ASTM D7644-23
(Test Method)Standard Test Method for Determination of Bromadiolone, Brodifacoum, Diphacinone and Warfarin in Water by Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS)
Standard Test Method for Determination of Bromadiolone, Brodifacoum, Diphacinone and Warfarin in Water by Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS)
SIGNIFICANCE AND USE
5.1 This test method has been developed by U.S. EPA Region 5 Chicago Regional Laboratory (CRL).
5.2 Bromadiolone, brodifacoum, diphacinone and warfarin are rodenticides for controlling mice, rats, and other rodents that pose a threat to public health, critical habitats, native plants and animals, crops, food and water supplies. These rodenticides also present human and environmental safety concerns. Warfarin and diphacinone are first-generation anticoagulants, while bromadiolone and brodifacoum are second-generation. The anticoagulants interfere with blood clotting, and death can result from excessive bleeding. The second-generation anticoagulants are especially hazardous for several reasons. They are highly toxic and persist a long time in body tissues. The second-generation anticoagulants are designed to be toxic in a single feeding, but time-to-death occurs in several days. This allows rodents to feed multiple times before death, leading to carcasses containing residues that may be many times the lethal dose.4
5.3 This test method has been investigated for use with reagent, surface, and drinking water for the selected rodenticides.
SCOPE
1.1 This test method covers the determination of bromadiolone, brodifacoum, diphacinone and warfarin (referred to collectively as rodenticides in this test method) in water by direct injection using liquid chromatography (LC) and detected with tandem mass spectrometry (MS/MS). These analytes are qualitatively and quantitatively determined by this test method. This test method adheres to multiple reaction monitoring (MRM) mass spectrometry.
1.2 The Detection Verification Level (DVL) and Reporting Range for the rodenticides are listed in Table 1.
1.2.1 The DVL is required to be at a concentration at least 3 times below the Reporting Limit (RL) and have a signal/noise ratio greater than 3:1. Fig. 1 displays the signal/noise ratios of the primary single reaction monitoring (SRM) transitions, and Fig. 2 displays the confirmatory SRM transitions at the DVLs for the rodenticides.
1.2.2 The reporting limit was calculated from the concentration of the Level 1 calibration standard, as shown in Table 4, accounting for the dilution of a 40 mL water sample up to a final volume of 50 mL with methanol to ensure analyte solubility.
1.3 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Apr-2023
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-May-2020
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Apr-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Mar-2010
- Effective Date
- 15-Jul-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-May-2007
- Effective Date
- 01-Sep-2006
Overview
ASTM D7644-23 is the internationally recognized standard test method for the determination of four major rodenticides-bromadiolone, brodifacoum, diphacinone, and warfarin-in water through liquid chromatography coupled with tandem mass spectrometry (LC/MS/MS). Established by ASTM International and further developed by the U.S. EPA Region 5 Chicago Regional Laboratory, this method provides laboratories with guidelines for qualitative and quantitative analysis to detect these toxic compounds in reagent, surface, and drinking water. The standard also sets strict parameters for calibration, sample preparation, and quality control to ensure the accuracy and reliability of results, addressing vital environmental and public health concerns.
Key Topics
- Rodenticide Monitoring: Focuses on monitoring and quantification of anticoagulant rodenticides in water matrices, including both first-generation (warfarin, diphacinone) and second-generation (bromadiolone, brodifacoum) types known for their persistence and toxicity.
- Sample Types: Applicable to reagent water, surface water, and potable water, especially important for environmental monitoring.
- Analytical Procedures: Direct injection of water samples into LC/MS/MS systems utilizing Multiple Reaction Monitoring (MRM) for enhanced specificity and sensitivity.
- Detection and Reporting: Establishes Detection Verification Levels (DVL) and reporting ranges for each analyte, ensuring results meet regulatory and safety standards. For each compound, the DVL is set at 20 ng/L with a reporting range from 125-2500 ng/L.
- Quality Assurance: Emphasizes rigorous calibration, use of surrogates, method blanks, laboratory control samples (LCS), and matrix spikes to validate data integrity.
- Safety and Environmental Practice: Includes guidance on safe laboratory handling and regulatory compliance for potentially hazardous rodenticides.
Applications
ASTM D7644-23 finds practical application in several critical areas:
- Water Quality Assessment: Used by environmental laboratories, utilities, and regulators to detect trace levels of rodenticides in natural and treated water supplies.
- Public Health Protection: Assists government agencies and health departments in monitoring water sources for rodenticide contamination, which, if undetected, poses serious risks to human health and wildlife.
- Wildlife and Habitat Conservation: Essential for monitoring environments where vulnerable animal and plant species are at risk from rodenticide exposure.
- Regulatory Compliance: Helps industries and public agencies comply with local, national, and international legislation on water safety and pesticide usage.
- Incident Response: Crucial in investigating contamination incidents or suspected poisoning related to rodenticide runoff or misuse.
Related Standards
For those implementing or referencing ASTM D7644-23, the following related ASTM and external standards are of importance:
- ASTM D1129 - Terminology Relating to Water
- ASTM D1193 - Specification for Reagent Water
- ASTM D2777 - Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
- ASTM D3694 - Practices for Preparation of Sample Containers and for Preservation of Organic Constituents
- ASTM D3856 - Guide for Management Systems in Laboratories Engaged in Analysis of Water
- ASTM D4841 - Practice for Estimation of Holding Time for Water Samples Containing Organic and Inorganic Constituents
- ASTM D5847 - Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
- ASTM E2554 - Practice for Estimating and Monitoring the Uncertainty of Test Results of a Test Method Using Control Chart Techniques
- U.S. EPA SW-846 - Test Methods for Evaluating Solid Waste, Physical/Chemical Methods
Conclusion
The ASTM D7644-23 standard is a pivotal resource for detecting and quantifying key rodenticides in water environments using state-of-the-art LC/MS/MS techniques. Its detailed protocols ensure quality assurance and regulatory compliance, supporting efforts to protect human health, water quality, and ecological systems from the risks posed by rodenticide contamination. For laboratories and agencies engaged in environmental analysis, adherence to this standard is essential for credible, reproducible monitoring results.
Buy Documents
ASTM D7644-23 - Standard Test Method for Determination of Bromadiolone, Brodifacoum, Diphacinone and Warfarin in Water by Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS)
REDLINE ASTM D7644-23 - Standard Test Method for Determination of Bromadiolone, Brodifacoum, Diphacinone and Warfarin in Water by Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS)
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D7644-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Bromadiolone, Brodifacoum, Diphacinone and Warfarin in Water by Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS)". This standard covers: SIGNIFICANCE AND USE 5.1 This test method has been developed by U.S. EPA Region 5 Chicago Regional Laboratory (CRL). 5.2 Bromadiolone, brodifacoum, diphacinone and warfarin are rodenticides for controlling mice, rats, and other rodents that pose a threat to public health, critical habitats, native plants and animals, crops, food and water supplies. These rodenticides also present human and environmental safety concerns. Warfarin and diphacinone are first-generation anticoagulants, while bromadiolone and brodifacoum are second-generation. The anticoagulants interfere with blood clotting, and death can result from excessive bleeding. The second-generation anticoagulants are especially hazardous for several reasons. They are highly toxic and persist a long time in body tissues. The second-generation anticoagulants are designed to be toxic in a single feeding, but time-to-death occurs in several days. This allows rodents to feed multiple times before death, leading to carcasses containing residues that may be many times the lethal dose.4 5.3 This test method has been investigated for use with reagent, surface, and drinking water for the selected rodenticides. SCOPE 1.1 This test method covers the determination of bromadiolone, brodifacoum, diphacinone and warfarin (referred to collectively as rodenticides in this test method) in water by direct injection using liquid chromatography (LC) and detected with tandem mass spectrometry (MS/MS). These analytes are qualitatively and quantitatively determined by this test method. This test method adheres to multiple reaction monitoring (MRM) mass spectrometry. 1.2 The Detection Verification Level (DVL) and Reporting Range for the rodenticides are listed in Table 1. 1.2.1 The DVL is required to be at a concentration at least 3 times below the Reporting Limit (RL) and have a signal/noise ratio greater than 3:1. Fig. 1 displays the signal/noise ratios of the primary single reaction monitoring (SRM) transitions, and Fig. 2 displays the confirmatory SRM transitions at the DVLs for the rodenticides. 1.2.2 The reporting limit was calculated from the concentration of the Level 1 calibration standard, as shown in Table 4, accounting for the dilution of a 40 mL water sample up to a final volume of 50 mL with methanol to ensure analyte solubility. 1.3 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method has been developed by U.S. EPA Region 5 Chicago Regional Laboratory (CRL). 5.2 Bromadiolone, brodifacoum, diphacinone and warfarin are rodenticides for controlling mice, rats, and other rodents that pose a threat to public health, critical habitats, native plants and animals, crops, food and water supplies. These rodenticides also present human and environmental safety concerns. Warfarin and diphacinone are first-generation anticoagulants, while bromadiolone and brodifacoum are second-generation. The anticoagulants interfere with blood clotting, and death can result from excessive bleeding. The second-generation anticoagulants are especially hazardous for several reasons. They are highly toxic and persist a long time in body tissues. The second-generation anticoagulants are designed to be toxic in a single feeding, but time-to-death occurs in several days. This allows rodents to feed multiple times before death, leading to carcasses containing residues that may be many times the lethal dose.4 5.3 This test method has been investigated for use with reagent, surface, and drinking water for the selected rodenticides. SCOPE 1.1 This test method covers the determination of bromadiolone, brodifacoum, diphacinone and warfarin (referred to collectively as rodenticides in this test method) in water by direct injection using liquid chromatography (LC) and detected with tandem mass spectrometry (MS/MS). These analytes are qualitatively and quantitatively determined by this test method. This test method adheres to multiple reaction monitoring (MRM) mass spectrometry. 1.2 The Detection Verification Level (DVL) and Reporting Range for the rodenticides are listed in Table 1. 1.2.1 The DVL is required to be at a concentration at least 3 times below the Reporting Limit (RL) and have a signal/noise ratio greater than 3:1. Fig. 1 displays the signal/noise ratios of the primary single reaction monitoring (SRM) transitions, and Fig. 2 displays the confirmatory SRM transitions at the DVLs for the rodenticides. 1.2.2 The reporting limit was calculated from the concentration of the Level 1 calibration standard, as shown in Table 4, accounting for the dilution of a 40 mL water sample up to a final volume of 50 mL with methanol to ensure analyte solubility. 1.3 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7644-23 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7644-23 has the following relationships with other standards: It is inter standard links to ASTM D3694-96(2024), ASTM D1129-13(2020)e2, ASTM E2554-18e1, ASTM E2554-18, ASTM E2554-13, ASTM D4841-88(2013)e1, ASTM D4841-88(2013), ASTM D2777-12, ASTM D3856-11, ASTM D3694-96(2011), ASTM D1129-10, ASTM D4841-88(2008), ASTM D2777-08, ASTM E2554-07, ASTM D1129-06ae1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7644-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7644 − 23
Standard Test Method for
Determination of Bromadiolone, Brodifacoum, Diphacinone
and Warfarin in Water by Liquid Chromatography/Tandem
Mass Spectrometry (LC/MS/MS)
This standard is issued under the fixed designation D7644; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.1 This test method covers the determination of
Barriers to Trade (TBT) Committee.
bromadiolone, brodifacoum, diphacinone and warfarin (re-
ferred to collectively as rodenticides in this test method) in
2. Referenced Documents
water by direct injection using liquid chromatography (LC) and
detected with tandem mass spectrometry (MS/MS). These 2.1 ASTM Standards:
D1129 Terminology Relating to Water
analytes are qualitatively and quantitatively determined by this
D1193 Specification for Reagent Water
test method. This test method adheres to multiple reaction
D2777 Practice for Determination of Precision and Bias of
monitoring (MRM) mass spectrometry.
Applicable Test Methods of Committee D19 on Water
1.2 The Detection Verification Level (DVL) and Reporting
D3694 Practices for Preparation of Sample Containers and
Range for the rodenticides are listed in Table 1.
for Preservation of Organic Constituents
1.2.1 The DVL is required to be at a concentration at least
D3856 Guide for Management Systems in Laboratories
3 times below the Reporting Limit (RL) and have a signal/
Engaged in Analysis of Water
noise ratio greater than 3:1. Fig. 1 displays the signal/noise
D4841 Practice for Estimation of Holding Time for Water
ratios of the primary single reaction monitoring (SRM)
Samples Containing Organic and Inorganic Constituents
transitions, and Fig. 2 displays the confirmatory SRM transi-
D5847 Practice for Writing Quality Control Specifications
tions at the DVLs for the rodenticides.
for Standard Test Methods for Water Analysis
1.2.2 The reporting limit was calculated from the concen-
E2554 Practice for Estimating and Monitoring the Uncer-
tration of the Level 1 calibration standard, as shown in Table 4,
tainty of Test Results of a Test Method Using Control
accounting for the dilution of a 40 mL water sample up to a
Chart Techniques
final volume of 50 mL with methanol to ensure analyte
2.2 Other Documents:
solubility.
U.S. EPA publication SW-846 Test Methods for Evaluating
1.3 Units—The values stated in SI units are to be regarded
Solid Waste, Physical/Chemical Methods
as standard. No other units of measurement are included in this
standard.
3. Terminology
1.4 This standard does not purport to address all of the
3.1 Definitions:
safety concerns, if any, associated with its use. It is the
3.1.1 For definitions of terms used in this standard, refer to
responsibility of the user of this standard to establish appro-
Terminology D1129.
priate safety, health, and environmental practices and deter-
3.2 Definitions of Terms Specific to This Standard:
mine the applicability of regulatory limitations prior to use.
3.2.1 detection verification level, DVL, n—a concentration
1.5 This international standard was developed in accor-
that has a signal/noise (S/N) ratio greater than 3:1 and is at least
dance with internationally recognized principles on standard-
3 times below the Reporting Limit (RL).
ization established in the Decision on Principles for the
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This test method is under the jurisdiction of ASTM Committee D19 on Water contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
and is the direct responsibility of Subcommittee D19.06 on Methods for Analysis for Standards volume information, refer to the standard’s Document Summary page on
Organic Substances in Water. the ASTM website.
Current edition approved April 15, 2023. Published June 2023. Originally Available from National Technical Information Service (NTIS), U.S. Depart-
approved in 2010. Last previous edition approved in 2016 as D7644 – 16. DOI: ment of Commerce, 5285 Port Royal Road, Springfield, VA, 22161 or at http://
10.1520/D7644-23. www.epa.gov/epawaste/hazard/testmethods/index.htm.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7644 − 23
TABLE 1 Detection Verification Level and Reporting Range
4. Summary of Test Method
DVL Reporting Range
Analyte 4.1 This is a performance based method, and modifications
(ng/L) (ng/L)
are allowed to improve performance.
Bromadiolone 20 125-2500
Brodifacoum 20 125-2500
4.2 For rodenticide analysis, samples are shipped to the lab
Diphacinone 20 125-2500
between above freezing and 6 °C and analyzed within 14 days
Warfarin 20 125-2500
of collection. In the lab, the samples are spiked with surrogates,
quantitatively transferred to a graduated cylinder using three
methanol rinses, filtered using a syringe driven filter unit, and
analyzed directly by LC/MS/MS.
3.2.2 independent reference material, IRM, n—a material of
4.3 Bromadiolone, brodifacoum, diphacinone, warfarin,
known purity and concentration obtained either from the
warfarin-D (surrogate) and 2-bromo-4-(1,1,3,3-
National Institute of Standards and Technology (NIST) or other
tetramethylbutyl)phenol (brominated octylphenol, Br-OP, sur-
reputable supplier; the IRM must be obtained from a different
rogate) are identified by retention time and two SRM transi-
lot of material than is used for calibration. tions. The target analytes and surrogates are quantitated using
the primary SRM transitions utilizing an external calibration.
3.2.3 reporting limit, RL, n—the concentration of the
The final report issued for each sample lists the concentration
lowest-level calibration standard used for quantification ac-
of bromadiolone, brodifacoum, diphacinone, warfarin, and
counting for the sample dilution.
surrogate recoveries.
3.2.3.1 Discussion—In this test method, a 40 mL sample
aliquot is diluted to a 50 mL final volume after thoroughly
5. Significance and Use
rinsing the collection vial with methanol for quantitative
transfer. In this case, the lowest calibration level of 100 ppt
5.1 This test method has been developed by U.S. EPA
would allow a reporting limit of 125 ppt to be achieved.
Region 5 Chicago Regional Laboratory (CRL).
3.2.4 rodenticides, n—in this test method, bromadiolone,
5.2 Bromadiolone, brodifacoum, diphacinone and warfarin
brodifacoum, diphacinone, and warfarin collectively.
are rodenticides for controlling mice, rats, and other rodents
that pose a threat to public health, critical habitats, native plants
3.3 Acronyms:
and animals, crops, food and water supplies. These rodenti-
3.3.1 CCC, n—Continuing Calibration Check
cides also present human and environmental safety concerns.
3.3.2 IC, n—Initial Calibration
Warfarin and diphacinone are first-generation anticoagulants,
3.3.3 LC, n—Liquid Chromatography
while bromadiolone and brodifacoum are second-generation.
The anticoagulants interfere with blood clotting, and death can
3.3.4 LCS/LCSD, n—Laboratory Control Sample/
result from excessive bleeding. The second-generation antico-
Laboratory Control Sample Duplicate
agulants are especially hazardous for several reasons. They are
3.3.5 MeOH, n—Methanol
highly toxic and persist a long time in body tissues. The
-3
3.3.6 mM, n—millimolar, 1 × 10 moles/L
second-generation anticoagulants are designed to be toxic in a
single feeding, but time-to-death occurs in several days. This
3.3.7 MRM, n—Multiple Reaction Monitoring
allows rodents to feed multiple times before death, leading to
3.3.8 MS/MSD, n—Matrix Spike/Matrix Spike Duplicate
carcasses containing residues that may be many times the lethal
3.3.9 NA, adj—Not Available
dose.
3.3.10 ND, n—non-detect
5.3 This test method has been investigated for use with
3.3.11 P&A, n—Precision and Accuracy
reagent, surface, and drinking water for the selected rodenti-
cides.
3.3.12 PPB, n—parts per billion
3.3.13 PPT, n—parts per trillion
6. Interferences
3.3.14 QA, adj—Quality Assurance
6.1 Method interferences may be caused by contaminants in
3.3.15 QC, adj—Quality Control
solvents, reagents, glassware and other apparatus producing
3.3.16 RL, n—Reporting Limit
discrete artifacts or elevated baselines. All of these materials
are demonstrated to be free from interferences by analyzing
3.3.17 RSD, n—Relative Standard Deviation
laboratory reagent blanks under the same conditions as
3.3.18 RT, n—Retention Time
samples.
3.3.19 SDS, n—Safety Data Sheets
6.2 All glassware is washed in hot water with detergent and
3.3.20 SRM, n—Single Reaction Monitoring
rinsed in hot water followed by distilled water. The glassware
is then dried and heated in an oven at 250 °C for 15 min to
3.3.21 SS, n—Surrogate Standard
3.3.22 TC, n—Target Compound
-6
3.3.23 μM, n—micromolar, 1 × 10 moles/L
Additional information about rodenticides is available from United States
3.3.24 VOA, n—Volatile Organic Analysis Environmental Protection Agency (EPA), http://www.epa.gov.
D7644 − 23
FIG. 1 Example Primary SRM Chromatograms Signal/Noise Ratios
FIG. 2 Example Confirmatory SRM Chromatograms Signal/Noise Ratios
D7644 − 23
30 min. All glassware is subsequently cleaned with acetone American Chemical Society. Other reagent grades may be
followed by methanol. used provided they are first determined to be of sufficiently
high purity to permit their use without affecting the accuracy of
6.3 All reagents and solvents should be of pesticide residue
the measurements.
purity or higher to minimize interference problems.
8.2 Purity of Water—Unless otherwise indicated, references
6.4 Matrix interferences may be caused by contaminants in
to water must be understood to mean reagent water conforming
the sample. The extent of matrix interferences can vary
to Type 1 of Specification D1193. It must be demonstrated that
considerably from sample source depending on variations of
this water does not contain contaminants at concentrations
the sample matrix.
sufficient to interfere with the analysis.
8.3 Gases—Ultrapure nitrogen and argon.
7. Apparatus
8.4 Methanol (CAS # 67-56-1).
7.1 LC/MS/MS System:
8.5 Acetonitrile (CAS # 75-05-8).
7.1.1 Liquid Chromatography (LC) System—A complete LC
system is needed to analyze samples. This should include a
8.6 Acetone (CAS # 67-64-1).
sample injection system, a solvent pumping system capable of
8.7 Ammonium Hydroxide (Concentrated, CAS # 1336-21-
mixing solvents, a sample compartment capable of maintaining
6).
required temperature and a temperature controlled column
8.8 Ascorbic Acid (CAS # 50-81-7).
compartment. A system that is capable of performing at the
flows, pressures, controlled temperatures, sample volumes, and
8.9 Bromadiolone (CAS # 28772-56-7).
requirements of the standard may be used.
8.10 Brodifacoum (CAS # 56073-10-0).
7.1.2 Analytical Column —A C18 column was used to
8.11 Diphacinone (CAS # 82-66-6).
develop this test method.
8.12 Warfarin (CAS # 81-81-2).
NOTE 1—Any column that can achieve baseline resolution of these
analytes may be used. Baseline resolution simplifies data analysis and can 8.13 Warfarin-D (Phenyl-D , CAS # (unlabeled) 81-81-2).
5 5
reduce the chance of ion suppression, leading to higher limits of detection.
8.13.1 Discussion—Warfarin-D is used as the electrospray
positive analyte surrogate in this standard.
7.1.3 Tandem Mass Spectrometer (MS/MS) System—A
MS/MS system capable of MRM analysis. Any system that is
8.14 2-Bromo-4-(1,1,3,3-tetramethylbutyl)phenol (Br-OP).
capable of performing at the requirements in this standard may
8.14.1 Discussion—Br-OP is used as the electrospray nega-
be used.
tive analyte surrogate in this standard.
7.2 Filtration Device:
9. Hazards
7.2.1 Hypodermic syringe—A Lock Tip Glass Syringe ca-
9.1 Normal laboratory safety applies to this method. Ana-
pable of holding a syringe-driven filter unit or similar may be
lysts should wear safety glasses, gloves, and lab coats when
used.
working in the lab. Analysts should review the Safety Data
7.2.1.1 A 50-mL lock tip glass syringe size is recommended
Sheets (SDS) for all reagents used in this test method.
since a 50-mL sample size is used in this test method.
7.2.2 Filter Unit —PVDF filter units were used to filter the 10. Sampling
samples.
10.1 Sampling—Grab samples must be collected in 40 mL
pre-cleaned amber glass vials with inert-lined caps demon-
8. Reagents and Materials
strated to be free of interferences. Surface water samples are
collected unpreserved, shipped between above freezing and
8.1 Purity of Reagents—High Performance Liquid Chroma-
6 °C, and stored in the laboratory between above freezing and
tography (HPLC) pesticide residue analysis and spectropho-
6 °C. Chlorinated drinking water samples are dechlorinated
tometry grade chemicals must be used in all tests. Unless
indicated otherwise, it is intended that all reagents must
conform to the Committee on Analytical Reagents of the
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by the American Chemical
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
A Waters ACQUITY UPLC (a trademark of the Waters Corporation, Milford, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
MA) BEH C18, 2.1 × 100 mm, 1.7 μm particle size was used, if you are aware of copeial Convention, Inc. (USPC), Rockville, MD.
an alternative column that meets the performance of the standard, please provide this A source of Warfarin-D is Cambridge Isotope Laboratories, 50 Frontage Road,
information to ASTM International Headquarters. Your comments will receive Andover, MA 01810-5413. If you are aware of an alternative source that meets the
careful consideration at a meeting of the responsible technical committee, which performance of the standard, please provide this information to ASTM International
you may attend. Headquarters. Your comments will receive careful consideration at a meeting of the
6 1
A Millex HV Syringe Driven Filter Unit PVDF 0.22 μm (Millipore responsible technical committee, which you may attend.
Corporation, Catalog #SLGV033NS; Millex is a trademark of Merck KGAA, A source of Br-OP is Accustandard, Inc., 125 Market Street, New Haven CT
Darmstadt, Germany) was used, if you are aware of an alternative filter unit that 06513. If you are aware of an alternative source that meets the performance of the
meets the performance of the standard, please provide this information to ASTM standard, please provide this information to ASTM International Headquarters. Your
International Headquarters. Your comments will receive careful consideration at a comments will receive careful consideration at a meeting of the responsible
1 1
meeting of the responsible technical committee, which you may attend. technical committee, which you may attend.
D7644 − 23
TABLE 2 Gradient Conditions for Liquid Chromatography
with ascorbic acid; 10 mg of ascorbic acid is added to each
40 mL vial prior to water collection. This test method requires Percent Percent
Time Flow 95 % Water/ 95 % Methanol/
a 40 mL sample size per analysis. Conventional sampling
(min) (μL/min) 5 % Methanol, 5 % Water,
practices should be followed. Refer to Guide D3856 and
5 mM NH OH 5 mM NH OH
4 4
Practices D3694.
0.0 300 100 0
2.0 300 100 0
10.1.1 Ammonium acetate was evaluated as an agent to
6.0 300 20 80
bind free chlorine in drinking water and was found to be
6.1 200 5 95
ineffective in the preservation of the rodenticides in chlorinated
7.0 200 5 95
drinking water. Ascorbic acid was effective as a dechlorinating 8.5 200 0 100
13.0 300 0 100
agent in chlorine fortified Chicago tap water, which contained
14.0 300 100 0
3.2 ppm free chlorine and was dechlorinated with 10 mg
16.0 300 100 0
ascorbic acid per 40 mL water sample.
10.2 The samples are collected using 40 mL glass vials. A
shown to carry-over when acetonitrile was used for this
40 mL volume is collected directly into the sample collection
analysis. The use of methanol corrected this problem while
vial without using any other measuring devices. This is a
providing separation and sensitivity. Instrument manufacturer
requirement due to the rodenticides’ affinity for surfaces, which
specifications should be followed in order to eliminate sample
will lead to biased low results if transferring between contain-
carry-over.
ers. Before collection, the vials must be evaluated to determine
11.2.2 Temperatures—Column, 30 °C; Sample
a 40 mL sample volume. For example, the vials used in this test
compartment, 15 °C.
method were calibrated before use to determine that filling the
11.2.3 Seal Wash—Solvent: 50 % Acetonitrile/50 % Water;
vial to approximately 1.6 cm below the rim would result in a
Time: 5 min.
40 mL sample volume. The greatest amount of water held by
the 40 mL vials used in this test method was approximately 42 11.3 Mass Spectrometer Parameters:
mL. Vials filled to 42 mL in the field would not allow the
11.3.1 To acquire the maximum number of data points per
laboratory to spike the samples before quantitatively transfer- SRM channel while maintaining adequate sensitivity, the tune
ring to the 50 mL graduated cylinder. It is imperative that the
parameters may be optimized according to the instrument.
samplers do not overfill the vials. Each peak requires at least 10 scans per peak for adequate
quantitation. This standard contains two surrogates and four
10.3 Preservation—Store samples between above freezing
target compounds that can be acquired in 5 MRM acquisition
and 6 °C from the time of collection until analysis. Analyze the
functions. Variable parameters regarding retention times, SRM
sample within 14 days of collection. Chlorinated drinking
transitions, and cone and collision energies are shown in Table
water samples are dechlorinated with ascorbic acid; 10 mg of
3. Mass spectrometer parameters used in the development of
ascorbic acid is added to each 40 mL vial prior to water
this test method are listed below:
collection.
The instrument is set in the Electrospray source setting.
Capillary Voltage: 3.5 kV
11. Preparation of LC/MS/MS
Cone: Variable depending on analyte (Table 3)
Extractor: 2 Volts
11.1 LC Chromatograph Operating Conditions:
RF Lens: 0.1 Volts
11.1.1 Injection volumes of all calibration standards and
Source Temperature: 120 °C
samples are made at 50 μL volume using a full loop injection.
Desolvation Temperature: 375 °C
Desolvation Gas Flow: 800 L/h
If a 50 μL volume loop is installed in the LC, a “full loop”
Cone Gas Flow: 25 L/h
mode is the preferred technique when performing fast, quali-
Low Mass Resolution 1: 14.0
tative analyses. This mode should be used whenever accuracy
High Mass Resolution 1: 14.0
Ion Energy 1: 0.5
and precision are the primary concerns. The first sample
Entrance Energy: -1
analyzed after the calibration curve is a blank to ensure there is
Collision Energy: Variable depending on analyte (Table 3)
no carry-over. The gradient conditions for the liquid chromato-
Exit Energy: 0
Low Mass Resolution 2: 14.0
graph are shown in Table 2.
High Mass resolution 2: 14.0
Ion Energy 2: 0.7
NOTE 2—If the instrument does not have a 50 μL injection capability a
Multiplier: 650
different volume may be used. This is a performance-based method and
-3
Gas Cell Pirani Gauge: 7.0 × 10 Torr
modifications are allowed as long as minimum performance criteria are
Inter-Channel Delay: 0.02 s
met.
Inter-Scan Delay: 0.02 s
Dwell: 0.1 s
11.2 LC Sample Manager Conditions:
11.2.1 Wash Solvents—Weak wash is 4.0 mL of 95 %
12. Calibration and Standardization
water/5 % methanol. Strong wash is 2.0 mL of methanol. The
12.1 The mass spectrometer must be calibrated per manu-
strong wash solvent is needed to eliminate carry-over between
facturer specifications before analysis. In order to obtain valid
injections of rodenticide samples. The weak wash is used to
and accurate analytical values through this test method within
remove the strong wash solvent. These rodenticides were
the confidence limits, the following procedures must be fol-
lowed when performing the test method. Prepare all solutions
A Pocket Chlorine meter was used to measure free chlorine. in the lab using Class A volumetric glassware.
D7644 − 23
TABLE 3 Retention Times, SRM Ions, and Analyte-Specific Mass Spectrometer Parameters
SRM Primary/
Primary/ Retention Time Cone Voltage Collision Energy
Analyte ESI Mass Transition Confirmatory
Confirmatory (min) (Volts) (eV)
(Parent > Product) SRM Area Ratio
Primary 7.45 and 30 22 509.1 > 251.1
Bromadiolone Positive 1.0
Confirmatory 7.59 30 22 511.1 > 251.1
Primary 42 20 523.1 > 335.1
Brodifacoum Positive 7.95 1.1
Confirmatory 42 33 523.1 > 178
Primary 32 25 339.1 > 166.8
Diphacinone Negative 6.36 3.0
Confirmatory 32 47 339.1 > 115.8
Primary 26 14 309.1 > 162.8
Warfarin Positive 5.38 1.7
Confirmatory 26 20 309.1 > 251.1
Warfarin-D Primary 26 14 314.2 > 162.8
Positive 5.37 1.7
(Surrogate) Confirmatory 26 19 314.2 > 256.1
Br-OP
Negative Primary 8.92 35 25 283.1 > 78.6 N/A
(Surrogate)
12.2 Calibration and Standardization—To calibrate the 12.2.3 Inject each standard and obtain its chromatogram. An
instrument, analyze 7 calibration standards containing the 7 external calibration technique is used monitoring the primary
concentration levels of the rodenticides and surrogates prior to and confirmatory SRM transition of each analyte. Calibration
analysis as shown in Table 4. A calibration stock standard software is utilized to conduct the quantitation of the target
solution is prepared from standard materials or they are analytes and surrogates using the primary SRM transition. The
purchased as certified solutions. Stock standard solution A ratios of the primary/confirmatory SRM transition area counts
containing bromadiolone, brodifacoum, diphacinone, warfarin, are given in Table 3 and will vary depending on the individual
warfarin-D (surrogate), and 2-Bromo-4-(1,1,3,3- tuning conditions. The primary/confirmatory SRM transition
tetramethylbutyl)phenol (surrogate) is prepared at Level 7 area ratio must be within 35 % of the individual labs accepted
concentration, and aliquots of that solution are diluted to primary/confirmatory SRM transition area ratio. The primary
prepare Levels 1 through 6. The following steps will produce SRM transition of each analyte is used for quantitation and the
standards with the concentration values shown in Table 4. The confirmatory SRM transition for confirmation. This gives
analyst is responsible for recording initial component weights additional confirmation by isolating the parent ion, forming
carefully when working with pure materials and correctly two product ions via fragmentation, and relating it to the
carrying the weights through the dilution calculations. retention time in the calibration standard.
12.2.1 Prepare stock standard solution A (Level 7) by 12.2.4 The calibration software manual should be consulted
adding to a 100 mL volumetric flask individual methanol to properly use the software. The quantitation method is set as
solutions of the following: 50 μL of bromadiolone, an external calibration using the peak areas in ppt or ppb units,
brodifacoum, diphacinone, warfarin, warfarin-D (surrogate) as long as the analyst is consistent. Concentrations may be
each at 4.0 mg/L and 50 μL of 2-Bromo-4-(1,1,3,3- calculated using the data system software to generate linear
tetramethylbutyl)phenol (surrogate) at 0.2 g/L, dilute to 100
regression or quadratic calibration curves. Forcing the calibra-
mL with 80 % water/20 % methanol. The preparation of the tion curve through the origin is not recommended. Curves
Level 7 standard can be accomplished using different volumes
should be evaluated using relative error or relative standard
and concentrations of stock solutions as is accustomed in the error.
individual laboratory. Depending on the prepared stock
12.2.5 Linear calibration may be used if the point of origin
concentrations, solubility at that concentration will have to be is excluded and a fit weighting of 1/X is used in order to give
ensured.
more emphasis to the lower concentrations. Each calibration
12.2.2 Aliquots of solution A are then diluted with 80 % point used to generate the curve must have a calculated percent
water/20 % methanol to prepare the desired calibration levels
deviation less than 25 % from the generated curve.
in 2 mL amber glass LC vials. The calibration vials must be
used within 24 h to ensure optimum results. Stock calibration
standards are routinely replaced every 7 days if not previously
Management and Technical Requirements for Laboratories Performing Envi-
discarded for quality control failure. Calibration standards are
ronmental Analysis; Module 4: Quality Systems for Chemical Testing; The NELAC
not filtered. Institute, 2017.
TABLE 4 Concentrations of Calibration Standards (ppt)
Analyte/Surrogate LV 1 LV 2 LV 3 LV 4 LV 5 LV 6 LV 7
Bromadiolone 100 200 500 750 1000 1500 2000
Brodifacoum 100 200 500 750 1000 1500 2000
Diphacinone 100 200 500 750 1000 1500 2000
Warfarin 100 200 500 750 1000 1500 2000
Warfarin-D (Surrogate) 100 200 500 750 1000 1500 2000
Br-OP (Surrogate) 5000 10 000 25 000 37 500 50 000 75 000 100 000
D7644 − 23
12.2.6 Quadratic calibration may be used if the point of similar to the solution used in this test method. Each replicate
origin is excluded, and a fit weighting of 1/X is used in order must be taken through the complete analytical test method
to give more emphasis to the lower concentrations. Each including any sample preservation and pretreatment steps.
calibration point used to generate the curve must have a
12.3.2 Calculate the mean (average) percent recovery and
calculated percent deviation less than 25 % from the generated
relative standard deviation (RSD) of the four values and
curve.
compare to the acceptable ranges of the QC acceptance criteria
12.2.7 The retention time window of the SRM transitions
for the Initial Demonstration of Performance in Table 5.
must be within 5 % of the retention time of the analyte in a
12.3.3 This study should be repeated until the single opera-
midpoint calibration standard. If this is not the case, re-analyze
tor precision and mean recovery are within the limits in Table
the calibration curve to determine if there was a shift in
5. If a concentration other than the recommended concentration
retention time during the analysis, and the sample needs to be
is used, refer to Practice D5847 for information on applying the
re-injected. If the retention time is still incorrect in the sample,
F-test and t-test in evaluating the acceptability of the mean and
refer to the analyte as an unknown.
standard deviation.
12.2.8 A midpoint calibration check standard must be ana-
12.3.3.1 The QC acceptance criteria for the Initial Demon-
lyzed at the end of each batch of 20 samples or within 24 h
stration of Performance in Table 5 were generated from a
after the initial calibration curve was generated. This end
single-laboratory. Data from reagent, surface, and drinking
calibration check should be the same calibration standard that
water matrices are shown in the Precision and Bias Section 16.
was used to generate the initial curve. The results from the end
It is recommended that the laboratory generate their own
calibration check standard must have a percent deviation less
in-house QC acceptance criteria which meets or exceeds the
than 30 % from the calculated concentration for the target
criteria in this standard. References on how to generate QC
analytes and surrogates. If the results are not within these
acceptance criteria are Practices D2777, D5847, E2554, or
criteria, the problem must be corrected, and either all samples
Method 8000B in U.S. EPA publication SW-846.
in the batch must be re-analyzed against a new calibration
12.4 Surrogate Spiking Solution:
curve or the affected results must be qualified with an indica-
12.4.1 A surrogate standard solution containing warfarin-D
tion that they do not fall within the performance criteria of the 5
and Br-OP is added to all samples. A stock surrogate spiking
test method. If the analyst inspects the vial containing the end
solution is prepared in methanol at 800 ppb for warfarin-D
calibration check standard and notices that the sample evapo- 5
and 40 ppm for Br-OP. Spiking 50 μL of this spiking solution
rated affecting the concentration, a new end calibration check
into a 40 mL water sample results in a concentration of
standard may be made and analyzed. If this new end calibration
1000 ppt for warfarin-D and 50 000 ppt for Br-OP in the
check standard has a percent deviation less than 30 % from the 5
sample. The result obtained for the surrogate recovery must fall
calculated concentration for the target analytes and surrogates,
within the limits of Table 5. If the limits are not met, the
the results may be reported unqualified.
affected results must be qualified with an indication that they
12.3 If a laboratory has not performed the test before or if
do not fall within the performance criteria of the test method.
there has been a major change in the measurement system, for
Surrogate spiking solutions are routinely replaced every 30
example, new analyst, new instrument, etc., a precision and
days if not previously discarded for quality control failure.
bias study must be performed to demonstrate laboratory
12.5 Method Blank:
capability.
12.3.1 Analyze at least four replicates of a sample solution 12.5.1 Analyze a reagent water blank with each batch of 20
containing bromadiolone, brodifacoum, diphacinone, warfarin, or fewer samples. The concentration of the rodenticides found
warfarin-D (surrogate) and Br-OP (surrogate) at a concentra- in the blank must be below the DVL. If the concentrations of
tion
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7644 − 16 D7644 − 23
Standard Test Method for
Determination of Bromadiolone, Brodifacoum, Diphacinone
and Warfarin in Water by Liquid Chromatography/Tandem
Mass Spectrometry (LC/MS/MS)
This standard is issued under the fixed designation D7644; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This procedure test method covers the determination of bromadiolone, brodifacoum, diphacinone and warfarin (referred to
collectively as rodenticides in this test method) in water by direct injection using liquid chromatography (LC) and detected with
tandem mass spectrometry (MS/MS). These analytes are qualitatively and quantitatively determined by this test method. This test
method adheres to multiple reaction monitoring (MRM) mass spectrometry.
1.2 The Detection Verification Level (DVL) and Reporting Range for the rodenticides are listed in Table 1.
1.2.1 The DVL is required to be at a concentration at least 3 times below the Reporting Limit (RL) and have a signal/noise ratio
greater than 3:1. Fig. 1 displays the signal/noise ratios of the primary single reaction monitoring (SRM) transitions, and Fig. 2
displays the confirmatory SRM transitions at the DVLs for the rodenticides.
1.2.2 The reporting limit was calculated from the concentration of the Level 1 calibration standard, as shown in Table 4,
accounting for the dilution of a 40 mL water sample up to a final volume of 50 mL with methanol to ensure analyte solubility.
1.3 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this
standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.06 on Methods for Analysis for
Organic Substances in Water.
Current edition approved Feb. 1, 2016April 15, 2023. Published May 2016June 2023. Originally approved in 2010. Last previous edition approved in 20102016 as
ɛ2
D7644D7644 – 16. – 10 . DOI: 10.1520/D7644-16.10.1520/D7644-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7644 − 23
TABLE 1 Detection Verification Level and Reporting Range
DVL Reporting Range
Analyte
(ng/L) (ng/L)
Bromadiolone 20 125-2500
Brodifacoum 20 125-2500
Diphacinone 20 125-2500
Warfarin 20 125-2500
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3694 Practices for Preparation of Sample Containers and for Preservation of Organic Constituents
D3856 Guide for Management Systems in Laboratories Engaged in Analysis of Water
D4841 Practice for Estimation of Holding Time for Water Samples Containing Organic and Inorganic Constituents
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
E2554 Practice for Estimating and Monitoring the Uncertainty of Test Results of a Test Method Using Control Chart Techniques
2.2 Other Documents:
U.S. EPA publication SW-846 Test Methods for Evaluating Solid Waste, Physical/Chemical Methods
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 detection verification level, DVL, n—a concentration that has a signal/noise (S/N) ratio greater than 3:1 and is at least 3 times
below the Reporting Limit (RL).
3.2.2 independent reference material, IRM, n—a material of known purity and concentration obtained either from the National
Institute of Standards and Technology (NIST) or other reputable supplier. The IRM shallsupplier; the IRM must be obtained from
a different lot of material than is used for calibration.
3.2.3 reporting limit, RL, n—the concentration of the lowest-level calibration standard used for quantification accounting for the
sample dilution.
3.2.3.1 Discussion—
In this test method, a 40 mL sample aliquot is diluted to a 50 mL final volume after thoroughly rinsing the collection vial with
methanol for quantitative transfer. In this case, the lowest calibration level of 100 ppt would allow a reporting limit of 125 ppt to
be achieved.
3.2.4 rodenticides, n—in this test method, bromadiolone, brodifacoum, diphacinone, and warfarin collectively.
3.3 Acronyms:
3.3.1 CCC, n—Continuing Calibration Check
3.3.2 IC, n—Initial Calibration
3.3.3 LC, n—Liquid Chromatography
3.3.4 LCS/LCSD, n—Laboratory Control Sample/Laboratory Control Sample Duplicate
3.3.5 MeOH, n—Methanol
-3
3.3.6 mM, n—millimolar, 1 × 10 moles/L
3.3.7 MRM, n—Multiple Reaction Monitoring
Available from National Technical Information Service (NTIS), U.S. Department of Commerce, 5285 Port Royal Road, Springfield, VA, 22161 or at http://www.epa.gov/
epawaste/hazard/testmethods/index.htm.
D7644 − 23
FIG. 1 Example Primary SRM Chromatograms Signal/Noise Ratios
FIG. 2 Example Confirmatory SRM Chromatograms Signal/Noise Ratios
D7644 − 23
3.3.8 MS/MSD, n—Matrix Spike/Matrix Spike Duplicate
3.3.9 NA, adj—Not Available
3.3.10 ND, n—non-detect
3.3.11 P&A, n—Precision and Accuracy
3.3.12 PPB, n—parts per billion
3.3.13 PPT, n—parts per trillion
3.3.14 QA, adj—Quality Assurance
3.3.15 QC, adj—Quality Control
3.3.16 RL, n—Reporting Limit
3.3.17 RSD, n—Relative Standard Deviation
3.3.18 RT, n—Retention Time
3.3.19 SDS, n—Safety Data Sheets
3.3.20 SRM, n—Single Reaction Monitoring
3.3.21 SS, n—Surrogate Standard
3.3.22 TC, n—Target Compound
-6
3.3.23 μM, n—micromolar, 1 × 10 moles/L
3.3.24 VOA, n—Volatile Organic Analysis
4. Summary of Test Method
4.1 This is a performance based method, and modifications are allowed to improve performance.
4.2 For rodenticide analysis, samples are shipped to the lab between 0°C and 6°Cabove freezing and 6 °C and analyzed within
14 days of collection. In the lab, the samples are spiked with surrogates, quantitatively transferred to a graduated cylinder using
three methanol rinses, filtered using a syringe driven filter unit, and analyzed directly by LC/MS/MS.
4.3 Bromadiolone, brodifacoum, diphacinone, warfarin, warfarin-D (surrogate) and 2-bromo-4-(1,1,3,3-tetramethylbutyl)phenol
(brominated octylphenol, Br-OP, surrogate) are identified by retention time and two SRM transitions. The target analytes and
surrogates are quantitated using the primary SRM transitions utilizing an external calibration. The final report issued for each
sample lists the concentration of bromadiolone, brodifacoum, diphacinone, warfarin, and surrogate recoveries.
5. Significance and Use
5.1 This test method has been developed by U.S. EPA Region 5 Chicago Regional Laboratory (CRL).
5.2 Bromadiolone, brodifacoum, diphacinone and warfarin are rodenticides for controlling mice, rats, and other rodents that pose
D7644 − 23
a threat to public health, critical habitats, native plants and animals, crops, food and water supplies. These rodenticides also present
human and environmental safety concerns. Warfarin and diphacinone are first-generation anticoagulants, while bromadiolone and
brodifacoum are second-generation. The anticoagulants interfere with blood clotting, and death can result from excessive bleeding.
The second-generation anticoagulants are especially hazardous for several reasons. They are highly toxic and persist a long time
in body tissues. The second-generation anticoagulants are designed to be toxic in a single feeding, but time-to-death occurs in
several days. This allows rodents to feed multiple times before death, leading to carcasses containing residues that may be many
times the lethal dose.
5.3 This test method has been investigated for use with reagent, surface, and drinking water for the selected rodenticides.
6. Interferences
6.1 Method interferences may be caused by contaminants in solvents, reagents, glassware and other apparatus producing discrete
artifacts or elevated baselines. All of these materials are demonstrated to be free from interferences by analyzing laboratory reagent
blanks under the same conditions as samples.
6.2 All glassware is washed in hot water with detergent and rinsed in hot water followed by distilled water. The glassware is then
dried and heated in an oven at 250°C for 15 to 30 minutes. 250 °C for 15 min to 30 min. All glassware is subsequently cleaned
with acetone followed by methanol.
6.3 All reagents and solvents should be of pesticide residue purity or higher to minimize interference problems.
6.4 Matrix interferences may be caused by contaminants in the sample. The extent of matrix interferences can vary considerably
from sample source depending on variations of the sample matrix.
7. Apparatus
7.1 LC/MS/MS System:
7.1.1 Liquid Chromatography (LC) System—A complete LC system is needed to analyze samples. This should include a sample
injection system, a solvent pumping system capable of mixing solvents, a sample compartment capable of maintaining required
temperature and a temperature controlled column compartment. A system that is capable of performing at the flows, pressures,
controlled temperatures, sample volumes, and requirements of the standard may be used.
7.1.2 Analytical Column —A C18 column was used to develop this test method.
NOTE 1—Any column that can achieve baseline resolution of these analytes may be used. Baseline resolution simplifies data analysis and can reduce the
chance of ion suppression, leading to higher limits of detection.
7.1.3 Tandem Mass Spectrometer (MS/MS) System—A MS/MS system capable of MRM analysis. Any system that is capable of
performing at the requirements in this standard may be used.
7.2 Filtration Device:
7.2.1 Hypodermic syringe—A Lock Tip Glass Syringe capable of holding a syringe-driven filter unit or similar may be used.
7.2.1.1 A 50-mL lock tip glass syringe size is recommended since a 50-mL sample size is used in this test method.
Additional information about rodenticides is available from United States Environmental Protection Agency (EPA), http://www.epa.gov.
A Waters ACQUITY UPLC (a trademark of the Waters Corporation, Milford, MA) BEH C18, 2.1 × 100 mm, 1.7 μm particle size was used to develop this test method.
Any column that achieves adequate resolution may be used. The retention times and order of elution may change depending on the column used andused, if you are aware
of an alternative column that meets the performance of the standard, please provide this information to ASTM International Headquarters. Your comments will receive careful
consideration at a meeting of the responsible technical committee, need to be monitored.which you may attend.
A Waters Quattro Premier (a trademark of the Waters Corporation, Milford, MA) XE tandem quadrupole mass spectrometer, or equivalent, was found suitable for use.
All parameters in this test method are based on this system and may vary depending on your instrument.
D7644 − 23
7.2.2 Filter Unit —PVDF filter units were used to filter the samples.
8. Reagents and Materials
8.1 Purity of Reagents—High Performance Liquid Chromatography (HPLC) pesticide residue analysis and spectrophotometry
grade chemicals shallmust be used in all tests. Unless indicated otherwise, it is intended that all reagents shallmust conform to the
Committee on Analytical Reagents of the American Chemical Society. Other reagent grades may be used provided they are first
determined to be of sufficiently high purity to permit their use without affecting the accuracy of the measurements.
8.2 Purity of Water—Unless otherwise indicated, references to water shallmust be understood to mean reagent water conforming
to Type 1 of Specification D1193. It must be demonstrated that this water does not contain contaminants at concentrations sufficient
to interfere with the analysis.
8.3 Gases—Ultrapure nitrogen and argon.
8.4 Methanol (CAS # 67-56-1).
8.5 Acetonitrile (CAS # 75-05-8).
8.6 Acetone (CAS # 67-64-1).
8.7 Ammonium Hydroxide (Concentrated, CAS # 1336-21-6).
8.8 Ascorbic Acid (CAS # 50-81-7).
8.9 Bromadiolone (CAS # 28772-56-7).
8.10 Brodifacoum (CAS # 56073-10-0).
8.11 Diphacinone (CAS # 82-66-6).
8.12 Warfarin (CAS # 81-81-2).
8.13 Warfarin-D (Phenyl-D , CAS # (unlabeled) 81-81-2).
5 5
8.13.1 Discussion—Warfarin-D is used as the electrospray positive analyte surrogate in this standard.
8.14 2-Bromo-4-(1,1,3,3-tetramethylbutyl)phenol (Br-OP).
8.14.1 Discussion—Br-OP is used as the electrospray negative analyte surrogate in this standard.
A Millex HV Syringe Driven Filter Unit PVDF 0.22 μm (Millipore Corporation, Catalog #SLGV033NS; Millex is a trademark of Merck KGAA, Darmstadt, Germany)
has been found suitable for use for this test method, any filter unit may be used was used, if you are aware of an alternative filter unit that meets the performance of this
test method the standard, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the
responsible technical committee, may be used. which you may attend.
Reagent Chemicals, American Chemical Society Specifications, ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, D.C. For SuggestionsDC. For suggestions on the testing of reagents not listed by the American Chemical Society, see
AnnualAnalar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulators,Formulary, U.S.
Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
A source of Warfarin-D is Cambridge Isotope Laboratories, 50 Frontage Road, Andover, MA 01810-5413. If you are aware of an alternative source that meets the
performance of the standard, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the
responsible technical committee, which you may attend.
A source of Br-OP is Accustandard, Inc., 125 Market Street, New Haven CT 06513. If you are aware of an alternative source that meets the performance of the standard,
please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical committee,
which you may attend.
Teflon is a trademark of The Chemours Company in Wilmington, DE.
D7644 − 23
9. Hazards
9.1 Normal laboratory safety applies to this method. Analysts should wear safety glasses, gloves, and lab coats when working in
the lab. Analysts should review the Safety Data Sheets (SDS) for all reagents used in this test method.
10. Sampling
10.1 Sampling—Grab samples must be collected in 40 mL pre-cleaned amber glass vials with Tefloninert-lined-lined caps
demonstrated to be free of interferences. Surface water samples are collected unpreserved, shipped between 0°C and 6°C,above
freezing and 6 °C, and stored in the laboratory between 0°C and 6°C.above freezing and 6 °C. Chlorinated drinking water samples
are dechlorinated with ascorbic acid; 10 mg of ascorbic acid is added to each 40 mL 40 mL vial prior to water collection. This test
method requires a 40 mL 40 mL sample size per analysis. Conventional sampling practices should be followed. Refer to Guide
D3856 and Practices D3694.
10.1.1 Ammonium acetate was evaluated as an agent to bind free chlorine in drinking water and was found to be ineffective in
the preservation of the rodenticides in chlorinated drinking water. Ascorbic acid was effective as a dechlorinating agent in chlorine
fortified Chicago tap water, which contained 3.2 ppm free chlorine and was dechlorinated with 10 mg ascorbic acid per 40 mL
water sample.
10.2 The samples are collected using 40 mL glass vials. A 40 mL volume is collected directly into the sample collection vial
without using any other measuring devices. This is a requirement due to the rodenticides’ affinity for surfaces, which will lead to
biased low results if transferring between containers. Before collection, the vials must be evaluated to determine a 40 mL 40 mL
sample volume. For example, the vials used in this test method were calibrated before use to determine that filling the vial to
approximately 1.6 cm below the rim would result in a 40 mL 40 mL sample volume. The greatest amount of water held by the
40 mL 40 mL vials used in this test method was approximately 42 mL. Vials filled to 42 mL in the field would not allow the
laboratory to spike the samples before quantitatively transferring to the 50 mL 50 mL graduated cylinder. It is imperative that the
samplers do not overfill the vials.
10.3 Preservation—Store samples between 0°C and 6°Cabove freezing and 6 °C from the time of collection until analysis.
Analyze the sample within 14 days of collection. Chlorinated drinking water samples are dechlorinated with ascorbic acid; 10 mg
of ascorbic acid is added to each 40 mL vial prior to water collection.
11. Preparation of LC/MS/MS
11.1 LC Chromatograph Operating Conditions:
11.1.1 Injection volumes of all calibration standards and samples are made at 50 μL volume using a full loop injection. If a 50
μL volume loop is installed in the LC, a “full loop” mode is the preferred technique when performing fast, qualitative analyses.
This mode should be used whenever accuracy and precision are the primary concerns. The first sample analyzed after the
calibration curve is a blank to ensure there is no carry-over. The gradient conditions for the liquid chromatograph are shown in
Table 2.
TABLE 2 Gradient Conditions for Liquid Chromatography
Percent Percent
Time Flow 95%95 % Water/ 95%95 % Methanol/
(min) (μL/min) 5%5 % Methanol, 5%5 % Water,
5 mM NH OH 5 mM NH OH
4 4
0.0 300 100 0
2.0 300 100 0
6.0 300 20 80
6.1 200 5 95
7.0 200 5 95
8.5 200 0 100
13.0 300 0 100
14.0 300 100 0
16.0 300 100 0
A Hach Pocket Colorimeter II (a trademark of Hach Company in Loveland, CO) Pocket Chlorine meter was used to measure free chlorine.
D7644 − 23
NOTE 2—If yourthe instrument does not have a 50 μL injection capability a different volume may be used. This is a performance-based method and
modifications are allowed as long as minimum performance criteria are met.
11.2 LC Sample Manager Conditions:
11.2.1 Wash Solvents—Weak wash is 4.0 mL of 95 % water/5 % methanol. Strong wash is 2.0 mL of methanol. The strong wash
solvent is needed to eliminate carry-over between injections of rodenticide samples. The weak wash is used to remove the strong
wash solvent. These rodenticides were shown to carry-over when acetonitrile was used for this analysis. The use of methanol
corrected this problem while providing separation and sensitivity. Instrument manufacturer specifications should be followed in
order to eliminate sample carry-over.
11.2.2 Temperatures—Column, 30°C;30 °C; Sample compartment, 15°C.15 °C.
11.2.3 Seal Wash—Solvent: 50% Acetonitrile/50%50 % Acetonitrile/50 % Water; Time: 5 minutes.min.
11.3 Mass Spectrometer Parameters:
11.3.1 To acquire the maximum number of data points per SRM channel while maintaining adequate sensitivity, the tune
parameters may be optimized according to yourthe instrument. Each peak requires at least 10 scans per peak for adequate
quantitation. This standard contains two surrogates and four target compounds that can be acquired in 5 MRM acquisition
functions. Variable parameters regarding retention times, SRM transitions, and cone and collision energies are shown in Table 3.
Mass spectrometer parameters used in the development of this test method are listed below:
The instrument is set in the Electrospray source setting.
Capillary Voltage: 3.5 kV
Cone: Variable depending on analyte (Table 3)
Extractor: 2 Volts
RF Lens: 0.1 Volts
Source Temperature: 120°C
Source Temperature: 120 °C
Desolvation Temperature: 375°C
Desolvation Temperature: 375 °C
Desolvation Gas Flow: 800 L/hr
Desolvation Gas Flow: 800 L/h
Cone Gas Flow: 25 L/hr
Cone Gas Flow: 25 L/h
Low Mass Resolution 1: 14.0
High Mass Resolution 1: 14.0
Ion Energy 1: 0.5
Entrance Energy: -1
Collision Energy: Variable depending on analyte (Table 3)
Exit Energy: 0
Low Mass Resolution 2: 14.0
High Mass resolution 2: 14.0
Ion Energy 2: 0.7
Multiplier: 650
-3
Gas Cell Pirani Gauge: 7.0 × 10 Torr
Inter-Channel Delay: 0.02 seconds
Inter-Channel Delay: 0.02 s
Inter-Scan Delay: 0.02 seconds
TABLE 3 Retention Times, SRM Ions, and Analyte-Specific Mass Spectrometer Parameters
SRM Primary/
Primary/ Retention Time Cone Voltage Collision Energy
Analyte ESI Mass Transition Confirmatory
Confirmatory (min) (Volts) (eV)
(Parent > Product) SRM Area Ratio
Primary 7.45 and 30 22 509.1 > 251.1
Bromadiolone Positive 1.0
Confirmatory 7.59 30 22 511.1 > 251.1
Primary 42 20 523.1 > 335.1
Brodifacoum Positive 7.95 1.1
Confirmatory 42 33 523.1 > 178
Primary 32 25 339.1 > 166.8
Diphacinone Negative 6.36 3.0
Confirmatory 32 47 339.1 > 115.8
Primary 26 14 309.1 > 162.8
Warfarin Positive 5.38 1.7
Confirmatory 26 20 309.1 > 251.1
Warfarin-D Primary 26 14 314.2 > 162.8
Positive 5.37 1.7
(Surrogate) Confirmatory 26 19 314.2 > 256.1
Br-OP
Negative Primary 8.92 35 25 283.1 > 78.6 N/A
(Surrogate)
D7644 − 23
Inter-Scan Delay: 0.02 s
Dwell: 0.1 seconds
Dwell: 0.1 s
12. Calibration and Standardization
12.1 The mass spectrometer must be calibrated per manufacturer specifications before analysis. In order to obtain valid and
accurate analytical values through this test method within the confidence limits, the following procedures must be followed when
performing the test method. Prepare all solutions in the lab using Class A volumetric glassware.
12.2 Calibration and Standardization—To calibrate the instrument, analyze 7 calibration standards containing the 7 concentration
levels of the rodenticides and surrogates prior to analysis as shown in Table 4. A calibration stock standard solution is prepared
from standard materials or they are purchased as certified solutions. Stock standard solution A containing bromadiolone,
brodifacoum, diphacinone, warfarin, warfarin-D (surrogate), and 2-Bromo-4-(1,1,3,3-tetramethylbutyl)phenol (surrogate) is
prepared at Level 7 concentration, and aliquots of that solution are diluted to prepare Levels 1 through 6. The following steps will
produce standards with the concentration values shown in Table 4. The analyst is responsible for recording initial component
weights carefully when working with pure materials and correctly carrying the weights through the dilution calculations.
12.2.1 Prepare stock standard solution A (Level 7) by adding to a 100 mL volumetric flask individual methanol solutions of the
following: 50 μL of bromadiolone, brodifacoum, diphacinone, warfarin, warfarin-D (surrogate) each at 4.0 mg/L and 50 μL of
2-Bromo-4-(1,1,3,3-tetramethylbutyl)phenol (surrogate) at 0.2 g/L, dilute to 100 mL with 80 % water/20 % methanol. The
preparation of the Level 7 standard can be accomplished using different volumes and concentrations of stock solutions as is
accustomed in the individual laboratory. Depending on the prepared stock concentrations, solubility at that concentration will have
to be ensured.
12.2.2 Aliquots of solution A are then diluted with 80 % water/20 % methanol to prepare the desired calibration levels in 2 mL
amber glass LC vials. The calibration vials must be used within 24 hours 24 h to ensure optimum results. Stock calibration
standards are routinely replaced every 7 days if not previously discarded for quality control failure. Calibration standards are not
filtered.
12.2.3 Inject each standard and obtain its chromatogram. An external calibration technique is used monitoring the primary and
confirmatory SRM transition of each analyte. Calibration software is utilized to conduct the quantitation of the target analytes and
surrogates using the primary SRM transition. The ratios of the primary/confirmatory SRM transition area counts are given in Table
3 and will vary depending on the individual tuning conditions. The primary/confirmatory SRM transition area ratio must be within
35 % of the individual labs accepted primary/confirmatory SRM transition area ratio. The primary SRM transition of each analyte
is used for quantitation and the confirmatory SRM transition for confirmation. This gives additional confirmation by isolating the
parent ion, forming two product ions via fragmentation, and relating it to the retention time in the calibration standard.
12.2.4 The calibration software manual should be consulted to properly use the software. The quantitation method is set as an
external calibration using the peak areas in ppt or ppb units, as long as the analyst is consistent. Concentrations may be calculated
using the data system software to generate linear regression or quadratic calibration curves. Forcing the calibration curve through
the origin is not recommended. Curves should be evaluated using relative error or relative standard error.
12.2.5 Linear calibration may be used if the coefficient of determination, r , is >0.98 for the analyte. The point of origin is excluded
and a fit weighting of 1/X is used in order to give more emphasis to the lower concentrations. If one of the calibration standards,
other than the high or low point, causes the rEach calibration of the curve to be <0.98, this point must be re-injected or a new
TABLE 4 Concentrations of Calibration Standards (ppt)
Analyte/Surrogate LV 1 LV 2 LV 3 LV 4 LV 5 LV 6 LV 7
Bromadiolone 100 200 500 750 1000 1500 2000
Brodifacoum 100 200 500 750 1000 1500 2000
Diphacinone 100 200 500 750 1000 1500 2000
Warfarin 100 200 500 750 1000 1500 2000
Warfarin-D (Surrogate) 100 200 500 750 1000 1500 2000
Br-OP (Surrogate) 5000 10 000 25 000 37 500 50 000 75 000 100 000
Management and Technical Requirements for Laboratories Performing Environmental Analysis; Module 4: Quality Systems for Chemical Testing; The NELAC Institute,
2017.
D7644 − 23
calibration curve must be regenerated. If the low or high point is excluded, minimally a five point curve is acceptable but the
reporting range must be modified to reflect this change.point used to generate the curve must have a calculated percent deviation
less than 25 % from the generated curve.
12.2.6 Quadratic calibration may be used if the coefficient of determination, r , is >0.99 for the analyte. The point of origin is
excluded, and a fit weighting of 1/X is used in order to give more emphasis to the lower concentrations. If one of the calibration
standards causes the curve to be <0.99, this point must be re-injected or a new calibration curve must be regenerated. Minimally
a six point curve is acceptable using a quadratic fit. Each calibration point used to generate the curve must have a calculated percent
deviation less than 25 % from the generated curve.
12.2.6.1 An initial seven point curve over the calibration range is encouraged in the event the low or high point must be excluded
to obtain a coefficient of determination >0.99. In this event, the reporting range must be modified to reflect this change.
12.2.7 The retention time window of the SRM transitions must be within 5 % of the retention time of the analyte in a midpoint
calibration standard. If this is not the case, re-analyze the calibration curve to determine if there was a shift in retention time during
the analysis, and the sample needs to be re-injected. If the retention time is still incorrect in the sample, refer to the analyte as an
unknown.
12.2.8 A midpoint calibration check standard must be analyzed at the end of each batch of 20 samples or within 24 hoursh after
the initial calibration curve was generated. This end calibration check should be the same calibration standard that was used to
generate the initial curve. The results from the end calibration check standard must have a percent deviation less than 30 % from
the calculated concentration for the target analytes and surrogates. If the results are not within these criteria, the problem must be
corrected, and either all samples in the batch must be re-analyzed against a new calibration curve or the affected results must be
qualified with an indication that they do not fall within the performance criteria of the test method. If the analyst inspects the vial
containing the end calibration check standard and notices that the sample evaporated affecting the concentration, a new end
calibration check standard may be made and analyzed. If this new end calibration check standard has a percent deviation less than
30 % from the calculated concentration for the target analytes and surrogates, the results may be reported unqualified.
12.3 If a laboratory has not performed the test before or if there has been a major change in the measurement system, for example,
new analyst, new instrument, etc., a precision and bias study must be performed to demonstrate laboratory capability.
12.3.1 Analyze at least four replicates of a sample solution containing bromadiolone, brodifacoum, diphacinone, warfarin,
warfarin-D (surrogate) and Br-OP (surrogate) at a concentration in the calibration range of Levels 3 to 5. A 750 ppt spike for
bromadiolone, brodifacoum, diphacinone and warfarin, 1000 ppt spike for warfarin-D (surrogate), and 50 000 ppt spike for Br-OP
(surrogate) were used to set the QC acceptance criteria in this test method. The matrix and chemistry should be similar to the
solution used in this test method. Each replicate must be taken through the complete analytical test method including any sample
preservation and pretreatment steps.
12.3.2 Calculate the mean (average) percent recovery and relative standard deviation (RSD) of the four values and compare to the
acceptable ranges of the QC acceptance criteria for the Initial Demonstration of Performance in Table 5.
12.3.3 This study should be repeated until the single operator precision and mean recovery are within the limits in Table 5. If a
concentration other than the recommended concentration is used, refer to Practice D5847 for information on applying the F test
and t test F-test and t-test in evaluating the acceptability of the mean and standard deviation.
TABLE 5 QC Acceptance Criteria
Initial Demonstration of Performance Lab Control Sample
Test Conc. Recovery (%) Precision Recovery (%)
Analyte
(ng/L)
Lower Upper Maximum Lower Up
...


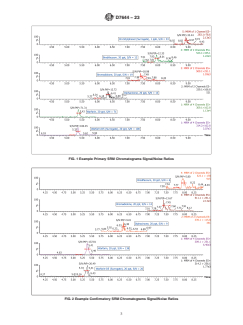



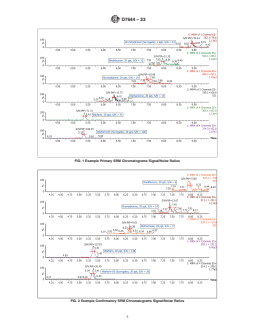

Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...