ASTM D4282-15(2022)
(Test Method)Standard Test Method for Determination of Free Cyanide in Water and Wastewater by Microdiffusion
Standard Test Method for Determination of Free Cyanide in Water and Wastewater by Microdiffusion
SIGNIFICANCE AND USE
5.1 This test method is useful in distinguishing between the potentially available free cyanide (total cyanide) and the free cyanide actually present.
5.2 This test method provides a convenient technique for making on-site free cyanide determinations.
SCOPE
1.1 This test method covers the determination of free cyanides in waters and wastewaters. Free cyanide is here defined as the cyanide which diffuses as cyanide (HCN), at room temperature, from a solution at pH 6.2
1.2 This test method does not include complexes that resist dissociation, such as hexacyanoferrates and gold cyanide, nor does it include thiocyanate and cyanohydrin.
1.3 This test method may be applied to water and wastewater samples containing free cyanide from 10 to 150 μg/L. Greater concentrations may be determined by appropriate dilution.
1.4 This test method has been fully validated by collaborative testing as specified by Practice D2777.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see 8.6, 8.9, Section 9, and 12.2.1.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2022
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-May-2020
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Refers
ASTM E275-08 - Describing and Measuring Performance of Ultraviolet and Visible Spectrophotometers - Effective Date
- 15-Oct-2008
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D4282-15(2022): Standard Test Method for Determination of Free Cyanide in Water and Wastewater by Microdiffusion provides a reliable and validated procedure for measuring the concentration of free cyanide in water and wastewater samples. The method specifically targets free cyanide species capable of diffusing as hydrogen cyanide (HCN) at room temperature and at pH 6.2. It does not include cyanide complexes that are resistant to dissociation, such as hexacyanoferrates and gold cyanide, nor does it measure thiocyanate or cyanohydrin.
This test method is intended for both laboratory and on-site applications, facilitating convenient and accurate free cyanide determinations. The test is applicable for samples with free cyanide concentrations ranging from 10 to 150 μg/L, with higher concentrations measurable following suitable dilution.
Key Topics
- Definition of Free Cyanide: Free cyanide is defined as those cyanide forms that can diffuse as HCN from an aqueous solution at pH 6 and room temperature. This distinction is important for environmental monitoring and regulatory compliance.
- Scope of Application: The method is applicable to various water matrices, including drinking water, surface water, and wastewater.
- Exclusions: The test does not measure strongly bound cyanide complexes like hexacyanoferrates or organocyanide compounds.
- Microdiffusion Principle: The test employs a microdiffusion cell which separates free cyanide from the sample matrix, allowing it to be trapped and subsequently quantified.
- Detection and Calibration: Colorimetric determination is performed using spectrophotometry. Calibration with known standards ensures the accuracy and precision of the results.
- Precision and Validation: The method has been collaboratively tested and validated as specified by ASTM Practice D2777.
- Quality Control: The standard emphasizes regular calibration, duplicate testing, and running blanks for reliable quality assurance and analytical performance.
- Safety: Given the toxicity of cyanide and certain reagents (e.g., pyridine, cadmium chloride), the standard outlines essential health, safety, and environmental precautions.
Applications
The ASTM D4282-15(2022) standard is vital for:
- Environmental Monitoring: Ensuring regulatory compliance for surface water, groundwater, and effluent discharge by determining free cyanide concentrations accurately.
- Wastewater Treatment Facilities: Routine monitoring of process streams and effluents to assess the effectiveness of cyanide removal processes and to maintain safe operating conditions.
- Industrial Water Quality Management: Supporting industries such as mining, plating, and chemical manufacturing in tracking and minimizing free cyanide releases.
- On-Site Testing: The method’s portability and ease-of-use make it practical for field engineers and environmental professionals conducting on-the-spot analyses.
- Research and Development: Facilitating studies into cyanide behavior in aquatic systems and treatment processes.
Related Standards
Understanding and implementing ASTM D4282-15(2022) may require reference to related ASTM standards and guides, such as:
- ASTM D1129: Terminology Relating to Water
- ASTM D1193: Specification for Reagent Water
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods
- ASTM D3370: Practices for Sampling Water from Flowing Process Streams
- ASTM D3856: Guide for Management Systems in Laboratories Engaged in Analysis of Water
- ASTM D4210: Practice for Intralaboratory Quality Control Procedures and a Discussion on Reporting Low-Level Data
- ASTM E275: Practice for Describing and Measuring Performance of Ultraviolet and Visible Spectrophotometers
Practical Value
Adopting ASTM D4282-15(2022) enables water and wastewater laboratories, as well as field teams, to reliably quantify free cyanide in compliance with regulatory requirements. Its robust validation and detailed quality control procedures help ensure accurate reporting, vital for public health, environmental protection, and industrial process control. The standard supports broader water quality initiatives by providing a clear, consistent method for one of the most critical water contaminants.
Keywords: ASTM D4282-15(2022), standard test method, free cyanide, microdiffusion, water, wastewater, environmental monitoring, cyanide analysis, spectrophotometer, quality control, laboratory, field testing.
Buy Documents
ASTM D4282-15(2022) - Standard Test Method for Determination of Free Cyanide in Water and Wastewater by Microdiffusion
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D4282-15(2022) is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Free Cyanide in Water and Wastewater by Microdiffusion". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is useful in distinguishing between the potentially available free cyanide (total cyanide) and the free cyanide actually present. 5.2 This test method provides a convenient technique for making on-site free cyanide determinations. SCOPE 1.1 This test method covers the determination of free cyanides in waters and wastewaters. Free cyanide is here defined as the cyanide which diffuses as cyanide (HCN), at room temperature, from a solution at pH 6.2 1.2 This test method does not include complexes that resist dissociation, such as hexacyanoferrates and gold cyanide, nor does it include thiocyanate and cyanohydrin. 1.3 This test method may be applied to water and wastewater samples containing free cyanide from 10 to 150 μg/L. Greater concentrations may be determined by appropriate dilution. 1.4 This test method has been fully validated by collaborative testing as specified by Practice D2777. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see 8.6, 8.9, Section 9, and 12.2.1. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is useful in distinguishing between the potentially available free cyanide (total cyanide) and the free cyanide actually present. 5.2 This test method provides a convenient technique for making on-site free cyanide determinations. SCOPE 1.1 This test method covers the determination of free cyanides in waters and wastewaters. Free cyanide is here defined as the cyanide which diffuses as cyanide (HCN), at room temperature, from a solution at pH 6.2 1.2 This test method does not include complexes that resist dissociation, such as hexacyanoferrates and gold cyanide, nor does it include thiocyanate and cyanohydrin. 1.3 This test method may be applied to water and wastewater samples containing free cyanide from 10 to 150 μg/L. Greater concentrations may be determined by appropriate dilution. 1.4 This test method has been fully validated by collaborative testing as specified by Practice D2777. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see 8.6, 8.9, Section 9, and 12.2.1. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4282-15(2022) is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4282-15(2022) has the following relationships with other standards: It is inter standard links to ASTM D5788-95(2024), ASTM D1129-13(2020)e2, ASTM D5788-95(2017), ASTM D2777-12, ASTM D3856-11, ASTM D5788-95(2011), ASTM D3370-10, ASTM D1129-10, ASTM E275-08, ASTM D3370-08, ASTM D2777-08, ASTM D3370-07, ASTM D1129-06ae1, ASTM D1129-06a, ASTM D2777-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4282-15(2022) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4282 − 15 (Reapproved 2022)
Standard Test Method for
Determination of Free Cyanide in Water and Wastewater by
Microdiffusion
This standard is issued under the fixed designation D4282; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This test method covers the determination of free 2.1 ASTM Standards:
cyanides in waters and wastewaters. Free cyanide is here D1129Terminology Relating to Water
defined as the cyanide which diffuses as cyanide (HCN), at D1192Guide for Equipment for Sampling Water and Steam
2 4
room temperature, from a solution at pH 6. in Closed Conduits (Withdrawn 2003)
D1193Specification for Reagent Water
1.2 This test method does not include complexes that resist
D2777Practice for Determination of Precision and Bias of
dissociation, such as hexacyanoferrates and gold cyanide, nor
Applicable Test Methods of Committee D19 on Water
does it include thiocyanate and cyanohydrin.
D3370Practices for Sampling Water from Flowing Process
1.3 This test method may be applied to water and wastewa-
Streams
ter samples containing free cyanide from 10 to 150 µg/L.
D3856Guide for Management Systems in Laboratories
Greater concentrations may be determined by appropriate
Engaged in Analysis of Water
dilution.
D4210Practice for Intralaboratory Quality Control Proce-
dures and a Discussion on Reporting Low-Level Data
1.4 This test method has been fully validated by collabora-
tive testing as specified by Practice D2777. (Withdrawn 2002)
D5788Guide for Spiking Organics into Aqueous Samples
1.5 The values stated in SI units are to be regarded as
D5789Practice for Writing Quality Control Specifications
standard. No other units of measurement are included in this
for Standard Test Methods for Organic Constituents
standard.
(Withdrawn 2002)
1.6 This standard does not purport to address all of the
D5847Practice for Writing Quality Control Specifications
safety concerns, if any, associated with its use. It is the
for Standard Test Methods for Water Analysis
responsibility of the user of this standard to establish appro-
E275PracticeforDescribingandMeasuringPerformanceof
priate safety, health, and environmental practices and deter-
Ultraviolet and Visible Spectrophotometers
mine the applicability of regulatory limitations prior to use.
For specific hazard statements, see 8.6, 8.9, Section 9, and 3. Terminology
12.2.1.
3.1 Definitions:
1.7 This international standard was developed in accor-
3.1.1 For definitions of terms used in this standard, refer to
dance with internationally recognized principles on standard-
Terminology D1129.
ization established in the Decision on Principles for the
3.2 Definitions of Terms Specific to This Standard:
Development of International Standards, Guides and Recom-
3.2.1 free cyanide—those simple cyanides or loosely held
mendations issued by the World Trade Organization Technical
cyanide complexes that diffuse at pH 6, at room temperature.
Barriers to Trade (TBT) Committee.
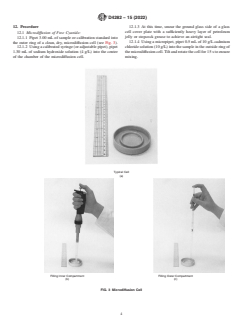
4. Summary of Test Method
4.1 The reactions are carried out in a microdiffusion cell.
This test method is under the jurisdiction ofASTM Committee D19 on Water
andisthedirectresponsibilityofSubcommitteeD19.06onMethodsforAnalysisfor
4.2 The sample is treated with cadmium ion to precipitate
Organic Substances in Water.
the hexacyanoferrates.
Current edition approved May 1, 2022. Published May 2022. Originally
approved in 1983. Last previous edition approved in 2015 as D4282–15. DOI:
10.1520/D4282-15R22. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
The paper by J. M. Kruse and L. E. Thibault, “Determination of Free Cyanide contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
in Ferro- and Ferricyanides,”Analytical Chemistry, 45(13): 2260–2261; 1973 Nov., Standards volume information, refer to the standard’s Document Summary page on
recommendsadiffusionatpH7.TheANSImodification(ANSIPH4.41-1978)uses the ASTM website.
pH 6. Using the conditions of the ANSI method, diffusion is completed within 4 The last approved version of this historical standard is referenced on
hoursatpH6.LongerdiffusiontimewasrequiredatpH7onthesamplesanalyzed. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4282 − 15 (2022)
4.3 The sample is buffered at pH 6 and allowed to stand for used provided it is first ascertained that the reagent is of
4h. sufficient purity to permit its use without lessening the accu-
racy of the determination.
4.4 The HCN diffuses into sodium hydroxide solution.
8.2 Purity of Water—Unless otherwise indicated, reference
4.5 An aliquot of the sodium hydroxide solution is treated
towatershallbeunderstoodtomeanreagentwaterconforming
with chloramine-T, and the cyanogen chloride formed is
to Type II of Specification D1193.
reacted with barbituric acid in pyridine. The absorbance of the
color formed is measured using a spectrophotometer at a 8.3 Cadmium Chloride Solution (10 g/L), CdCl —Dissolve
wavelength of 580 nm. 10.0 g of anhydrous cadmium chloride in 750 mL of water in
a 1 L volumetric flask. Dilute to volume with water.
5. Significance and Use
8.4 Chloramine-T Reagent (10 g/L)—Dissolve 1.00 g of
5.1 This test method is useful in distinguishing between the
chloramine-Tin 50 mLof water in a 100 mLvolumetric flask.
potentially available free cyanide (total cyanide) and the free
Dilute to volume with water. Make this reagent fresh daily.
cyanide actually present.
−
8.5 Cyanide Solution, Standard (1.00 mL=2 µg CN )
5.2 This test method provides a convenient technique for
—Pipet 2.00 mL of cyanide stock solution (approximately 1.0
−
making on-site free cyanide determinations.
g/LCN )intoa1Lvolumetricflaskanddilutetovolumewith
sodium hydroxide solution (2.05 g/L).
6. Interferences
8.6 Cyanide Solution Stock—Dissolve 2.51 g of potassium
6.1 Decomposition of Hexacyanoferrates During Diffusion:
cyanide, KCN, in 500 mL of sodium hydroxide solution (2.05
6.1.1 This decomposition is virtually eliminated by allow-
g/L) ina1L volumetric flask. Dilute to volume with sodium
ing the sample to diffuse in the dark, and by precipitating the
hydroxide solution (2.05 g/L). This solution contains approxi-
hexacyanoferrates with cadmium ion.
−
mately 1.0 g/L cyanide (CN ). (Warning—KCN is highly
6.2 Instability of Free Cyanide in Effluents—The reactivity toxic, avoid contact or inhalation. Prepare and standardize this
of free cyanide with such chemicals as aldehydes or oxidizing
solution weekly.)
agents, is not really a method interference. However, because 8.6.1 Standardizing Cyanide Stock Solution:
of this instability, it is important for the diffusion to begin as
8.6.1.1 Using a silver electrode and a reference electrode,
soon after sampling as possible. It is beyond the scope of this titrate 20.0 mL of the cyanide stock solution (in a beaker also
testmethodtolistallthepossiblecyanidereactionsthatmaybe
containing 50 mL of sodium hydroxide solution (2.05 g/L))
encountered. with the silver nitrate standard solution.
8.6.1.2 Record the mLof titration for use in the calculation
7. Apparatus
(see Fig. 1 for an example of a typical titration curve).
8.6.1.3 Calculate the concentration of the cyanide stock
7.1 Diffusion Cell, microdiffusion cell, Conway type, 68
solution using the following equation:
mm outside diameter.
50 3 mLsilvernitrate 5mg/L CN instocksolution
~ !
7.2 Micropipets, 0.10 mL, 1.00 mL.
1.00mLofsilvernitratesolutionisequalto1 mgofCN .
7.3 Spectrophotometer, conforming to Practice E275.
8.7 Potassium Phosphate Buffer Solution (Acidified)—Add
7.4 Spectrophotometer Cell, 1 cm equipped with a stopper.
8.0mLofconcentratedphosphoricacid(spgr1.69),H PO,to
3 4
7.5 Pipet or Syringe, adjustable (to deliver 1.30 mL).
100 mL of potassium phosphate solution.
7.6 Calomel Reference Electrode, with saturated KNO
electrolyte, or the equivalent.
7.7 pH Meter.
7.8 Silver Electrode.
8. Reagents
8.1 Purity of Reagents—Reagent grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that
reagents shall conform to the specifications of the Committee
on Analytical Reagents of the American Chemical Society
where such specifications are available. Other grades may be
One source of supply for these cells is Arthur H. Thomas, No. 3806-F-10.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
NOTE 1—Twenty millilitres of 2.51 g/L KCN titrated with AgNO .
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. FIG. 1 Typical Titration Curve Standardizing KCN Solution
D4282 − 15 (2022)
8.8 Potassium Phosphate Solution, 190 g/L—Add 400 mL 9.2.5 Make the seal between the microdiffusion cell and lid
of water toa2L beaker. Add and dissolve 14.5 g of sodium airtight.
hydroxide, NaOH. Add and dissolve 190 g of potassium 9.2.6 It is important to observe the specified time periods in
phosphate, monobasic, KH PO . Add water to 950 mL to aid those steps where such is noted. In particular, make the
2 4
dissolution. Adjust the pH of the solution to pH 5.9 to 6.1, spectrophotometer measurements in the 3 to 6 min interval.
using100g/Lsodiumhydroxidesolution.Transferthesolution 9.2.7 Full color development in the spectrophotometer cell
toa1L volumetric flask, and dilute to volume with water. requires that after each addition, mix the solution thoroughly
without loss of material.
8.9 Pyridine-Barbituric Acid Reagent—Add 15.0 g of bar-
bituricacidtoa250mLvolumetricflask.Washdownthesides
10. Sampling and Sample Preservation
of the flask with just enough water to moisten the barbituric
10.1 Collect the sample in accordance with Guide D1192
acid. Add 75 mL of pyridine and swirl to mix. Slowly add
and Practices D3370.
15mLofconcentratedhydrochloricacid(spgr1.19)andswirl
to mix. Cool the solution to room temperature. Dilute to
10.2 A satisfactory preservation technique is not available.
−
volume and mix. It is recommended that this reagent be
Reactions between CN and aldehydes, oxidizing agents, or
prepared fresh weekly and stored in a dark place. (Warning—
sulfides will continue. However, if the sample cannot be
Pyridine is toxic; avoid contact or inhalation. Prepare this
analyzed immediately, some steps can be taken to slow down
reagent in an exhaust hood.)
the reactions taking place.
10.2.1 Adjust the sample to pH 12 or more.This minimizes
8.10 Silver Nitrate Solution, Standard (1 mL=1 mg of
−
−
CN losses due to vaporization.
CN )—Weigh 3.2647 g of silver nitrate on an analytical
10.2.2 Store the samples in the dark to prevent hexacyano-
balance. Quantitatively transfer the silver nitrate toa1L
ferrate breakdown.
volumetric flask. Dissolve and dilute to volume with water.
10.2.3 Keepthesamplecool(forexample,inarefrigerator).
Store in a dark glass bottle.
8.11 Sodium Hydroxide Solution (4.1 g/L), NaOH—Add
11. Calibration
4.10 g of sodium hydroxide to 800 mL of water ina1L
11.1 Calibration Standards—Pipet 0.00 mL (Note 2),
volumetric flask. Stir until dissolved, and cool the solution to
5.00mL, 10.0mL, and 15.0 mL of the 2.00 mg/L cyanide
room temperature before adjusting the final volume to 1 L.
standard solution into four 200 mL volumetric flasks. Dilute
8.12 Sodium Hydroxide Solution (2.05 g/L), NaOH—Add
each of the flasks to volume with sodium
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...