ASTM D8024-23
(Test Method)Standard Test Method for Determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in Water by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
Standard Test Method for Determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in Water by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
SIGNIFICANCE AND USE
5.1 TTPC may be used in various industrial and commercial products for use as a biocide. Products containing TTPC have been approved for controlling algal, bacterial, and fungal slimes in industrial water systems.2 TTPC should not be persistent in water but may be deposited in sediments at concentrations of concern. Hence, there is a need for quick, easy and robust method to determine TTPC concentration at trace levels in water matrices for understanding the sources and concentration levels in affected areas.
5.2 This method has been used to determine TTPC in reagent water and a river water (Table 8). (A) Solution A: Level 8 stock solution prepared according to Section 12 and at Table 4 concentrations.(B) Solution B: 75 % Acetone, 25 % Water.
Note 1: This test method has been used to characterize TTPC in real world water samples with success and similar recoveries as shown in Table 8.
SCOPE
1.1 This test method covers the determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in water by dilution with acetone, filtration and analysis by liquid chromatography/tandem mass spectrometry. This test method is not amenable for the analysis of isomeric mixtures of Tributyl-tetradecylphosphonium chloride. TTPC is a biocide that strongly adsorbs to soils.2 The water samples are prepared in a solution of 75 % acetone and 25 % water because TTPC has an affinity for surfaces and particles. The reporting range for this method is from 100 ng/L to 4000 ng/L. This analyte is qualitatively and quantitatively determined by this method. This test method adheres to multiple reaction monitoring (MRM) mass spectrometry.
1.2 A full collaborative study to meet the requirements of Practice D2777 has not been completed. This test method contains single-operator precision and bias based on single-operator data. Publication of standards that have not been fully validated is done to make the current technology accessible to users of standards, and to solicit additional input from the user community.
1.3 The Method Detection Limit3 (MDL) and Reporting Range4 for the target analyte are listed in Table 1.
1.3.1 The reporting limit in this test method is the minimum value below which data are documented as non-detects. Analyte detections between the method detection limit and the reporting limit are estimated concentrations and are not reported following this test method. The reporting limit is calculated from the concentration of the Level 1 calibration standard as shown in Table 4 for TTPC after taking into account a 2.5 mL water sample volume and a final diluted sample volume of 10 mL (75 % acetone/25 % water). The final solution volume is 10 mL because a 7.5 mL volume of acetone is added to each 2.5 mL water sample which is shaken and filtered.
1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Apr-2023
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Apr-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 01-Mar-2010
- Effective Date
- 15-Jul-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-May-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D8024-23 is the internationally recognized standard test method developed by ASTM for the quantitative and qualitative determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in water matrices. The method employs advanced multiple reaction monitoring liquid chromatography tandem mass spectrometry (LC/MS/MS) techniques, enabling detection and measurement of TTPC at trace levels in various water samples. This standard supports environmental monitoring, water quality assessment, and regulatory compliance by providing a robust, performance-based approach for detecting TTPC, a biocide commonly used in industrial water treatment.
Key Topics
Scope and Applicability
- Focuses on test method for detecting TTPC in water, including reagent and river waters
- Reporting range: 100 ng/L to 4000 ng/L
- Not suitable for isomeric mixtures of tributyl-tetradecylphosphonium chloride
- Emphasizes single-operator precision, with ongoing efforts toward multi-lab validation
Significance and Use
- TTPC is used as a biocide in industrial products for controlling algal, bacterial, and fungal slimes in water systems
- Detecting TTPC in water is vital since it may deposit in sediments, raising environmental concerns
- Provides a reliable method for tracing sources and quantifying TTPC concentrations in environmental and process waters
Test Method Details
- Involves acetone dilution, filtration, and LC/MS/MS analysis using multiple reaction monitoring
- Requires advanced instruments capable of specified flow, temperature, and detection parameters
- Utilizes robust sample preparation and quality control procedures, including calibration and matrix spike checks
Quality Control and Reporting
- Sets method detection limit (MDL) for TTPC at 13 ng/L
- Outlines procedures for calibration, surrogate addition, method blanks, and reporting limit check samples
- Stresses the importance of laboratory quality systems, precise sample handling, and thorough cleaning to prevent contamination
Applications
Environmental Monitoring
- Assessment of TTPC contamination in river, lake, and industrial water supplies
- Supports environmental impact studies and helps identify pollution sources
Industrial Water Quality
- Ensures compliance with regulations on biocide residues in water treatment systems
- Supports operational safety and product stewardship in manufacturing and utilities
Regulatory Compliance
- Assists laboratories and water authorities in meeting national and international environmental standards
- Provides data for reporting to regulatory agencies and stakeholders on TTPC levels in water resources
Related Standards
- ASTM D1129 - Terminology relating to water
- ASTM D1193 - Specification for reagent water
- ASTM D2777 - Practice for determining precision and bias of water methods
- ASTM D3856 - Water analysis laboratory management systems
- ASTM D4841 - Estimation of holding time for water samples
- ASTM D5847 - Writing quality control specifications for water analysis
- ASTM E2554 - Estimating and monitoring uncertainty using control charts
- EPA 40 CFR Part 136 - Procedures for method detection limits
- EPA SW-846 - Test methods for evaluating solid waste
ASTM D8024-23 forms a critical resource for laboratories, environmental specialists, and regulatory agencies needing accurate, sensitive, and repeatable testing protocols for TTPC in water using state-of-the-art LC/MS/MS technology. Implementing this standard ensures that monitoring of biocidal residues supports water quality and environmental safety objectives worldwide.
Buy Documents
ASTM D8024-23 - Standard Test Method for Determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in Water by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
REDLINE ASTM D8024-23 - Standard Test Method for Determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in Water by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D8024-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in Water by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)". This standard covers: SIGNIFICANCE AND USE 5.1 TTPC may be used in various industrial and commercial products for use as a biocide. Products containing TTPC have been approved for controlling algal, bacterial, and fungal slimes in industrial water systems.2 TTPC should not be persistent in water but may be deposited in sediments at concentrations of concern. Hence, there is a need for quick, easy and robust method to determine TTPC concentration at trace levels in water matrices for understanding the sources and concentration levels in affected areas. 5.2 This method has been used to determine TTPC in reagent water and a river water (Table 8). (A) Solution A: Level 8 stock solution prepared according to Section 12 and at Table 4 concentrations.(B) Solution B: 75 % Acetone, 25 % Water. Note 1: This test method has been used to characterize TTPC in real world water samples with success and similar recoveries as shown in Table 8. SCOPE 1.1 This test method covers the determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in water by dilution with acetone, filtration and analysis by liquid chromatography/tandem mass spectrometry. This test method is not amenable for the analysis of isomeric mixtures of Tributyl-tetradecylphosphonium chloride. TTPC is a biocide that strongly adsorbs to soils.2 The water samples are prepared in a solution of 75 % acetone and 25 % water because TTPC has an affinity for surfaces and particles. The reporting range for this method is from 100 ng/L to 4000 ng/L. This analyte is qualitatively and quantitatively determined by this method. This test method adheres to multiple reaction monitoring (MRM) mass spectrometry. 1.2 A full collaborative study to meet the requirements of Practice D2777 has not been completed. This test method contains single-operator precision and bias based on single-operator data. Publication of standards that have not been fully validated is done to make the current technology accessible to users of standards, and to solicit additional input from the user community. 1.3 The Method Detection Limit3 (MDL) and Reporting Range4 for the target analyte are listed in Table 1. 1.3.1 The reporting limit in this test method is the minimum value below which data are documented as non-detects. Analyte detections between the method detection limit and the reporting limit are estimated concentrations and are not reported following this test method. The reporting limit is calculated from the concentration of the Level 1 calibration standard as shown in Table 4 for TTPC after taking into account a 2.5 mL water sample volume and a final diluted sample volume of 10 mL (75 % acetone/25 % water). The final solution volume is 10 mL because a 7.5 mL volume of acetone is added to each 2.5 mL water sample which is shaken and filtered. 1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 TTPC may be used in various industrial and commercial products for use as a biocide. Products containing TTPC have been approved for controlling algal, bacterial, and fungal slimes in industrial water systems.2 TTPC should not be persistent in water but may be deposited in sediments at concentrations of concern. Hence, there is a need for quick, easy and robust method to determine TTPC concentration at trace levels in water matrices for understanding the sources and concentration levels in affected areas. 5.2 This method has been used to determine TTPC in reagent water and a river water (Table 8). (A) Solution A: Level 8 stock solution prepared according to Section 12 and at Table 4 concentrations.(B) Solution B: 75 % Acetone, 25 % Water. Note 1: This test method has been used to characterize TTPC in real world water samples with success and similar recoveries as shown in Table 8. SCOPE 1.1 This test method covers the determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in water by dilution with acetone, filtration and analysis by liquid chromatography/tandem mass spectrometry. This test method is not amenable for the analysis of isomeric mixtures of Tributyl-tetradecylphosphonium chloride. TTPC is a biocide that strongly adsorbs to soils.2 The water samples are prepared in a solution of 75 % acetone and 25 % water because TTPC has an affinity for surfaces and particles. The reporting range for this method is from 100 ng/L to 4000 ng/L. This analyte is qualitatively and quantitatively determined by this method. This test method adheres to multiple reaction monitoring (MRM) mass spectrometry. 1.2 A full collaborative study to meet the requirements of Practice D2777 has not been completed. This test method contains single-operator precision and bias based on single-operator data. Publication of standards that have not been fully validated is done to make the current technology accessible to users of standards, and to solicit additional input from the user community. 1.3 The Method Detection Limit3 (MDL) and Reporting Range4 for the target analyte are listed in Table 1. 1.3.1 The reporting limit in this test method is the minimum value below which data are documented as non-detects. Analyte detections between the method detection limit and the reporting limit are estimated concentrations and are not reported following this test method. The reporting limit is calculated from the concentration of the Level 1 calibration standard as shown in Table 4 for TTPC after taking into account a 2.5 mL water sample volume and a final diluted sample volume of 10 mL (75 % acetone/25 % water). The final solution volume is 10 mL because a 7.5 mL volume of acetone is added to each 2.5 mL water sample which is shaken and filtered. 1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D8024-23 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8024-23 has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM E2554-18e1, ASTM E2554-18, ASTM E2554-13, ASTM D4841-88(2013)e1, ASTM D4841-88(2013), ASTM D2777-12, ASTM D3856-11, ASTM D1129-10, ASTM D4841-88(2008), ASTM D2777-08, ASTM E2554-07, ASTM D1129-06ae1, ASTM D1129-06a, ASTM D2777-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8024-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D8024 − 23
Standard Test Method for
Determination of (Tri-n-butyl)-n-tetradecylphosphonium
chloride (TTPC) in Water by Multiple Reaction Monitoring
Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
This standard is issued under the fixed designation D8024; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.3.1 The reporting limit in this test method is the minimum
value below which data are documented as non-detects. Ana-
1.1 This test method covers the determination of (Tri-n-
lyte detections between the method detection limit and the
butyl)-n-tetradecylphosphonium chloride (TTPC) in water by
reporting limit are estimated concentrations and are not re-
dilution with acetone, filtration and analysis by liquid
ported following this test method. The reporting limit is
chromatography/tandem mass spectrometry. This test method
calculated from the concentration of the Level 1 calibration
is not amenable for the analysis of isomeric mixtures of
standard as shown in Table 4 for TTPC after taking into
Tributyl-tetradecylphosphonium chloride. TTPC is a biocide
2 account a 2.5 mL water sample volume and a final diluted
that strongly adsorbs to soils. The water samples are prepared
sample volume of 10 mL (75 % acetone/25 % water). The final
in a solution of 75 % acetone and 25 % water because TTPC
solution volume is 10 mL because a 7.5 mL volume of acetone
has an affinity for surfaces and particles. The reporting range
is added to each 2.5 mL water sample which is shaken and
for this method is from 100 ng ⁄L to 4000 ng ⁄L. This analyte is
filtered.
qualitatively and quantitatively determined by this method.
This test method adheres to multiple reaction monitoring 1.4 Units—The values stated in SI units are to be regarded
(MRM) mass spectrometry. as standard. No other units of measurement are included in this
standard.
1.2 A full collaborative study to meet the requirements of
1.5 This standard does not purport to address all of the
Practice D2777 has not been completed. This test method
safety concerns, if any, associated with its use. It is the
contains single-operator precision and bias based on single-
responsibility of the user of this standard to establish appro-
operator data. Publication of standards that have not been fully
priate safety, health, and environmental practices and deter-
validated is done to make the current technology accessible to
mine the applicability of regulatory limitations prior to use.
users of standards, and to solicit additional input from the user
1.6 This international standard was developed in accor-
community.
dance with internationally recognized principles on standard-
1.3 The Method Detection Limit (MDL) and Reporting
ization established in the Decision on Principles for the
Range for the target analyte are listed in Table 1.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
This test method is under the jurisdiction of ASTM Committee D19 on Water
and is the direct responsibility of Subcommittee D19.06 on Methods for Analysis for
2. Referenced Documents
Organic Substances in Water.
Current edition approved April 15, 2023. Published May 2023. Originally
2.1 ASTM Standards:
approved in 2016. Last previous edition approved in 2016 as D8024 – 16. DOI:
D1129 Terminology Relating to Water
10.1520/D8024-23.
2 D1193 Specification for Reagent Water
More information on TTPC can be found at http://www.buruenergy.com/wp-
content/uploads/BE-Environmental-Properties-of-Proposed-Biocide-BE-91.pdf
D2777 Practice for Determination of Precision and Bias of
(2014) and http://iaspub.epa.gov/sor_internet/registry/substreg/searchandretrieve/
Applicable Test Methods of Committee D19 on Water
advancedsearch/externalSearch.do?p_type=CASNO&p=81741-28-8 (2014).
D3856 Guide for Management Systems in Laboratories
The MDL is determined following the Code of Federal Regulations, 40 CFR
Engaged in Analysis of Water
Part 136, Appendix B, as a guide. A detailed process determining the MDL is
explained in the reference and is beyond the scope of this test method to be
explained here.
Reporting range concentration is calculated from Table 4 concentrations
assuming a 50 μL injection of the Level 1 calibration standard for TTPC, and the For referenced ASTM standards, visit the ASTM website, www.astm.org, or
highest level calibration standard with a 10 mL final diluted sample volume starting contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
with a 2.5 mL water sample. Volume variations will change the reporting limit and Standards volume information, refer to the standard’s Document Summary page on
ranges. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8024 − 23
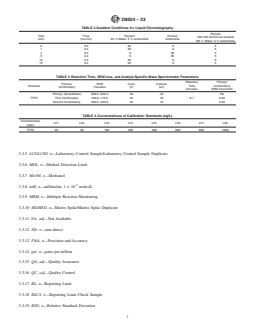
TABLE 1 Method Detection Limit and Reporting Range
3.3.11 NA, adj—Not Available
MDL Reporting Range
3.3.12 ND, n—non-detect
Analyte
(ng/L) (ng/L)
3.3.13 P&A, n—Precision and Accuracy
TTPC 13 100–4000
3.3.14 ppt, n—parts-per-trillion
3.3.15 QA, adj—Quality Assurance
D4841 Practice for Estimation of Holding Time for Water
3.3.16 QC, adj—Quality Control
Samples Containing Organic and Inorganic Constituents
3.3.17 RL, n—Reporting Limit
D5847 Practice for Writing Quality Control Specifications
for Standard Test Methods for Water Analysis 3.3.18 RLCS, n—Reporting Limit Check Sample
E2554 Practice for Estimating and Monitoring the Uncer-
3.3.19 RSD, n—Relative Standard Deviation
tainty of Test Results of a Test Method Using Control
3.3.20 RT, n—Retention Time
Chart Techniques
3.3.21 SDS, n—Safety Data Sheets
2.2 Other Standards:
3.3.22 SRM, n—Single Reaction Monitoring
40 CFR Part 136, Appendix B Definition and Procedure for
the Determination of the Method Detection Limit
3.3.23 SS, n—Surrogate Standard
EPA Publication SW-846 Test Methods for Evaluating Solid
3.3.24 TC, n—Target Compound
Waste, Physical/Chemical Methods
3.3.25 TTPBr, n—(Tri-n-butyl)-n-tetradecylphosphonium
3. Terminology bromide
3.3.26 TTPC, n—(Tri-n-butyl)-n-tetradecylphosphonium
3.1 Definitions:
chloride
3.1.1 For definitions of terms used in this standard, refer to
Terminology D1129.
3.3.27 VOA, n—Volatile Organic Analysis
3.2 Definitions of Terms Specific to This Standard:
3.2.1 reporting limit, RL, n—the minimum concentration
4. Summary of Test Method
below which data are documented as non-detects.
4.1 The operating conditions presented in this test method
3.2.2 reporting limit check sample, RLCS, n—a sample used
have been successfully used in the determination of TTPC in
to ensure that if the analyte was present at the reporting limit,
water; however, this test method is intended to be performance
it would be confidently identified.
based and alternative operating conditions can be used to
perform this method provided data quality objectives are
3.2.3 batch QC, n—all the quality control samples and
standards included in an analytical procedure. attained.
3.2.4 independent reference material, IRM, n—a material of 4.2 For TTPC analysis, samples are shipped to the lab on ice
known purity and concentration obtained either from the
and analyzed within 14 days of collection. A sample (2.5 mL)
National Institute of Standards and Technology (NIST) or other is transferred to a VOA vial, a TTPC spike solution is added to
reputable supplier.
Laboratory Control and Matrix Spike samples before the
3.2.4.1 Discussion—The IRM must be obtained from a addition of acetone. An isotopically labeled TTPC surrogate
different lot of material than is used for calibration.
would be added at this point. An isotopically labeled TTPBr-
D29 surrogate is now available and should be incorporated into
3.3 Acronyms:
this method by the user if requested by the customer. 7.5 mL
3.3.1 CCC, n—Continuing Calibration Check
of acetone is them added and the solution is mixed by hand or
3.3.2 CRW, n—Chicago River Water
vortex for 1 min. The samples are then filtered through a Nylon
3.3.3 IC, n—Initial Calibration
membrane syringe driven filter unit and then analyzed by
LC/MS/MS. All concentrations reported only to the reporting
3.3.4 LC, n—Liquid Chromatography
limit.
3.3.5 LCS/LCSD, n—Laboratory Control Sample/
Laboratory Control Sample Duplicate
3.3.6 MDL, n—Method Detection Limit
A surrogate, TTPBr-D29, was custom synthesized by Cambridge Isotope
3.3.7 MeOH, n—Methanol Laboratories Inc., Andover, MA 01810 and was found to be acceptable. Any
commercial source of TTPBr-D29 surrogate may be used, SRM transition 428.6 >
-3
3.3.8 mM, n—millimolar, 1 × 10 moles/L
372.5 was used. If you are aware of an alternative source that meets the performance
of the standard, please provide this information to ASTM International Headquar-
3.3.9 MRM, n—Multiple Reaction Monitoring
ters. Your comments will receive careful consideration at a meeting of the
3.3.10 MS/MSD, n—Matrix Spike/Matrix Spike Duplicate
responsible technical committee, which you may attend.
A Whatman Puradisc (a trademark of Whatman International Limited of
Maidstone, Kent) 25 NYL Disposable Filter unit (Diameter 25 mm, 0.2 μm Nylon
Available from U.S. Government Printing Office, Superintendent of membrane syringe driven filter unit has been found suitable for use for this method,
Documents, 732 N. Capitol St., NW, Washington, DC 20401-0001, http:// any filter unit may be used that meets the performance of this method may be used.
www.access.gpo.gov. If you are aware of an alternative filter that meets the performance of the standard,
Available from United States Environmental Protection Agency (EPA), William please provide this information to ASTM International Headquarters. Your com-
Jefferson Clinton Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20460, ments will receive careful consideration at a meeting of the responsible technical
http://www.epa.gov. committee, which you may attend.
D8024 − 23
TABLE 2 Gradient Conditions for Liquid Chromatography
Percent
Time Flow Percent Percent
400 mM Ammonium Acetate
(min) (mL/min) 95 % Water: 5 % Acetonitrile Acetonitrile
(95 % Water: 5 % Acetonitrile)
0 0.3 95 0 5
1 0.3 95 0 5
4 0.4 0 95 5
11 0.4 0 95 5
12 0.4 95 0 5
15 0.4 95 0 5
TABLE 3 Retention Time, SRM Ions, and Analyte-Specific Mass Spectrometer Parameters
Retention Primary/
Primary/ SRM Cone Collision
Chemical Time Confirmatory
Confirmatory Transition (V) (eV)
(minutes) SRM Area Ratio
Primary (Quantitation) 40 45 NA
399.5→229.3
TTPC First Confirmatory 399.5→75.9 40 46 8.1 0.92
Second Confirmatory 40 10 3.02
399.5→343.5
TABLE 4 Concentrations of Calibration Standards (ng/L)
Concentrations
LV1 LV2 LV3 LV4 LV5 LV6 LV7 LV8
(ng/L)
TTPC 25 50 100 200 400 600 800 1000
4.3 TTPC is identified by comparing the single reaction 6. Interferences
monitoring (SRM) transition and its confirmatory SRM tran-
6.1 All glassware is washed in hot water with detergent and
sitions if correlated to the known standard SRM transition
rinsed in hot water followed by distilled water. The glassware
(Table 3) and quantitated utilizing an external calibration. The
is then dried and heated in an oven at 250 °C for 15 min to
final report issued for each sample lists the concentration of
30 min. All glassware is subsequently rinsed or sonicated, or
TTPC, if detected, or RL, if not detected, in ng/L and surrogate
both, with acetone, n-propanol, or acetonitrile, or combinations
recovery, if available.
thereof.
5. Significance and Use
6.2 TTPC should not be a common contaminant found in a
5.1 TTPC may be used in various industrial and commercial
laboratory, unless involved in the analysis or matrices that
products for use as a biocide. Products containing TTPC have
contain TTPC. TTPC has been found to continue to adhere to
been approved for controlling algal, bacterial, and fungal
glassware and syringes after routine glassware washing. Rins-
slimes in industrial water systems. TTPC should not be
ing glassware with acetone, n-propanol, or acetonitrile, or
persistent in water but may be deposited in sediments at
combinations thereof, or even sonication, may be required to
concentrations of concern. Hence, there is a need for quick,
remove TTPC. All of the materials and supplies are routinely
easy and robust method to determine TTPC concentration at
demonstrated to be free from interferences of TTPC by
trace levels in water matrices for understanding the sources and
analyzing laboratory blanks under the same conditions as the
concentration levels in affected areas.
samples. If found, measures should be taken to remove the
5.2 This method has been used to determine TTPC in
contamination or data should be qualified, background subtrac-
reagent water and a river water (Table 8).
tion of blank contamination is not allowed.
NOTE 1—This test method has been used to characterize TTPC in real
6.3 All reagents and solvents should be pesticide residue
world water samples with success and similar recoveries as shown in
Table 8. purity or higher to minimize interference problems.
TABLE 5 Preparation of Calibration Standards
Solution LV1 LV2 LV3 LV4 LV5 LV6 LV7 LV8
A
A 25 μL 50 μL 100 μL 200 μL 400 μL 600 μL 800 μL 1000 μL
B
B 975 μL 950 μL 900 μL 800 μL 600 μL 400 μL 200 μL 0 μL
A
Solution A: Level 8 stock solution prepared according to Section 12 and at Table 4 concentrations.
B
Solution B: 75 % Acetone, 25 % Water.
D8024 − 23
TABLE 6 QC Acceptance Criteria
NOTE 1—Table 6 data is preliminary until a multi-lab validation study is completed.
Initial Demonstration of Performance Laboratory Control Sample
Spike
Recovery (%) Precision Recovery (%)
Analyte Conc.
Maximum
Lower Control Limit Upper Control Limit
ng/L
Lower Limit Upper Limit
(LCL) % (UCL) %
% RSD
TTPC 2000 70 130 30 70 130
TABLE 7 MS/MSD QC Acceptance Criteria
NOTE 1—Table 7 data is preliminary until a multi-lab validation study is completed.
MS/MSD Precision
Spike
Analyte Conc. Recovery (%)
RPD (%)
ng/L
Lower Limit Upper Limit
TTPC 2000 70 130 30
TABLE 8 Single-Laboratory Recovery Data in Water
Reagent Water P&A Data CRW P&A Data
(2000 ng/L spike) (2000 ng/L spike)
Sample
TTPC TTPC
MB 1
MB 2
P&A 1 1861 1898
P&A 2 1891 1965
P&A 3 1882 1933
P&A 4 1894 1871
P&A 5 1975 1929
P&A 6 1975 1974
Average Recovery (ng/L) 1893 1928
% Average Recovery 94.6 96.4
Standard Deviation 42.8 39.1
RSD (%) 2.26 2.03
6.4 Matrix interferences may be caused by contaminants in column compartment. A LC system that is capable of perform-
the sample. The extent of matrix interferences can vary ing at the flows, pressures, controlled temperatures, sample
considerably depending on variations in the sample matrices. volumes, and requirements of this test method must be used.
7.1.2 Analytical Column —A reverse phase C18 particle
6.5 Automatic pipettes with polypropylene tips were used to
column was used to develop this test method. Any column that
prepare the standards, spiking, and calibration solutions. The
achieves adequate resolution may be used. The retention times
use of glass syringes for preparing standards, spiking, and
and order of elution may change depending on the column used
calibration solutions generated erratic results; glass syringes
and need to be monitored.
should be avoided for these critical tasks. A thoroughly cleaned
10 mL or 20 mL hypodermic glass syringe with a nylon filter 7.2 Tandem Mass Spectrometer System—A MS/MS system
was found to perform well to filter the 10 mL prepared sample. capable of multiple reaction monitoring (MRM) analysis or
20 mL Luer-Lock polypropylene syringes have also been any system that is capable of meeting the requirements in this
shown to meet the performance of this test method; these are test method must be used.
disposable and do not require cleaning before use. It seems
7.3 Adjustable Volume Pipettes—10 μL, 20 μL, 100 μL,
when preparing small volumes of solutions, concentrations are
1000 μL, 5 mL, and 10 mL.
affected by adhesion of TTPC to the syringe barrel or plunger.
NOTE 3—Any pipette may be used providing the data generated meets
NOTE 2—The use of PTFE, PVDF and polypropylene filter units
the performance of this test method.
resulted in poor performance and low recoveries.
7.3.1 Pipette Tips—Polypropylene pipette tips free of re-
lease agents or low retention coating of various sizes.
7. Apparatus
7.1 LC/MS/MS System:
7.1.1 Liquid Chromatography System—A complete LC sys-
A Waters Acquity UPLC BEH C18, 2.1 mm × 100 mm and 1.7 μm particle size
tem is required in order to analyze samples, this should include
column was used. If you are aware of an alternative column that meets the
a sample injection system, a solvent pumping system capable
performance of the standard, please provide this information to ASTM International
of mixing solvents, a sample compartment capable of main-
Headquarters. Your comments will receive careful consideration at a meeting of the
taining required temperature and a temperature controlled responsible technical committee, which you may attend.
D8024 − 23
7.4 Class A Volumetric Glassware. 8.10 Ottawa Sand (CAS # 14808-60-7).
7.5 Filtration Device. 8.11 (Tri-n-butyl)-n-tetradecylphosphonium chloride (CAS
7.5.1 Hypodermic Syringe—A Luer-Lock tip glass or poly- # 81741-28-8).
propylene syringe capable of holding a syringe driven filter
8.12 (Tri-n-butyl)-n-tetradecylphosphonium bromide –D29
unit.
(TTPBr-D29, CAS # NA, optional surrogate).
7.5.2 A 10 mL or 20 mL Lock Tip Syringe size is recom-
mended since a 10 mL prepared sample size is used in this test
9. Hazards
method. If a smaller volume syringe is used, do not wash out
9.1 Normal laboratory safety applies to this method. Ana-
the syringe or change filters while filtering the same sample if
lysts should wear safety glasses, gloves, and lab coats when
multiple refills of the syringe is required in order to filter the
working in the lab. Analysts should review the Safety Data
10 mL prepared sample.
Sheets (SDS) for all reagents used in this method.
7.5.3 Filter Unit —Nylon filter units were used to filter the
samples.
10. Sampling and Preservation
7.6 Vials—2-mL autosampler vials (LC vials) with pre-slit
10.1 Grab samples are collected in glass containers with
PTFE/silicone septa or equivalent.
inert-lined caps. As part of the overall quality assurance
program for this test method, field blanks exposed to the same
7.7 VOA Vials—40 mL.
field conditions as samples are collected and analyzed accord-
7.8 Sonicator.
ing to this test method to assess the potential for field
7.9 Oven, capable to achieve 250 °C.
contamination, refer to Guide D3856 as a guide for sampling.
This test method is based on a 2.5 mL sample size per analysis.
8. Reagents and Materials If different sample sizes are used, spiking solution amounts
may need to be modified. EPA Publication SW-8466 may be
8.1 Purity of Reagents—High Performance Liquid Chroma-
used as a sampling guide. Samples must be shipped with a trip
tography (HPLC) pesticide residue analysis and spectropho-
blank between above freezing and 6 °C.
tometry grade chemicals must be used in all tests. Unless
indicated otherwise, it is intended that all reagents must
10.2 Once received the sample temperature is taken and
conform to the Committee on Analytical Reagents of the should be less than 6 °C. If the receiving temperature is greater
American Chemical Society. Other reagent grades may be
than 6 °C, the sample temperature is noted in the case narrative
used provided they are first determined to be of sufficiently accompanying the data. Samples should be stored refrigerated
high purity to permit their use without affecting the accuracy of
between above freezing and 6 °C from the time of collection
the measurements. until analysis.
8.2 Purity of Water—Unless otherwise indicated, references 10.3 The sample should be analyzed within 14 days of
to water must be understood to mean reagent water conforming collection. No holding time study has been done on water
to Type 1 of Specification D1193. It must be demonstrated that matrices tested in this test method. Holding time may vary
this water does not contain contaminants at concentrations depending on the matrix and individual laboratories should
sufficient to interfere with the analysis. determine the holding time in their matrix, refer to Practice
D4841.
8.3 All prepared solutions are routinely replaced every year
if not previously discarded for quality control failure.
11. Preparation of LC/MS/MS
8.4 Gases—Ultrapure nitrogen and argon.
11.1 LC Chromatograph Operating Conditions:
8.5 Acetone (CAS # 67-64-1). 11.1.1 Injections of all standards and samples are made at a
50 μL volume. Other injection volumes may be used to
8.6 Acetonitrile (CAS # 75-05-8).
optimize conditions. Standards and sample extracts must be in
8.7 Methanol (CAS # 67-56-1).
a 75:25 acetone:water solution. In the case of extreme concen-
8.8 Ammonium Acetate (CAS # 631-61-8). tration differences amongst samples, it is wise to analyze a
blank after a concentrated sample and before a dilute sample to
8.9 2-Propanol (isopropyl alcohol, CAS # 67-63-0).
minimize carry-over of analytes from injection to injection.
However, there should not be carry-over between samples. The
LC utilized to develop this test method has a flow through LC
A Whatman Puradisc 25 NYL Disposable Filter unit (diameter 25 mm, 0.2 μm
needle design. The gradient conditions for liquid chromatog-
nylon membrane syringe driven filter unit) was used. If you are aware of an
raphy are shown in Table 2.
alternative filter that meets the performance of the standard, please provide this
information to ASTM International Headquarters. Your comments will receive
11.2 LC Sample Manager Conditions:
careful consideration at a meeting of the responsible technical committee, which
11.2.1 Needle Wash Solvent—60 % acetonitrile ⁄40 %
you may attend.
2-propanol. Eight-second wash time before and after injection.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
Instrument manufacturer’s specifications should be followed in
DC. For suggestions on the testing of reagents not listed by the American Chemical
order to eliminate sample carry-over.
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
11.2.2 Temperatures—Column, 35 °C; sample
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. compartment, 15 °C.
D8024 − 23
11.2.3 Seal Wash—Solvent: 50 % methanol ⁄50 % water; consistency. 500 μL of TTPC Target Spike Solution (100 μg ⁄L,
time: 5 min. 12.7) is added to a 50 mL volumetric flask and diluted to
50 mL with 75:25 acetone:water. The preparation of the Level
11.3 Mass Spectrometer Parameters:
8 standard can be accomplished using appropriate volumes and
11.3.1 To acquire the maximum number of data points per
concentrations of stock solutions as in accordance with a
SRM channel while maintaining adequate sensitivity, the tune
particular laboratory’s standard procedure.
parameters may be optimized according to the instrument used.
12.3.2 Aliquots of Solution A are then diluted with 75:25
Each peak requires at least 10 scans per peak for adequate
acetone:water to prepare the 1 mL desired calibration levels in
quantitation. Variable parameters regarding SRM transitions,
2 mL amber glass LC vials (Table 5). The calibration vials
and cone and collision energies are shown in Table 3. The
must be used w
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D8024 − 16 D8024 − 23
Standard Test Method for
Determination of (Tri-n-butyl)-n-tetradecylphosphonium
chloride (TTPC) in Water by Multiple Reaction Monitoring
Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
This standard is issued under the fixed designation D8024; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the determination of (Tri-n-butyl)-n-tetradecylphosphonium chloride (TTPC) in water by dilution with
acetone, filtration and analysis by liquid chromatography/tandem mass spectrometry. This test method is not amenable for the
analysis of isomeric mixtures of Tributyl-tetradecylphosphonium chloride. TTPC is a biocide that strongly adsorbs to soils. The
water samples are prepared in a solution of 75 % acetone and 25 % water because TTPC has an affinity for surfaces and particles.
The reporting range for this method is from 100–4000100 ng ng/L. ⁄L to 4000 ng ⁄L. This analyte is qualitatively and quantitatively
determined by this method. This test method adheres to multiple reaction monitoring (MRM) mass spectrometry.
1.2 A full collaborative study to meet the requirements of Practice D2777 has not been completed. This test method contains
single-operator precision and bias based on single-operator data. Publication of standards that have not been fully validated is done
to make the current technology accessible to users of standards, and to solicit additional input from the user community.
3 4
1.3 The Method Detection Limit (MDL) and Reporting Range for the target analyte are listed in Table 1.
1.3.1 The reporting limit in this test method is the minimum value below which data are documented as non-detects. Analyte
detections between the method detection limit and the reporting limit are estimated concentrations and are not reported following
this test method. The reporting limit is calculated from the concentration of the Level 1 calibration standard as shown in Table 4
for TTPC after taking into account a 2.5 mL water sample volume and a final diluted sample volume of 10 mL (75 % acetone/25
% water). The final solution volume is 10 mL because a 7.5 mL volume of acetone is added to each 2.5 mL water sample which
is shaken and filtered.
1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this
standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.06 on Methods for Analysis for
Organic Substances in Water.
Current edition approved June 1, 2016April 15, 2023. Published August 2016May 2023. Originally approved in 2016. Last previous edition approved in 2016 as
D8024 – 16. DOI: 10.1520/D8024-16.10.1520/D8024-23.
More information on TTPC can be found at http://www.buruenergy.com/wp-content/uploads/BE-Environmental-Properties-of-Proposed-Biocide-BE-91.pdf (2014) and
http://iaspub.epa.gov/sor_internet/registry/substreg/searchandretrieve/advancedsearch/externalSearch.do?p_type=CASNO&p=81741-28-8 (2014).
The MDL is determined following the Code of Federal Regulations, 40 CFR Part 136, Appendix B, as a guide. A detailed process determining the MDL is explained
in the reference and is beyond the scope of this test method to be explained here.
Reporting range concentration is calculated from Table 4 concentrations assuming a 50 μL injection of the Level 1 calibration standard for TTPC, and the highest level
calibration standard with a 10 mL final diluted sample volume starting with a 2.5 mL water sample. Volume variations will change the reporting limit and ranges.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8024 − 23
TABLE 1 Method Detection Limit and Reporting Range
MDL Reporting Range
Analyte
(ng/L) (ng/L)
TTPC 13 100–4000
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3856 Guide for Management Systems in Laboratories Engaged in Analysis of Water
D4841 Practice for Estimation of Holding Time for Water Samples Containing Organic and Inorganic Constituents
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
E2554 Practice for Estimating and Monitoring the Uncertainty of Test Results of a Test Method Using Control Chart Techniques
2.2 Other Standards:
40 CFR Part 136, Appendix B Definition and Procedure for the Determination of the Method Detection Limit
EPA Publication SW-846 Test Methods for Evaluating Solid Waste, Physical/Chemical Methods
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 reporting limit, RL, n—the minimum concentration below which data are documented as non-detects.
3.2.2 reporting limit check sample, RLCS, n—a sample used to ensure that if the analyte was present at the reporting limit, it would
be confidently identified.
3.2.3 batch QC, n—all the quality control samples and standards included in an analytical procedure.
3.2.4 independent reference material, IRM, n—a material of known purity and concentration obtained either from the National
Institute of Standards and Technology (NIST) or other reputable supplier.
3.2.4.1 Discussion—
The IRM shallmust be obtained from a different lot of material than is used for calibration.
3.3 Acronyms:
3.3.1 CCC, n—Continuing Calibration Check
3.3.2 CRW, n—Chicago River Water
3.3.3 IC, n—Initial Calibration
3.3.4 LC, n—Liquid Chromatography
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from U.S. Government Printing Office, Superintendent of Documents, 732 N. Capitol St., NW, Washington, DC 20401-0001, http://www.access.gpo.gov.
Available from United States Environmental Protection Agency (EPA), William Jefferson Clinton Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20460,
http://www.epa.gov.
D8024 − 23
TABLE 2 Gradient Conditions for Liquid Chromatography
Percent
Time Flow Percent Percent
400 mM Ammonium Acetate
(min) (mL/min) 95 % Water: 5 % Acetonitrile Acetonitrile
(95 % Water: 5 % Acetonitrile)
0 0.3 95 0 5
1 0.3 95 0 5
4 0.4 0 95 5
11 0.4 0 95 5
12 0.4 95 0 5
15 0.4 95 0 5
TABLE 3 Retention Time, SRM Ions, and Analyte-Specific Mass Spectrometer Parameters
Retention Primary/
Primary/ SRM Cone Collision
Chemical Time Confirmatory
Confirmatory Transition (V) (eV)
(minutes) SRM Area Ratio
Primary (Quantitation) 40 45 NA
399.5→229.3
TTPC First Confirmatory 399.5→75.9 40 46 8.1 0.92
Second Confirmatory 40 10 3.02
399.5→343.5
TABLE 4 Concentrations of Calibration Standards (ng/L)
Concentrations
LV1 LV2 LV3 LV4 LV5 LV6 LV7 LV8
(ng/L)
TTPC 25 50 100 200 400 600 800 1000
3.3.5 LCS/LCSD, n—Laboratory Control Sample/Laboratory Control Sample Duplicate
3.3.6 MDL, n—Method Detection Limit
3.3.7 MeOH, n—Methanol
-3
3.3.8 mM, n—millimolar, 1 × 10 moles/L
3.3.9 MRM, n—Multiple Reaction Monitoring
3.3.10 MS/MSD, n—Matrix Spike/Matrix Spike Duplicate
3.3.11 NA, adj—Not Available
3.3.12 ND, n—non-detect
3.3.13 P&A, n—Precision and Accuracy
3.3.14 ppt, n—parts-per-trillion
3.3.15 QA, adj—Quality Assurance
3.3.16 QC, adj—Quality Control
3.3.17 RL, n—Reporting Limit
3.3.18 RLCS, n—Reporting Limit Check Sample
3.3.19 RSD, n—Relative Standard Deviation
D8024 − 23
3.3.20 RT, n—Retention Time
3.3.21 SDS, n—Safety Data Sheets
3.3.22 SRM, n—Single Reaction Monitoring
3.3.23 SS, n—Surrogate Standard
3.3.24 TC, n—Target Compound
3.3.25 TTPBr, n—(Tri-n-butyl)-n-tetradecylphosphonium bromide
3.3.26 TTPC, n—(Tri-n-butyl)-n-tetradecylphosphonium chloride
3.3.27 VOA, n—Volatile Organic Analysis
4. Summary of Test Method
4.1 The operating conditions presented in this test method have been successfully used in the determination of TTPC in water;
however, this test method is intended to be performance based and alternative operating conditions can be used to perform this
method provided data quality objectives are attained.
4.2 For TTPC analysis, samples are shipped to the lab on ice and analyzed within 14 days of collection. A sample (2.5 mL) is
transferred to a VOA vial, a TTPC spike solution is added to Laboratory Control and Matrix Spike samples before the addition
of acetone. An isotopically labeled TTPC surrogate would be added at this point. An isotopically labeled TTPBr-D29 surrogate
is now available and should be incorporated into this method by the user if requested by the customer. 7.5 mL of acetone is them
added and the solution is mixed by hand or vortex for 1 minute. 1 min. The samples are then filtered through a Nylon membrane
syringe driven filter unit and then analyzed by LC/MS/MS. All concentrations reported only to the reporting limit.
4.3 TTPC is identified by comparing the single reaction monitoring (SRM) transition and its confirmatory SRM transitions if
correlated to the known standard SRM transition (Table 3) and quantitated utilizing an external calibration. The final report issued
for each sample lists the concentration of TTPC, if detected, or RL, if not detected, in ng/L and surrogate recovery, if available.
5. Significance and Use
5.1 TTPC may be used in various industrial and commercial products for use as a biocide. Products containing TTPC have been
approved for controlling algal, bacterial, and fungal slimes in industrial water systems. TTPC should not be persistent in water
TABLE 5 Preparation of Calibration Standards
Solution LV1 LV2 LV3 LV4 LV5 LV6 LV7 LV8
A
A 25 μL 50 μL 100 μL 200 μL 400 μL 600 μL 800 μL 1000 μL
B
B 975 μL 950 μL 900 μL 800 μL 600 μL 400 μL 200 μL 0 μL
A
Solution A: Level 8 stock solution prepared according to Section 12 and at Table 4 concentrations.
B
Solution B: 75 % Acetone, 25 % Water.
A surrogate, TTPBr-D29, was custom synthesized by Cambridge Isotope Laboratories Inc., Andover, MA 01810 and was found to be acceptable. Any commercial source
of TTPBr-D29 surrogate may be used, SRM transition 428.6 > 372.5 was used. If you are aware of an alternative source that meets the performance of the standard, please
provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical committee, which
you may attend.
A Whatman Puradisc (a trademark of Whatman International Limited of Maidstone, Kent) 25 NYL Disposable Filter unit (Diameter 25 mm, 0.2 μm Nylon membrane
syringe driven filter unit has been found suitable for use for this method, any filter unit may be used that meets the performance of this method may be used. If you are aware
of an alternative filter that meets the performance of the standard, please provide this information to ASTM International Headquarters. Your comments will receive careful
consideration at a meeting of the responsible technical committee, which you may attend.
D8024 − 23
but may be deposited in sediments at concentrations of concern. Hence, there is a need for quick, easy and robust method to
determine TTPC concentration at trace levels in water matrices for understanding the sources and concentration levels in affected
areas.
5.2 This method has been used to determine TTPC in reagent water and a river water (Table 8).
NOTE 1—This test method has been used to characterize TTPC in real world water samples with success and similar recoveries as shown in Table 8.
6. Interferences
6.1 All glassware is washed in hot water with detergent and rinsed in hot water followed by distilled water. The glassware is then
dried and heated in an oven at 250°C for 15 to 30 minutes. 250 °C for 15 min to 30 min. All glassware is subsequently rinsed and/or
sonicated with acetone, n-propanol and/or acetonitrile.or sonicated, or both, with acetone, n-propanol, or acetonitrile, or
combinations thereof.
6.2 TTPC should not be a common contaminant found in a laboratory, unless involved in the analysis or matrices that contain
TTPC. TTPC has been found to continue to adhere to glassware and syringes after routine glassware washing. Rinsing glassware
with acetone, n-propanol and/orn-propanol, or acetonitrile, or combinations thereof, or even sonication, may be required to remove
TTPC. All of the materials and supplies are routinely demonstrated to be free from interferences of TTPC by analyzing laboratory
blanks under the same conditions as the samples. If found, measures should be taken to remove the contamination or data should
be qualified, background subtraction of blank contamination is not allowed.
6.3 All reagents and solvents should be pesticide residue purity or higher to minimize interference problems.
6.4 Matrix interferences may be caused by contaminants in the sample. The extent of matrix interferences can vary considerably
depending on variations in the sample matrices.
6.5 Automatic pipettes with polypropylene tips were used to prepare the standards, spiking, and calibration solutions. The use of
glass syringes for preparing standards, spiking, and calibration solutions generated erratic results; glass syringes should be avoided
for these critical tasks. A thoroughly cleaned 1010 mL or 20 mL hypodermic glass syringe with a nylon filter was found to perform
well to filter the 10 mL prepared sample. 20 mL 20 mL Luer-Lock polypropylene syringes have also been shown to meet the
performance of this test method; these are disposable and do not require cleaning before use. It seems when preparing small
volumes of solutions, concentrations are affected by adhesion of TTPC to the syringe barrel or plunger.
TABLE 6 QC Acceptance Criteria
NOTE 1—Table 6 data is preliminary until a multi-lab validation study is completed.
Initial Demonstration of Performance Laboratory Control Sample
Spike
Recovery (%) Precision Recovery (%)
Analyte Conc.
Maximum
Lower Control Limit Upper Control Limit
ng/L
Lower Limit Upper Limit
(LCL) % (UCL) %
% RSD
TTPC 2000 70 130 30 70 130
D8024 − 23
TABLE 7 MS/MSD QC Acceptance Criteria
NOTE 1—Table 7 data is preliminary until a multi-lab validation study is completed.
MS/MSD Precision
Spike
Analyte Conc. Recovery (%)
RPD (%)
ng/L
Lower Limit Upper Limit
TTPC 2000 70 130 30
TABLE 8 Single-Laboratory Recovery Data in Water
Reagent Water P&A Data CRW P&A Data
(2000 ng/L spike) (2000 ng/L spike)
Sample
TTPC TTPC
MB 1
MB 2
P&A 1 1861 1898
P&A 2 1891 1965
P&A 3 1882 1933
P&A 4 1894 1871
P&A 5 1975 1929
P&A 6 1975 1974
Average Recovery (ng/L) 1893 1928
% Average Recovery 94.6 96.4
Standard Deviation 42.8 39.1
RSD (%) 2.26 2.03
NOTE 2—The use of PTFE, PVDF and polypropylene filter units resulted in poor performance and low recoveries.
7. Apparatus
7.1 LC/MS/MS System:
7.1.1 Liquid Chromatography SystemSystem——A complete LC system is required in order to analyze samples, this should
include a sample injection system, a solvent pumping system capable of mixing solvents, a sample compartment capable of
maintaining required temperature and a temperature controlled column compartment. A LC system that is capable of performing
at the flows, pressures, controlled temperatures, sample volumes, and requirements of this test method shallmust be used.
7.1.2 Analytical Column —A reverse phase C18 particle column was used to develop this test method. Any column that achieves
adequate resolution may be used. The retention times and order of elution may change depending on the column used and need
to be monitored.
7.2 Tandem Mass Spectrometer SystemSystem——A MS/MS system capable of multiple reaction monitoring (MRM) analysis or
any system that is capable of meeting the requirements in this test method shallmust be used.
7.3 Adjustable Volume Pipettes—10, 20, 100, and 1000 μL and 5 and 10 mL.10 μL, 20 μL, 100 μL, 1000 μL, 5 mL, and 10 mL.
NOTE 3—Any pipette may be used providing the data generated meets the performance of this test method.
7.3.1 Pipette Tips—Polypropylene pipette tips free of release agents or low retention coating of various sizes.
7.4 Class A Volumetric Glassware.
7.5 Filtration Device.
A Waters Acquity UPLC BEH C18, 2.12.1 mm × 100 mm and 1.7 μm particle size column, or equivalent, has been found suitable for use. It was used to develop this
test method and generate the precision and biascolumn was used. If you are aware of an alternative column that meets the performance of the standard, please provide this
information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical committee, data presented in
Section which you may attend.16.
D8024 − 23
7.5.1 Hypodermic Syringe—A Luer-Lock tip glass or polypropylene syringe capable of holding a syringe driven filter unit.
7.5.2 A 1010 mL or 20 mL Lock Tip Syringe size is recommended since a 10 mL prepared sample size is used in this test method.
If a smaller volume syringe is used, do not wash out the syringe or change filters while filtering the same sample if multiple refills
of the syringe is required in order to filter the 10 mL 10 mL prepared sample.
7.5.3 Filter Unit —Nylon filter units were used to filter the samples.
7.6 Vials—2-mL autosampler vials (LC vials) with pre-slit PTFE/silicone septa or equivalent.
7.7 VOA Vials—40 mL.
7.8 Sonicator.
7.9 Oven, capable to achieve 250°C.250 °C.
8. Reagents and Materials
8.1 Purity of Reagents—High Performance Liquid Chromatography (HPLC) pesticide residue analysis and spectrophotometry
grade chemicals shallmust be used in all tests. Unless indicated otherwise, it is intended that all reagents shallmust conform to the
Committee on Analytical Reagents of the American Chemical Society. Other reagent grades may be used provided they are first
determined to be of sufficiently high purity to permit their use without affecting the accuracy of the measurements.
8.2 Purity of Water—Unless otherwise indicated, references to water shallmust be understood to mean reagent water conforming
to Type 1 of Specification D1193. It shallmust be demonstrated that this water does not contain contaminants at concentrations
sufficient to interfere with the analysis.
8.3 All prepared solutions are routinely replaced every year if not previously discarded for quality control failure.
8.4 Gases—Ultrapure nitrogen and argon.
8.5 Acetone (CAS # 67-64-1).
8.6 Acetonitrile (CAS # 75-05-8).
8.7 Methanol (CAS # 67-56-1).
8.8 Ammonium Acetate (CAS # 631-61-8).
8.9 2-Propanol (isopropyl alcohol, CAS # 67-63-0).
8.10 Ottawa Sand (CAS # 14808-60-7).
8.11 (Tri-n-butyl)-n-tetradecylphosphonium chloride (CAS # 81741-28-8).
8.12 (Tri-n-butyl)-n-tetradecylphosphonium bromide –D29 (TTPBr-D29, CAS # NA, optional surrogate).
A Whatman Puradisc 25 NYL Disposable Filter unit (diameter 25 mm, 0.2 μm nylon membrane syringe driven filter unit) has been found suitable for use for this method.
Any filter unit may be used was used. If you are aware of an alternative filter that meets the performance of this method. the standard, please provide this information to
ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical committee, which you may attend.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
D8024 − 23
9. Hazards
9.1 Normal laboratory safety applies to this method. Analysts should wear safety glasses, gloves, and lab coats when working in
the lab. Analysts should review the Safety Data Sheets (SDS) for all reagents used in this method.
10. Sampling and Preservation
10.1 Grab samples are collected in glass containers with Tefloninert-lined-lined caps. As part of the overall quality assurance
program for this test method, field blanks exposed to the same field conditions as samples are collected and analyzed according
to this test method to assess the potential for field contamination, refer to Guide D3856 as a guide for sampling. This test method
is based on a 2.5 mL sample size per analysis. If different sample sizes are used, spiking solution amounts may need to be modified.
EPA Publication SW-8466 may be used as a sampling guide. Samples shallmust be shipped with a trip blank and at less than
6°C.between above freezing and 6 °C.
10.2 Once received the sample temperature is taken and should be less than 6°C.6 °C. If the receiving temperature is greater than
6°C,6 °C, the sample temperature is noted in the case narrative accompanying the data. Samples should be stored refrigerated
between 0°C and 6°Cabove freezing and 6 °C from the time of collection until analysis.
10.3 The sample should be analyzed within 14 days of collection. No holding time study has been done on water matrices tested
in this test method. Holding time may vary depending on the matrix and individual laboratories should determine the holding time
in their matrix, refer to Practice D4841.
11. Preparation of LC/MS/MS
11.1 LC Chromatograph Operating Conditions:
11.1.1 Injections of all standards and samples are made at a 50 μL volume. Other injection volumes may be used to optimize
conditions. Standards and sample extracts shallmust be in a 75:25 acetone:water solution. In the case of extreme concentration
differences amongst samples, it is wise to analyze a blank after a concentrated sample and before a dilute sample to minimize
carry-over of analytes from injection to injection. However, there should not be carry-over between samples. The LC utilized to
develop this test method has a flow through LC needle design. The gradient conditions for liquid chromatography are shown in
Table 2.
11.2 LC Sample Manager Conditions:
11.2.1 Needle Wash Solvent—60 %60 % acetonitrile acetonitrile/40 % ⁄40 % 2-propanol. Eight-second wash time before and after
injection. Instrument manufacturer’s specifications should be followed in order to eliminate sample carry-over.
11.2.2 Temperatures—Column, 35°C;35 °C; sample compartment, 15°C.15 °C.
11.2.3 Seal Wash—Solvent: 50 %50 % methanol methanol/5 0 % ⁄50 % water; time: 5 minutes.5 min.
11.3 Mass Spectrometer Parameters:
11.3.1 To acquire the maximum number of data points per SRM channel while maintaining adequate sensitivity, the tune
parameters may be optimized according to the instrument used. Each peak requires at least 10 scans per peak for adequate
quantitation. Variable parameters regarding SRM transitions, and cone and collision energies are shown in Table 3. The values for
the following parameters are shown here for information only. These conditions should be checked and optimized when required.
Mass spectrometer parameters used in the development of this method are listed below:
The instrument is set in the Electrospray positive source setting:
Capillary Voltage: 1 kV
Cone: Variable depending on analyte
Source Temperature: 150°C
Source Temperature: 150 °C
Desolvation Gas Temperature: 500°C
Desolvation Gas Temperature: 500 °C
Desolvation Gas Flow: 900 L/hr
D8024 − 23
Desolvation Gas Flow: 900 L/h
Cone Gas Flow: 150 L/hr
Cone Gas Flow: 150 L/h
Collision Gas Flow: 0.15 mL/min
Low Mass Resolution 1: 3
Low Mass Resolution 1:3
High Mass Resolution 1: 14
High Mass Resolution 1:14
Ion Energy 1: 1
Ion Energy 1:1
Entrance Ener
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...