variations in fixation hole configuration may be required for staple legs with noncircular cross sections. Also, it is necessary to provide a
gap between the underside of the staple bridge and edge of the staple extender in most cases. This is necessary to eliminate contact between the staple
bridge (or other bridge features such as tissue spikes) and the staple extender. However, this gap should be standardized within any test group as required.
A1.4.2.2 4-Point Bend Fixture—A standard or modified bending fixture that produces pure bending in the staple without
appreciable shear or torsion when used to apply load to the staple through the staple extensions.
A1.4.2.3 Pin and Clevice Fixture—A standard or modified fixture used to apply a distractive or compressive load to the staple
through the staple extensions to produce bending in the staple similar to that seen in vivo.
A1.4.3 Filling or Grouting Agent—A stiff, moldable filler, such as epoxy, acrylic cement, or a low-melting point alloy (for
example, Wood’s metal) used to secure the staple leg within the staple extension.
A1.4.4 Aqueous Solution—Tap water, distilled water, physiological saline, or similar aqueous solutions, used to immerse the test
specimens fully during the test.
FIG. A1.1 4-Point Bending of Staples in Extension
F564 − 17
FIG. A1.2 Combined Tension (or Compression) and Bending of Staples
FIG. A1.3 Diagram of Extender-Staple Forces Under Combined Bending and Tension
A1.4.5 Constant Temperature Bath—An aqueous bath capable of maintaining the samples and containers at physiologic
temperatures, 37 6 2°C, for the specified testing periods.
A1.5. Test Specimen
A1.5.1 Staple—A generally U-shaped metal loop, with at least two legs, that is driven into the surface of bone to either fix or
immobilize adjacent pieces of bone or to fasten soft tissue or other material to bone. All test samples shall be representative of the
material under evaluation. Samples for comparative tests shall be produced from the same material lot or batch and under the same
fabricating conditions, unless noted specifically.
F564 − 17
A1.5.2 Staple Leg—The parallel or nearly parallel extensions that are intended to penetrate the bone tissue; these may be round,
square, or polygonal in cross section, and they may possess serrations or barbs to increase the fixation or purchase strength in the
bone.
A1.5.3 Staple Bridge—The cross member of the staple connecting the legs; the bridge may be smooth or possess spikes or
projections on the underside for the retention of soft tissue or other material.
A1.6. Procedure
A1.6.1 Mounting the Specimen—Fix the staple leg in an extension block using the filling agent. A fixture should be used to ensure
proper in-plane alignment of the two extensions and the staple during this process. Also, each staple should be fixed such that the
bridge is the same distance from the top of each extension.
NOTE A1.2—This distance is at the discretion of the operator, but it determines the portion of the staple subjected to the bending loads.
A1.6.2 4-Point Bend Testing:
A1.6.2.1 Place the staple and attached staple extensions in the 4-point bending fixture such that the loading point and support
rollers contact the staple extensions on either side of the staple; direct contact of the rollers with the staple shall not be permitted
during the test. Alignment of the loading point rollers shall be symmetric on the centerline between the support rollers (see Fig.
A1.1).
A1.6.2.2 Apply cyclic loads (sinusoidal, sawtooth, and so forth), generating bending moments in the staple without permanent
deformation. Appropriate starting loads should be 50 to 75 % of the static bending strength, unless indicated otherwise.
NOTE A1.3—It may be necessary to provide a low-friction means of maintaining the position of the staple and attached extensions. Also, the fixtures
should be designed so that loads are applied equally at the loading points during each deflection throughout the test.
A1.6.2.3 Compute the bending moment, M, by the following formula, where F = force applied at each loading point and A =
distance between the loading point and support roller,roller:
M 5 FA (A1.1)
bending
M = FA.
bending
A1.6.2.4 Continue the test until failure of the staple, the fixation, or a predetermined number of load cycles hashave been applied.
A1.6.3 Combined Tension or (Compression) and Bending:
A1.6.3.1 Place the staple and attached staple extensions in the axial bending fixture. The pins and clevices should permit free
rotation of the staple extensions, with minimal friction, while maintaining alignment of the staple legs (and extensions) in the same
plane (see Fig. A1.2).
A1.6.3.2 Apply cyclic loads (sinusoidal, sawtooth, and so forth), generating bending moments in the staple without permanent
deformation. Appropriate starting loads should be 50 to 75 % of the static yield strength, unless indicated otherwise.
NOTE A1.4—It may be necessary to maintain a minimum tensile or compressive load on the specimen throughout the test, since operating at or near zero
load may result in either loss of machine control due to discontinuity in the load feedback loop or undesirable transient loading of the staple.
A1.6.3.3 Compute the bending moment in the staple bridge, M, by the following formula, where F = force applied at each center
of each pin and L = distance between the load application axis, thataxis (that is, the pin center,center) and the neutral axis of the
staple bridge, bridge:
M 5 FL ~see Fig. A1.3! (A1.2)
bending
M = FL (see Fig. A1.3).
bending
NOTE A1.5—The application of this test method produces bending, tensile (or compressive), and shear stresses in the staple. The direction and magnitudes
of these stresses should be analyzed using superposition theory or other suitable methods.
F564 − 17
A1.6.3.4 Continue the test until failure of the staple or the fixation or a predetermined number of load cycles hashave been applied.
A1.6.4 Stress Verification—It is recommended that strain gages (or extensometry) be used to measure the bending strains induced
in the specimen. This is accomplished most easily on the staple bridge, but it may be possible to perform on a portion of the staple
leg or at the leg-bridge junction under certain circumstances and with certain staple designs. The recommended technique is to
strain gage the actual fatigue test specimens, if possible, provided that the installation of the strain gage will not influence the test
results.
A1.7 Test Termination
A1.7.1 Continue the tests until the specimen fails or a predetermined number of cycles hashave been applied to the specimen.
Failure should be defined as complete separation, a crack visible at a specified magnification, a crack of certain dimensions, or by
some other criterion. State the criterion selected for defining failure when reporting the results.
A1.7.2 A test shall be considered invalid if loosening of the staple occurs in the embedding medium, if noticeable yielding occurs,
or permanent deformation occurs in the specimen.
A1.8. Report
A1.8.1 Report the following information:
A1.8.1.1 Staple Description—Type, size, special features (barbs, spikes, and so forth), manufacturer, material, batch or lot number,
and dimensions (including leg length, bridge width, and length), as appropriate.
A1.8.1.2 Test Type—4-point or combined tension (or compression) and bending.
A1.8.1.3 Fixation Geometry—Load point separation distances (4-point bending), load offset distance (combined tension and
bending), staple bridge-extension distance, and so forth.
A1.8.1.4 Minimum and maximum cycle loads, test frequency (for example, cycles/s), and forcing function type (sine, ramp, saw
tooth, and so forth).
A1.8.1.5 Bending moment, M (N-m).
A1.8.1.6 Load ratio, R, where R = minimum load/maximum load.
A1.8.1.7 Test Environment—Ambient air or physiological solution.
A1.8.1.8 Number of cycles at failure or test termination (runout).
A1.8.1.9 Location of fatigue fracture (if applicable).
A1.8.1.10 Reason for test termination, thattermination (that is, staple failure, fixation failure, runout to specified cycle limit, and
so forth.forth).
A1.9. Precision
A1.9.1 Intralaboratory and interlaboratory reproducibility have not been determined systematically.
A1.10 Rationale (Nonmandatory Information)
A1.10.1 This test method is intended to aid in characterizing the fatigue behavior of metallic bone staples used for the fixation
of bone to bone or soft tissue to bone. The data obtained using this test method may be used to compare staple materials, designs,
or placements under certain circumstances.
F564 − 17
A1.10.2 This test method is intended to simulate potential in vivo staple loading conditions. However, in vitro testing of these
devices may not duplicate their in vivo behavior adequately.
A2. TEST METHOD FOR PULL-OUT FIXATION STRENGTH OF METALLIC BONE STAPLES
A2.1. Scope
A2.1.1 This test method covers testing of the hard tissue pull-out fixation strength of metallic staples used in the internal fixation
of the musculoskeletal system. This test method may be used with physiologic bone or a synthetic substitute. It may also be used
when testing in an aqueous or physiological solution.
A2.1.2 The values stated in SI units are to be regarded as standard. NoAny other units of measurement are included in this
standard.standard are shown for reference only.
A2.1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
A2.1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
A2.2. Summary of Test Method
A2.2.1 A thin, flat, flexible metal or fabric strap is fixed to bone (or a substitute) using a single staple. Tension is applied equally
to both ends of the strap in a direction parallel to the staple legs until loss of staple fixation occurs. Tests using this test method
may be conducted at ambient conditions or in aqueous or physiological solutions at either room temperature or 37°C.
A2.3. Significance and Use
A2.3.1 This test method is intended to help assess the axial holding strength of various metallic bone staples used to fix either soft
or hard tissue to bone. This information may be useful for comparisons of staple materials, designs, or placement under certain
circumstances.
A2.3.2 This test method may not be appropriate for all types of bone staple applications. The user is cautioned to consider the
appropriateness of the test method in view of the materials being tested and their potential application.
A2.4. Apparatus
A2.4.1 Testing Machines, conforming to the requirements of Practices E4. The loads used for determining strengths shall be within
the loading range of the testing machine as defined in Practices E4.
A2.4.2 Gripping Devices:
A2.4.2.1 Hard Tissue Grips—Grips designed specifically for secure grasping of bones, bone segments, or bone substitutes with
minimal material effects at the staple fixation site during the application of tension to the metal or fabric strap. These grips
mustshall allow for variation in bone geometry and permit proper orientation and alignment of the bone (or substitute) during the
test.
A2.4.2.2 Metal or Fabric Strap Grips—Grips designed for secure grasping of the metal or fabric strap with minimal damage
during the application of tension. Typical grips may be actuated mechanically, pneumatically, or hydraulically to compress the strap
between two flat grip faces parallel to the line of load application. The grip faces may be smooth or serrated.
A2.4.2.3 Constant Temperature Bath—An aqueous bath capable of maintaining the samples and containers at physiologic
temperatures, 37 6 2°C, for the specified testing periods.
F564 − 17
A2.5. Test Specimen
A2.5.1 Staple—A generally U-shaped metal loop, with at least two legs, that is driven into the surface of bone to either fix or
immobilize adjacent pieces of bone or to fasten soft tissue or other material to bone. All test samples shall be representative of the
material under evaluation. Samples for comparative tests shall be produced from the same material lot or batch and under the same
fabricating conditions, unless noted specifically.
A2.5.2 Staple Leg—The parallel or nearly parallel extensions that are intended to penetrate the bone tissue; these may be round,
square, or polygonal in cross section, and they may possess serrations or barbs to increase the fixation or purchase strength in the
bone.
A2.5.3 Staple Bridge—The cross member of the staple connecting the legs; the bridge may be smooth or possess spikes or
projections on the underside for the retention of soft tissue or other material.
A2.5.4 Bone—Fresh, fresh frozen, or preserved bone with material properties representative of the staple placement site; either
cadaveric or animal bone may be suitable.
A2.5.5 Bone Substitute—A material with known, consistent properties, similar in strength, density, or rigidity to fresh, fresh
frozen, or preserved bone. Substitutes such as natural wood, solid plastics, composites, or rigid foams may be suitable, depending
on type, material properties, and configuration.
A2.5.6 Metal or Fabric Tension Strap—A thin, flat, flexible, highly conformable metal strip or fabric (woven or braided) that is
used to apply a balanced lifting or pull-out force under the staple bridge. The strap should have a width sufficient to support 90 %
of the staple bridge and have sufficient tensile properties so that it does not deform plastically or rupture under the applied loads.
A2.5.7 Aqueous Solution—Tap water, distilled water, physiological saline, or similar aqueous solutions, used to either maintain
hydration of the soft tissue and bone used in the test or bathe or immerse the test specimens fully during the test.
A2.6. Procedure
A2.6.1 Prepare the staple fixation site(s) on the bone (or substitute); this may require predrilling to accommodate the staple legs
without damagedamaging or fracture of fracturing the bone (or substitute).
A2.6.2 Insert and seat the staple in the bone (or substitute) in a standardized, repeatable manner using all appropriate surgical
instrumentation specified by the manufacturer. The staple should be positioned over the tension strap, interposing the metal or
fabric between the staple bridge and bone surface, leaving equal lengths of the strap on either side of the staple bridge. The strap
should also be centered under the staple bridge, and the strap bulk should permit the staple to be seated against the bone fully. The
staple bridge should be perpendicular to the long axis of the strap.
NOTE A2.1—In order to either reduce the inherent variability of manual staple insertion techniques or investigate the effects of deliberate variation of
insertion parameters, it is permissible to devise special instrumentation in addition to that provided by the manufacturer. These additional instruments may
be used to limit staple angulation, control insertion depth, or standardize insertion forces. However, any use of nonstandard surgical instrumentation for
staple insertion mustshall be described and reported fully.
A2.6.3 Record the insertion method (manual or mechanical), staple orientation, and position from known landmarks (if
applicable).
A2.6.4 Fix the bone (or substitute) in the machine grips and align so that tension can be applied to the strap ends parallel to the
staple legs. The bone should be gripped at a sufficient distance from the staple fixation site so that the fixation site is left undamaged
and undisturbed (see the schematic of staple pull-out testing in Fig. A2.1).
A2.6.5 Grasp the ends of the strap, place them together, and secure them between the flat grip faces under equal tension (see Fig.
A2.1).
NOTE A2.2—It is recommended that self-alignment devices such as universal joints, pin pivots, and so forth be placed in the load train in order to minimize
F564 − 17
FIG. A2.1 Schematic of Staple Pull-Out Testing
the eccentric loading of either the tension strap or the staple.
A2.6.6 Apply the load to the strap at a constant displacement, load, or strain rate.
A2.6.7 Monitor the load-displacement continuously until one of the test termination criteria is reached.
NOTE A2.3—Elastic deformation in the strap may require a separate method of monitoring staple movement during the test.
A2.7 Test Termination
A2.7.1 Testing of the samples shall be terminated when one or more of the following has occurred:
A2.7.1.1 A significant drop in tensile load after peak,
A2.7.1.2 Visible tearing or rupture of the strap (or substitute),
A2.7.1.3 Fracture of the bone (or bone substitute), or
A2.7.1.4 Loosening of the staple(s) or disruption of the staple-bone interface.
A2.8. Report
A2.8.1 Report the following information:
A2.8.1.1 Staple Description—Type, size, special features (barbs, spikes, and so forth), manufacturer, material, batch or lot number,
and dimensions (including leg length, bridge width, and length), as appropriate.
F564 − 17
A2.8.1.2 Fixation site and orientation (if applicable).
A2.8.1.3 Staple
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...