ASTM D932-20
(Practice)Standard Practice for Filamentous Iron Bacteria in Water and Water-Formed Deposits
Standard Practice for Filamentous Iron Bacteria in Water and Water-Formed Deposits
SIGNIFICANCE AND USE
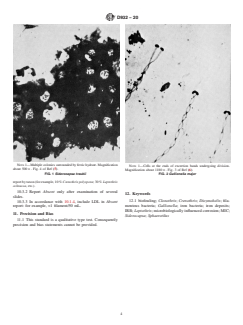
5.1 Filamentous iron bacteria is a general classification for microorganisms that utilize ferrous iron as a source of energy and are characterized by the deposition of ferric hydroxide in their mucilaginous sheaths. The process is continuous with these growths, and over a period of time large accumulations of slimy brown deposits can occur. Iron bacteria may clog water lines, reduce heat transfer, and cause staining; objectionable odors may arise following death of the bacteria. The organic matter in the water is consequently increased, and this in turn favors the multiplication of other bacteria.
SCOPE
1.1 This practice covers the determination of filamentous iron bacteria (FIB) by microscopic examination. This practice provides for the identification of the following genera of bacteria found in water and water-formed deposits: Siderocapsa, Gallionella (Dioymohelix), Sphaerotilus, Crenothrix, Leptothrix, and Clonothrix.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2020
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.24 - Water Microbiology
Relations
- Replaces
ASTM D932-15 - Standard Practice for Filamentous Iron Bacteria in Water and Water-Formed Deposits - Effective Date
- 01-May-2020
- Effective Date
- 01-Jul-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-Jun-2016
- Effective Date
- 01-Jun-2012
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-May-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 15-Feb-2006
- Effective Date
- 01-Jun-2004
Overview
ASTM D932-20: Standard Practice for Filamentous Iron Bacteria in Water and Water-Formed Deposits provides guidelines for identifying and evaluating the presence of filamentous iron bacteria (FIB) in water systems and water-formed deposits through microscopic examination. Filamentous iron bacteria, such as Siderocapsa, Gallionella (Dioymohelix), Sphaerotilus, Crenothrix, Leptothrix, and Clonothrix, are microorganisms that utilize ferrous iron for growth, producing deposits of ferric hydroxide. Over time, these bacteria can cause significant operational and maintenance issues in water infrastructure due to the accumulation of slimy brown deposits.
Key Topics
Significance of Detection:
- Filamentous iron bacteria are associated with biofouling, clogging of water lines, reduction in heat transfer, staining, and unpleasant odors in water systems.
- Their presence increases organic matter, enabling the proliferation of additional bacteria and potentially leading to microbiologically influenced corrosion (MIC).
Identification and Microscopy:
- The standard describes procedures for sampling, concentrating, and microscopic analysis of water and deposit samples.
- Identifies multiple genera by morphology, including distinctive iron or manganese-encrusted sheaths and specific structural features.
- Utilizes staining techniques (such as Hucker’s modified Gram stain) for clear differentiation.
Sampling and Testing Procedures:
- Provides guidance for collecting representative water and deposit samples for analysis.
- Describes sample concentration techniques including filtration and centrifugation to ensure the detection of even low populations.
- Outlines steps for slide preparation, staining, and systematic microscopic examination to identify and quantify FIB presence.
Reporting Requirements:
- Advises documentation of FIB as present or absent, with quantitative details such as relative abundance or coverage per microscope field.
- Encourages detailed reporting by bacterial taxon, particularly in mixed populations, to assess severity and inform appropriate mitigation strategies.
Applications
Water Utility and Treatment Plants:
- Regular monitoring of water supplies for iron bacteria is vital to prevent blockages, maintain system efficiency, and ensure water quality.
- Early detection supports timely intervention, reducing costly maintenance and mitigating risks such as pipe corrosion and staining.
Industrial Water Systems:
- Applicable in industrial cooling systems, heat exchangers, and process water lines where iron bacteria can decrease efficiency and lead to equipment failure.
- Inspection of water-formed deposits for FIB assists in diagnosing and resolving operational issues.
Environmental and Regulatory Compliance:
- Supports compliance with environmental and public health regulations related to water microbiology and infrastructure maintenance.
- Ensures that organizations can detect and control biofouling to minimize adverse impacts on distribution systems and the environment.
Related Standards
Several ASTM and referenced standards complement ASTM D932-20:
- ASTM D887: Practices for Sampling Water-Formed Deposits
- ASTM D1129: Terminology Relating to Water
- ASTM D1193: Specification for Reagent Water
- ASTM D3370: Practices for Sampling Water from Flowing Process Streams
- ASTM D5465: Practices for Determining Microbial Colony Counts from Waters Analyzed by Plating Methods
By following ASTM D932-20 in conjunction with these related standards, water professionals can ensure accurate and reliable identification of filamentous iron bacteria, support proactive maintenance, and safeguard water system integrity.
Keywords: filamentous iron bacteria, iron deposits, water-formed deposits, biofouling, microbiologically influenced corrosion, FIB detection, iron bacteria identification, ASTM D932-20, water sampling, water quality standards, Siderocapsa, Gallionella, Leptothrix, water microbiology
Buy Documents
ASTM D932-20 - Standard Practice for Filamentous Iron Bacteria in Water and Water-Formed Deposits
REDLINE ASTM D932-20 - Standard Practice for Filamentous Iron Bacteria in Water and Water-Formed Deposits
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D932-20 is a standard published by ASTM International. Its full title is "Standard Practice for Filamentous Iron Bacteria in Water and Water-Formed Deposits". This standard covers: SIGNIFICANCE AND USE 5.1 Filamentous iron bacteria is a general classification for microorganisms that utilize ferrous iron as a source of energy and are characterized by the deposition of ferric hydroxide in their mucilaginous sheaths. The process is continuous with these growths, and over a period of time large accumulations of slimy brown deposits can occur. Iron bacteria may clog water lines, reduce heat transfer, and cause staining; objectionable odors may arise following death of the bacteria. The organic matter in the water is consequently increased, and this in turn favors the multiplication of other bacteria. SCOPE 1.1 This practice covers the determination of filamentous iron bacteria (FIB) by microscopic examination. This practice provides for the identification of the following genera of bacteria found in water and water-formed deposits: Siderocapsa, Gallionella (Dioymohelix), Sphaerotilus, Crenothrix, Leptothrix, and Clonothrix. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Filamentous iron bacteria is a general classification for microorganisms that utilize ferrous iron as a source of energy and are characterized by the deposition of ferric hydroxide in their mucilaginous sheaths. The process is continuous with these growths, and over a period of time large accumulations of slimy brown deposits can occur. Iron bacteria may clog water lines, reduce heat transfer, and cause staining; objectionable odors may arise following death of the bacteria. The organic matter in the water is consequently increased, and this in turn favors the multiplication of other bacteria. SCOPE 1.1 This practice covers the determination of filamentous iron bacteria (FIB) by microscopic examination. This practice provides for the identification of the following genera of bacteria found in water and water-formed deposits: Siderocapsa, Gallionella (Dioymohelix), Sphaerotilus, Crenothrix, Leptothrix, and Clonothrix. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D932-20 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D932-20 has the following relationships with other standards: It is inter standard links to ASTM D932-15, ASTM D5465-16(2020), ASTM D1129-13(2020)e2, ASTM D5465-16, ASTM D5465-93(2012), ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D887-08, ASTM D3370-07, ASTM D1129-06ae1, ASTM D1129-06a, ASTM D1193-06, ASTM D1129-06, ASTM D5465-93(2004). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D932-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D932 − 20
Standard Practice for
Filamentous Iron Bacteria in Water and Water-Formed
Deposits
This standard is issued under the fixed designation D932; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3.1.1 For definitions of terms used in this standard, refer to
Terminology D1129.
1.1 This practice covers the determination of filamentous
iron bacteria (FIB) by microscopic examination. This practice
4. Summary of Practice
provides for the identification of the following genera of
4.1 The iron bacteria are generally filamentous, typically
bacteria found in water and water-formed deposits:
found in fresh water, and frequently surrounded by a sheath
Siderocapsa, Gallionella (Dioymohelix), Sphaerotilus,
which is usually encrusted with iron or manganese, or both (1,
Crenothrix, Leptothrix, and Clonothrix.
2). However, Starkey (3) reports another type which is
1.2 The values stated in SI units are to be regarded as
classified among the true bacteria. Detection and identification
standard. No other units of measurement are included in this
is accomplished by microscopic examination of sediment from
standard.
the sample.
1.3 This standard does not purport to address all of the
4.2 This practice provides a qualitative indication of the
safety concerns, if any, associated with its use. It is the
density of the filamentous iron bacteria and the severity of the
responsibility of the user of this standard to establish appro-
clogging problem in pipes caused by these bacteria.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
5. Significance and Use
1.4 This international standard was developed in accor-
5.1 Filamentous iron bacteria is a general classification for
dance with internationally recognized principles on standard-
microorganisms that utilize ferrous iron as a source of energy
ization established in the Decision on Principles for the
and are characterized by the deposition of ferric hydroxide in
Development of International Standards, Guides and Recom-
their mucilaginous sheaths. The process is continuous with
mendations issued by the World Trade Organization Technical
thesegrowths,andoveraperiodoftimelargeaccumulationsof
Barriers to Trade (TBT) Committee.
slimy brown deposits can occur. Iron bacteria may clog water
2. Referenced Documents
lines, reduce heat transfer, and cause staining; objectionable
2.1 ASTM Standards:
odors may arise following death of the bacteria. The organic
D887Practices for Sampling Water-Formed Deposits matter in the water is consequently increased, and this in turn
D1129Terminology Relating to Water
favors the multiplication of other bacteria.
D1193Specification for Reagent Water
D3370Practices for Sampling Water from Flowing Process 6. Apparatus
Streams
6.1 Centrifuge, complete with 250 mL conical bottles.
D5465Practices for Determining Microbial Colony Counts
6.2 Cover Glasses, round or square type, 19 mm ( ⁄4 in.) in
from Waters Analyzed by Plating Methods
diameter.
3. Terminology
6.3 Filter Paper or Blotter.
3.1 Definitions:
6.3.1 For 8.3.2.1 – Grade 5 (nominal 2.5 µm particle-size
retention).
This practice is under the jurisdiction ofASTM Committee D19 on Water and
6.3.2 For 9.3 – any absorbent paper medium will suffice.
is the direct responsibility of Subcommittee D19.24 on Water Microbiology.
6.4 Containers, sterile 1 L glass or plastic (can be autocla-
Current edition approved May 1, 2020. Published May 2020. Originally
approved in 1947. Last previous edition approved in 2015 as D932– 15. DOI:
vable).
10.1520/D0932-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on The boldface numbers in parentheses refer to a list of references at the end of
the ASTM website. this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D932 − 20
6.5 Membrane Filter, 0.45 µ nominal pore size, with appro- 8.3 Sampleconcentrationbyfollowingeither8.3.2or8.3.3.
priate filter-holding and vacuum assembly (see 9.2). 8.3.1 If the population is not sufficiently dense to be visible
to the naked eye, samples should be concentrated before
6.6 Microscope that provides a magnification of 400 to
staining and microscopic examination.
1000×andiscompletewithasuitablelightsource.Adark-field
8.3.2 Filtration—Useasmallsidestreamfiltertocollectthe
condenser is desirable.
sample to be examined.
6.7 Pipets, Mohr-type, 10-mL, with an opening 3 to 4 mm
8.3.2.1 Filterthewatersuspectedofcontainingironbacteria
in diameter, for thick samples, and 1-mLMohr-type pipets for
through a Grade 5 (nominal 2.5 µm particle-size retention)
thin samples or equivalent disposable plastic pipettes.
filter paper (6.3.1 or some other comparable media) for 24 h.
6.8 Slides, glass, standard type, 25 by 76 mm (1 by 3 in.) 8.3.2.2 Adjust the side-stream filter flow rate to match the
maximum filtration capacity of the filter medium used.
with either plain or frosted end.
8.3.3 Centrifugation:
6.9 Spatula, small and narrow, for handling thick samples.
8.3.3.1 Divide the 500 mLsample (8.2) equally, by weight,
among four 250 mL centrifuge bottles (6.1).
7. Reagents
8.3.3.2 Centrifuge the subsamples at 9000 to 12 000×gfor
7.1 Purity of Reagents—Reagent grade chemicals shall be
10 min.
used in all tests. Unless otherwise indicated, it is intended that
8.3.3.3 Decant the supernate from each 250-mL bottle.
all reagents shall conform to the specifications of the Commit-
8.3.3.4 Resuspend the pellet from one centrifuge bottle into
tee onAnalytical Reagents of theAmerican Chemical Society,
20 mL of phosphate buffer or physiological saline (Practices
where such specifications are available. Other grades may be
D5465).
used, provided it is first ascertained that the reagent is of
8.3.3.5 Transfer the suspension (8.3.3.4) to a second, pellet-
sufficiently high purity to permit its use without lessening the
containing centrifuge bottle and repeat 8.3.3.4.
accuracy of the determination.
8.3.3.6 Repeat 8.3.3.4 and 8.3.3.5 until all pellets and been
7.2 Purity of Water—Unless otherwise indicated, references
consolidated into a single 20-mL suspension.
towatershallbeunderstoodtomeanreagentwaterconforming
8.4 Regardless of the method used to concentrate the solids
to Specification D1193, Type II.
in the water, keep them moist until examined.
7.3 Hucker’s modification of the Gram stain (4).
8.5 Collect mud samples from the mud-water interface in
7.3.1 Crystal Violet Solution—Dissolve 2.0 g of crystal
order to obtain maximum bacterial populations.
v
violet (90% dye content) in 20 mLof ethyl alcohol (95% ⁄v).
8.6 Transfer the deposit or mud samples to wide-mouth
7.3.2 Ammonium Oxylate Solution—Dissolve 0.8 g of am-
bottles and add sterile phosphate buffer or physiological saline
monium oxalate monohydrate (NH ) C O •H O) in 80 mL of
4 2 2 4 2
(Practices D5465) to cover the deposits and maintain moisture
water.
until examined. Protect the samples from sunlight and hold at
7.3.3 Ammonium Oxalate-Crystal Violet Solution—
4°C during transportation and storage.
Combine crystal violet (7.3.1) and ammonium oxylate (7.3.2)
solutions and mix well to ensure that the salts are dissolved
8.7 As soon as possible after collection of the solids,
completely.
microscopically examine them for the presence of iron bacte-
ria.
7.4 3N Acid (1+4)—Mix 1 volume of hydrochloric acid
(HCl, sp gr 1.19) with 4 volumes of water.
9. Procedure
7.5 Iodine Solution—Prepare Gram’s modification of
9.1 Placeaportionofthesampleontheslide(6.8)andapply
Lugol’s solution (4) by dissolving1gof iodine in a solute
a cover glass (6.2).
containing2gof potassium iodide (KI) in 10 mLof water and
9.1.1 Useaspatula(6.9)orwide-mouthpipettotransferthe
diluting the resulting solution to 300 mL with water.
sample to the slide.
9.1.2 Whenflocsofmaterialareencountered,useapipet;as
8. Sampling
the flocs settle to the tip when the pipet is held in a vertical
8.1 Collect the samples in accordance with either Practices
position, and concentrate in the first drop.
D887 or D3370, whichever is applicable.
9.1.3 In the case of very dilute solids or a water sample,
8.2 Obtain a 500-m
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D932 − 15 D932 − 20
Standard Practice for
Filamentous Iron Bacteria in Water and Water-Formed
Deposits
This standard is issued under the fixed designation D932; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice covers the determination of filamentous iron bacteria (FIB) by examination under the microscope. The
microscopic examination. This practice provides for the identification of the following genera of bacteria found in water and
water-formed deposits: Siderocapsa,Gallionella (Dioymohelix),Sphaerotilus,Crenothrix,Leptothrix, and Clonothrix.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D887 Practices for Sampling Water-Formed Deposits
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D3370 Practices for Sampling Water from Flowing Process Streams
D5465 Practices for Determining Microbial Colony Counts from Waters Analyzed by Plating Methods
3. Terminology
3.1 Definitions—For definitions of terms used in this practice, refer to Terminology D1129. Definitions:
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
4. Summary of Test MethodPractice
4.1 The iron bacteria are generally filamentous, typically found in fresh water, and frequently surrounded by a sheath which is
usually encrusted with iron or manganese, or both (1, 2). However, Starkey (3) reports another type which is classified among
the true bacteria. Detection and identification is accomplished by microscopic examination of sediment from the sample.
4.2 This practice provides a qualitative indication of the density of the filamentous iron bacteria and the severity of the clogging
problem in pipes caused by these bacteria.
5. Significance and Use
5.1 Filamentous iron bacteria is a general classification for microorganisms that utilize ferrous iron as a source of energy and
are characterized by the deposition of ferric hydroxide in their mucilaginous sheaths. The process is continuous with these growths,
and over a period of time large accumulations of slimy brown deposits can occur. Iron bacteria may clog water lines, reduce heat
This practice is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.24 on Water Microbiology.
Current edition approved Feb. 1, 2015May 1, 2020. Published March 2015May 2020. Originally approved in 1947. Last previous edition approved in 20092015 as D932 –
85 (2009). 15. DOI: 10.1520/D0932-15.10.1520/D0932-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D932 − 20
transfer, and cause staining; objectionable odors may arise following death of the bacteria. The organic matter in the water is
consequently increased, and this in turn favors the multiplication of other bacteria.
6. Apparatus
6.1 Centrifuge, complete with 250 mL conical bottles.
6.2 Cover Glasses, round or square type, 19 mm ( ⁄4 in.) in diameter.
6.3 Filter Paper or Blotter.
6.3.1 For 8.3.2.1 – Grade 5 (nominal 2.5 μm particle-size retention).
6.3.2 For 9.3 – any absorbent paper medium will suffice.
6.4 Containers, sterile 1 L glass or plastic (can be autoclavable).
6.5 Membrane Filter, 0.45 μ nominal pore size, with appropriate filter-holding and vacuum assembly (see 9.2).
6.6 Microscope that provides a magnification of 400 to 1000× and is complete with a suitable light source. A dark-field
condenser is desirable.
6.7 Pipets, Mohr-type, 10-mL, with an opening 3 to 4 mm in diameter, for thick samples, and 1-mL Mohr-type pipets for thin
samples or equivalent disposable plastic pipettes.
6.8 Slides, glass, standard type, 25 by 76-mm 76 mm (1 by 3 in.) with either plain or frosted end.
6.9 Spatula, small and narrow, for handling thick samples.
7. Reagents
7.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without lessening the accuracy of the determination.
7.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water conforming to
Specification D1193, Type II.
7.3 Hucker’s modification of the Gram stain (4).
v
7.3.1 Crystal Violet Solution—Dissolve 2.0 g of crystal violet (90 % dye content) in 20 mL of ethyl alcohol (95 % ⁄v).
7.3.2 Ammonium Oxylate Solution—Dissolve 0.8 g of ammonium oxalate monohydrate (NH ) C O •H O) in 80 mL of water.
4 2 2 4 2
7.3.3 Ammonium Oxalate-Crystal Violet Solution—Combine crystal violet (2.3.1)(7.3.1) and ammonium oxylate (2.3.2)(7.3.2)
solutions and mix well to ensure that the salts are dissolved completely.
7.4 3N Acid (1 + 4)—Mix 1 volume of hydrochloric acid (HCl, sp gr 1.19) with 4 volumes of water.
7.5 Iodine Solution—Prepare Gram’sGram’s modification of Lugol’sLugol’s solution (4) by dissolving 1 g of iodine in a solute
containing 2 g of potassium iodide (KI) in 10 mL of water and diluting the resulting solution to 300 mL with water.
8. Sampling
8.1 Collect the samples in accordance with either Practices D887 or D3370, whichever is applicable.
8.2 Obtain a 500-mL (1-pt) sample of water, using a sterile 1-L (1-qt) bottle.
NOTE 1—The bottle should not be more than half-filled because of the oxygen demand of suspended matter; filling the bottle may cause the sample
to become anaerobic.
8.3 Sample concentration by following either 8.3.2 or 8.3.3.
8.3.1 If the population is not sufficiently dense to be visible to the naked eye, samples should be concentrated before staining
and microscopic examination.
8.3.2 Filtration—Use a small side stream filter to collect the sample to be examined.
8.3.2.1 Filter the water suspected of containing iron bacteria through a Grade 5 (nominal 2.5 μm particle-size retention) filter
paper (6.3.1 or some other comparable media) for 24 h.
8.3.2.2 Adjust the side-stream filter flow rate to match the maximum filtration capacity of the filter medium used.
8.3.3 Centrifugation:
8.3.3.1 Divide the 500 mL sample (8.2) equally, by weight, among four 250 mL centrifuge bottles (6.1).
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For Suggestionssuggestions on the testing of reagents not listed by the American Chemical Society, see
AnnualAnalar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial
Convention, Inc. (USPC), Rockville, MD.
D932 − 20
8.3.3.2 Centrifuge the subsamples at 9000 to 12 000 × g for 10 min.
8.3.3.3 Decant the supernate from each 250-mL bottle.
8.3.3.4 Resuspend the pellet from one centrifuge bottle into 20 mL of phosphate buffer or physiological saline (Practice(Prac-
tices D5465)).
8.3.3.5 Transfer the suspension (8.3.3.4) to a second, pellet-containing centrifuge bottle and repeat 8.3.3.4.
8.3.3.6 Repeat 8.3.3.4 and 8.3.3.5 until all pellets and been consolidated into a single 20-mL suspension.
8.4 Regardless of the method used to concentrate the solids in the water, keep them moist until examined.
8.5 Collect mud samples from the mud-water interface in order to obtain maximum bacterial populations.
8.6 Transfer the deposit or mud samples to wide-mouth bottles and add sterile phosphate buffer or physiological saline
(Practice(Practices D5465) to cover the deposits and maintain moisture until examined. Protect the samples from sunlight and hold
at 4°C during transportation and storage.
8.7 As soon as possible after collection of the solids, microscopically examine them for the presence of iron bacteria.
9. Procedure
9.1 Place a portion of the sample on the slide (6.8) and apply a cover glass (6.2).
9.1.1 Use a spatula (6.9) or wide-mouth pipet to transfer the sample to the slide.
9.1.2 When flocs of mater
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...