ASTM D6317-98(2009)
(Test Method)Standard Test Method for Low Level Determination of Total Carbon, Inorganic Carbon and Organic Carbon in Water by Ultraviolet, Persulfate Oxidation, and Membrane Conductivity Detection
Standard Test Method for Low Level Determination of Total Carbon, Inorganic Carbon and Organic Carbon in Water by Ultraviolet, Persulfate Oxidation, and Membrane Conductivity Detection
SIGNIFICANCE AND USE
This test method is used for determination of the carbon content of water from a variety of natural, domestic, and industrial sources. In its most common form, this test method is used to measure organic carbon as a means of monitoring organic impurities in high purity process water used in industries such as nuclear power, pharmaceutical, and electronics.
SCOPE
1.1 This test method covers the determination of total carbon (TC), inorganic carbon (IC), and total organic carbon (TOC) in water in the range from 10 to 1000 μg/L of carbon. This method is for laboratory or grab sample applications and has been subjected to an interlaboratory study under the guidelines of D2777. Test Method D5997 can be used for on-line determinations. The test method utilizes persulfate or ultraviolet oxidation of organic carbon, or both coupled with a CO2 selective membrane to recover the CO2 into deionized water. The change in conductivity of the deionized water is measured and related to carbon concentration in the oxidized sample. Inorganic carbon is determined in a similar manner without the oxidation step. In both cases, the sample is acidified to facilitate CO2 recovery through the membrane. The relationship between the conductivity measurement and carbon concentration is described by a set of chemometric equations for the chemical equilibrium of CO2, HCO3–, and H+, and the relationship between the ionic concentrations and the conductivity. The chemometric model includes the temperature dependence of the equilibrium constants and the specific conductances resulting in linear response of the method over the stated range of TOC. See Test Method D4519 for a discussion of the measurement of CO2 by conductivity.
1.2 This test method has the advantage of a very high sensitivity detector that allows very low detection levels on relatively small volumes of sample. Also, use of two measurement channels allows determination of CO2 in the sample independently of organic carbon. Isolation of the conductivity detector from the sample by the CO2 selective membrane results in a very stable calibration, with minimal interferences.
1.3 This test method was used successfully with reagent water spiked with various organic materials. It is the user's responsibility to ensure the validity of this test method for waters of untested matrices.
1.4 In addition to laboratory analyses, this test method may be adapted to on line monitoring. See Test Method D5997.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
Relations
Standards Content (Sample)
NOTICE: This standard has either been superseded and replaced by a new version or withdrawn.
Contact ASTM International (www.astm.org) for the latest information
Designation: D6317 − 98(Reapproved 2009)
Standard Test Method for
Low Level Determination of Total Carbon, Inorganic Carbon
and Organic Carbon in Water by Ultraviolet, Persulfate
Oxidation, and Membrane Conductivity Detection
This standard is issued under the fixed designation D6317; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope responsibility to ensure the validity of this test method for
waters of untested matrices.
1.1 This test method covers the determination of total
carbon (TC), inorganic carbon (IC), and total organic carbon 1.4 In addition to laboratory analyses, this test method may
(TOC) in water in the range from 10 to 1000 µg/L of carbon. be adapted to on line monitoring. See Test Method D5997.
This method is for laboratory or grab sample applications and
1.5 This standard does not purport to address all of the
has been subjected to an interlaboratory study under the
safety concerns, if any, associated with its use. It is the
guidelines of D2777. Test Method D5997 can be used for
responsibility of the user of this standard to establish appro-
on-line determinations. The test method utilizes persulfate or
priate safety and health practices and determine the applica-
ultraviolet oxidation of organic carbon, or both coupled with a
bility of regulatory limitations prior to use.
CO selective membrane to recover the CO into deionized
2 2
water. The change in conductivity of the deionized water is
2. Referenced Documents
measured and related to carbon concentration in the oxidized
2.1 ASTM Standards:
sample. Inorganic carbon is determined in a similar manner
D1129 Terminology Relating to Water
without the oxidation step. In both cases, the sample is
D1192 Guide for Equipment for Sampling Water and Steam
acidifiedtofacilitateCO recoverythroughthemembrane.The
2 3
in Closed Conduits (Withdrawn 2003)
relationshipbetweentheconductivitymeasurementandcarbon
D1193 Specification for Reagent Water
concentration is described by a set of chemometric equations
D2777 Practice for Determination of Precision and Bias of
– +
for the chemical equilibrium of CO , HCO , and H , and the
2 3
Applicable Test Methods of Committee D19 on Water
relationship between the ionic concentrations and the conduc-
D3370 Practices for Sampling Water from Closed Conduits
tivity. The chemometric model includes the temperature de-
D4210 Practice for Intralaboratory Quality Control Proce-
pendence of the equilibrium constants and the specific conduc-
dures and a Discussion on Reporting Low-Level Data
tancesresultinginlinearresponseofthemethodoverthestated
(Withdrawn 2002)
range of TOC. See Test Method D4519 for a discussion of the
D5997 Test Method for On-Line Monitoring of Total
measurement of CO by conductivity.
Carbon, Inorganic Carbon in Water by Ultraviolet, Persul-
1.2 This test method has the advantage of a very high
fate Oxidation, and Membrane Conductivity Detection
sensitivity detector that allows very low detection levels on
D4519 Test Method for On-Line Determination of Anions
relatively small volumes of sample.Also, use of two measure-
and Carbon Dioxide in High Purity Water by Cation
ment channels allows determination of CO in the sample
2 Exchange and Degassed Cation Conductivity
independently of organic carbon. Isolation of the conductivity
detector from the sample by the CO selective membrane
3. Terminology
results in a very stable calibration, with minimal interferences.
3.1 Definitions—For definitions of terms used in this test
1.3 This test method was used successfully with reagent
method, refer to Terminology D1129.
water spiked with various organic materials. It is the user’s
3.2 Definitions of Terms Specific to This Standard:
This test method is under the jurisdiction of ASTM Committee D19 on Water
and is the direct responsibility of Subcommittee D19.03 on Sampling Water and For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Water-Formed Deposits, Analysis of Water for Power Generation and Process Use, contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
On-Line Water Analysis, and Surveillance of Water. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Oct. 1, 2009. Published November 2009. Originally the ASTM website.
approved in 1998. Last previous edition approved in 2004 as D6317 – 98 (2004). The last approved version of this historical standard is referenced on
DOI: 10.1520/D6317-98R09. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6317 − 98 (2009)
3.2.1 inorganic carbon (IC)—carbon in the form of carbon 6.2 Chloride ion above 250 mg/L tends to interfere with
dioxide, carbonate ion, or bicarbonate ion. oxidative reaction mechanisms in this test method. Follow
manufacturer’s instructions for dealing with this problem.
3.2.2 refractory material—that which cannot be oxidized
Other interferences have been investigated and found to be
completely under the test method conditions.
minimal under most conditions. Refer to the reference (2) for
3.2.3 total carbon (TC)—the sum of IC and TOC.
more information.
3.2.4 total organic carbon (TOC)—carbon in the form of
6.3 Note that error will be introduced when the method of
organic compounds.
difference is used to derive a relatively small level from two
large levels. In this case the vacuum degassing unit on the
4. Summary of Test Method
instrument should be used to reduce the concentration of IC
4.1 Carbon can occur in water as inorganic and organic
prior to measurement. Alternatively, the sample can be acidi-
compounds.This test method can be used to make independent
fied and sparged prior to introduction into the instrument.
measurementsofICandTCandcanalsodetermineTOCasthe
6.4 Use of the vacuum degassing unit or sparging the
difference of TC and IC. If IC is high relative to TOC it is
sample may cause loss of volatile organic compounds, thus
desirable to use a vacuum degassing unit to reduce the IC
yielding a value lower than the true TOC level. At low TOC
concentration as part of the measurement.Alternatively, the IC
levels,thedegassingunitmayintroduceameasurableTOCand
can be removed by acidifying and sparging the sample prior to
IC background. The user should characterize the background
injection into the instrument. The basic steps of the procedure
and performance of the degassing module for their application.
are as follows:
Table 1 provides typical IC removal performance and back-
(1) Removal of IC, if desired, by vacuum degassing;
ground levels of the vacuum degassing unit.
(2) Conversion of remaining inorganic carbon to CO by
6.5 Contamination of the sample with both CO and organic
action of acid in both channels and oxidation of total carbon to
carbon is a severe problem as lower levels of analyte are
CO by action of ultraviolet (UV) radiation in the TC channel.
attempted.Throughoutthismethodtheanalystmustbevigilant
(Acid-persulfate can be added but is usually not required at
for all potential sources of contamination and must monitor
TOC levels below 1 ppm).
blanks and adjust operations to prevent contamination.
(3) Detection of CO that is swept out of the U.V. reactor
and delay coil by the liquid stream and passed through
7. Apparatus
membranes that allow the specific passage of CO to high
purity water where change in conductivity is measured and; 7.1 Apparatus for Carbon Determination—Atypical instru-
(4) Conversion of the conductivity detector signal to a
ment consists of reagent and sample introduction mechanism,
display of carbon concentration in parts per million reaction vessel, detector, control system, and a display. Fig. 1
(ppm=mg/L) or parts per billion (ppb=µg/L). The IC channel
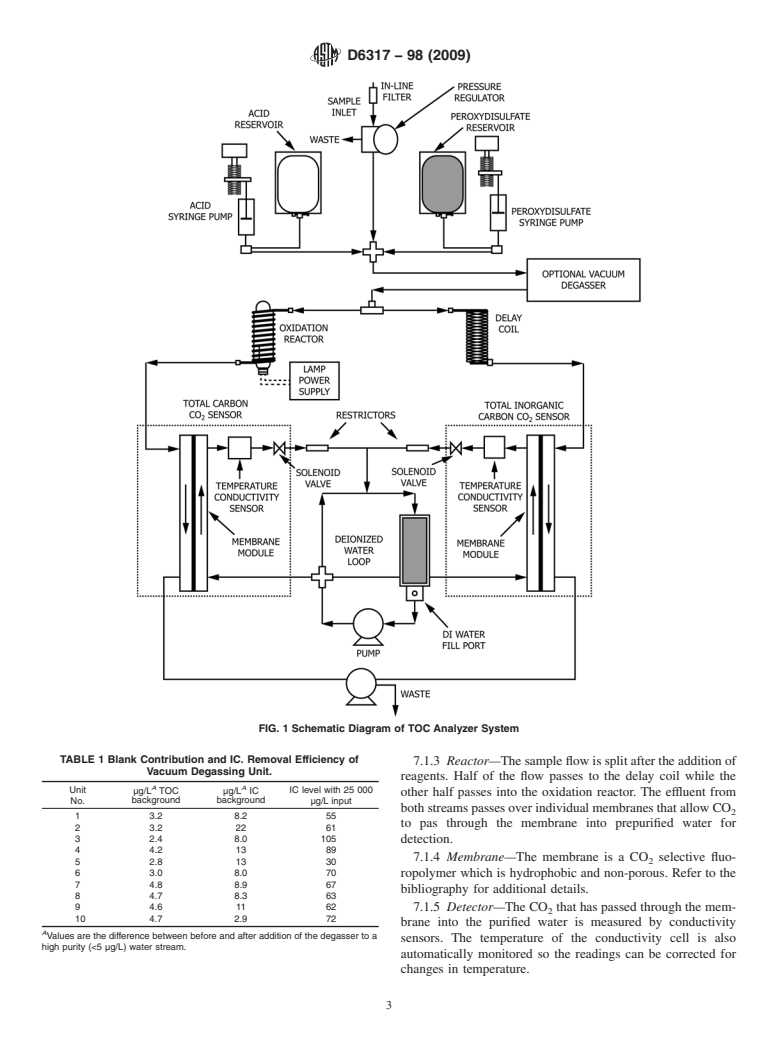
shows a diagram of such an arrangement.
reading is subtracted from the TC channel to give a TOC 7.1.1 Sampling Needle—A double chambered needle ca-
reading. A diagram of suitable apparatus is given in Fig. 1.
pable of piercing the sample bottle septum and pulling sample
References 1-5 provide additional information on the method. from the bottom of the bottle is used. The second chamber
vents the top of the bottle to prevent vacuum build up as the
5. Significance and Use sample is withdrawn. Typically this needle is mounted on an
autosampler to provide unattended analysis of several samples.
5.1 This test method is used for determination of the carbon
7.1.2 I.C. Removal—Vacuum degassing requires the manu-
content of water from a variety of natural, domestic, and
facturer’s module which includes a vacuum pump and a
industrial sources. In its most common form, this test method
hollow fiber membrane assembly. Use of this vacuum degasser
is used to measure organic carbon as a means of monitoring
will remove essentially all IC as part of the analysis. The
organic impurities in high purity process water used in indus-
membrane module consists of a tube and shell arrangement of
tries such as nuclear power, pharmaceutical, and electronics.
microporous polypropylene hollow fibers. Sample flows along
the inside of the fibers, while air is passed on the shell
6. Interferences and Limitations
side-counterflow to the sample flow. The shell side pressure is
6.1 The oxidation of dissolved carbon to CO is brought
reduced by means of a vacuum pump on the air outlet. The
about at relatively low temperatures by the chemical action of
sample is acidified before introduction into the degasser to
reactive species produced by UV-irradiated persulfate ions and
facilitate CO transport through the hollow fibers. Sparging
water. Not all suspended or refractory material may be oxi-
requires an inert vessel with provision for sparging the acidi-
dized under these conditions; analysts should take steps to
fied sample with 50 to 100 mL/min of carbon free gas. This
determine what recovery is being obtained. This may be done
procedure will remove essentially all IC in 2 to 10 min,
by several methods: by rerunning the sample under more
depending on design.
vigorousreactionconditionsorbyspikingsampleswithknown
refractories and determining recovery.
Instruments manufactured and marketed by Sievers Instruments, Inc., 6185
Arapahoe Ave., Suite H1, Boulder, CO 80303 have been found satisfactory. If you
are aware of alternative suppliers, please provide this information to ASTM
The boldface numbers in parentheses refer to the list of references found at the International Headquarters. Your comments will receive careful consideration at a
end of this test method. meeting of the responsible technical committee, which you may attend.
D6317 − 98 (2009)
FIG. 1 Schematic Diagram of TOC Analyzer System
TABLE 1 Blank Contribution and IC. Removal Efficiency of
7.1.3 Reactor—The sample flow is split after the addition of
Vacuum Degassing Unit.
reagents. Half of the flow passes to the delay coil while the
A A
Unit µg/L TOC µg/L IC IC level with 25 000
other half passes into the oxidation reactor. The effluent from
background background
No. µg/L input
bothstreamspassesoverindividualmembranesthatallowCO
1 3.2 8.2 55
to pas through the membrane into prepurified water for
2 3.2 22 61
3 2.4 8.0 105 detection.
4 4.2 13 89
7.1.4 Membrane—The membrane is a CO selective fluo-
5 2.8 13 30
6 3.0 8.0 70
ropolymer which is hydrophobic and non-porous. Refer to the
7 4.8 8.9 67
bibliography for additional details.
8 4.7 8.3 63
94.6 11 62 7.1.5 Detector—The CO that has passed through the mem-
10 4.7 2.9 72
brane into the purified water is measured by conductivity
A
Values are the difference between before and after addition of the degasser to a
sensors. The temperature of the conductivity cell is also
high purity (<5 µg/L) water stream.
automatically monitored so the readings can be corrected for
changes in temperature.
D6317 − 98 (2009)
7.1.6 Data Display—The conductivity detector output is 8.5 Organic Carbon, Standard Solution (1000 mg/L)—
relatedtostoredcalibrationdataandthendisplayedaspartsper Choose a water-soluble, stable reagent grade compound, such
million, (ppm = mg of carbon per litre) or parts per billion, as benzoic acid or anhydrous potassium hydrogen phthalate
(ppb = µg of carbon per L). Values are given for TC, IC, and (KHC H O ). Calculate the weight of compound required to
8 4 4
TOC by difference. make 1 L of organic carbon standard solution; for example,
KHC H O = 0.471 g of carbon per g, so one L of 1 g/L of
4 4
8. Reagents and Materials
standard requires 1/0.471, or 2.12, grams of KHP. Dissolve the
required amount of standard in some CO -free water in a 1-L
8.1 Purity of Reagents—Reagent grade chemicals shall be 2
volumetric flask, add 1 mL of sulfuric acid, and dilute to
used in all tests. Unless otherwise indicated, it is intended that
volume. Dilutions of this stock solution are to be used to
all reagents conform to the specifications of the Committee on
calibrate and test performance of the carbon analyzer.
AnalyticalReagentsoftheAmericanChemicalSociety, where
such specifications are available. Other grades may be used,
9. Sampling and Sample Preservation
provided it is first ascertained that the reagent is of sufficient
9.1 Collect the sample in accordance with Specification
purity to permit its use without lessening the accuracy of the
D1192 and Practices D3370.
determination.
9.2 Samples must be collected in contamination free bottles
8.2 Purity of Water—Unless otherwise indicated, references
sealed with a fluoropolymer lined septa. Specially cleaned (for
towatershallbeunderstoodtomeanreagentwaterconforming
TOC) 40 ml bottles are commercially available. The sample
to Type I or Type II in Specification D1193. The indicated
bottle should be rinsed several times with the sample, filled,
specification does not actually specify inorganic carbon or
and then tightly sealed.
organic carbon levels.These levels can affect the results of this
test method, especially at progressively lower levels of the
9.3 To preserve samples for this analysis, store samples in
carboncontentinthesamplestobemeasured.Whereinorganic
glass at 4 °C. To aid preservation, acidify the samples to a pH
carbon in reagent water is significant, CO -free water may be
2 of 2. It should be noted that acidification will enhance loss of
prepared from reagent water by acidifying to pH 2, then
inorganiccarbon.Ifthepurgeableorganicfractionisimportant,
sparging with fritted-glass
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.