ASTM D6581-18
(Test Method)Standard Test Methods for Bromate, Bromide, Chlorate, and Chlorite in Drinking Water by Suppressed Ion Chromatography
Standard Test Methods for Bromate, Bromide, Chlorate, and Chlorite in Drinking Water by Suppressed Ion Chromatography
SIGNIFICANCE AND USE
4.1 The oxyhalides chlorite, chlorate, and bromate are inorganic disinfection by-products (DBPs) of considerable health risk concern worldwide. The occurrence of chlorite and chlorate is associated with the use of chlorine dioxide, as well as hypochlorite solutions used for drinking water disinfection. The occurrence of bromate is associated with the use of ozone for disinfection, wherein naturally occurring bromide is oxidized to bromate. Bromide is a naturally occurring precursor to the formation of bromate.
SCOPE
1.1 These multi-test methods cover the determination of the oxyhalides—chlorite, bromate, and chlorate, and bromide, in raw water, finished drinking water and bottled (non-carbonated) water by chemically and electrolytically suppressed ion chromatography. The ranges tested using these test methods for each analyte were as follows:
Range
Sections
Test Method A:
Chemically Suppressed Ion Chromatography
8 to 20
Chlorite
5 to 500 µg/L
Bromate
1 to 25 µg/L
Bromide
5 to 250 µg/L
Chlorate
5 to 500 µg/L
Test Method B:
Electrolytically Suppressed Ion Chromatography
21 to 31
Chlorite
20 to 1000 µg/L
Bromate
1 to 30 µg/L
Bromide
20 to 200 µg/L
Chlorate
20 to 1000 µg/L
1.1.1 The upper limits may be extended by appropriate sample dilution or by the use of a smaller injection volume. Other ions of interest, such as fluoride, chloride, nitrite, nitrate, phosphate, and sulfate may also be determined using these test methods. However, analysis of these ions is not the object of these test methods.
1.2 It is the user’s responsibility to ensure the validity of these test methods for waters of untested matrices.
1.3 These test methods are technically equivalent with Part B of U.S. EPA Method 300.1,2 titled “The Determination of Inorganic Anions in Drinking Water by Ion Chromatography.”
1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2018
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-May-2018
- Effective Date
- 01-May-2020
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
- Effective Date
- 15-Aug-2006
- Effective Date
- 01-Mar-2006
Overview
ASTM D6581-18 is the standard test method developed by ASTM International for detecting and quantifying bromate, bromide, chlorate, and chlorite in drinking water through suppressed ion chromatography. These analytes are considered inorganic disinfection by-products (DBPs) arising from the use of disinfectants like chlorine dioxide, hypochlorite solutions, and ozone in water treatment processes. Monitoring and controlling these oxyhalides is crucial due to their significant health risks, making this test method an essential part of water quality assessment and public safety.
Key Topics
Scope of Analysis:
This standard covers the determination of four key oxyhalides-chlorite, bromate, chlorate, and bromide-in raw water, finished (treated) drinking water, and bottled (non-carbonated) water.Test Methods Included:
- Chemically Suppressed Ion Chromatography:
Suitable for measuring chlorite (5–500 µg/L), bromate (1–25 µg/L), bromide (5–250 µg/L), and chlorate (5–500 µg/L). - Electrolytically Suppressed Ion Chromatography:
Suitable for chlorite (20–1000 µg/L), bromate (1–30 µg/L), bromide (20–200 µg/L), and chlorate (20–1000 µg/L).
- Chemically Suppressed Ion Chromatography:
Extension of Range:
The test methods’ upper detection limits can be increased by appropriate sample dilution or by using a smaller injection volume.Technical Equivalence:
ASTM D6581-18 is technically equivalent to Part B of U.S. EPA Method 300.1, ensuring broad regulatory acceptance and harmonization.Health Significance:
Oxyhalides are monitored because of their association with disinfection by-products, which pose global health risks. For example, ozone disinfection may convert naturally occurring bromide into the potentially harmful bromate.
Applications
Regulatory Compliance:
Water treatment facilities, regulatory agencies, and bottled water manufacturers use this standard method to verify compliance with national and international drinking water quality regulations concerning DBPs.Quality Assurance:
Laboratories and water utilities implement ASTM D6581-18 for regular monitoring of disinfection by-products to ensure consumer safety and maintain water quality certifications.Source and Process Water Assessment:
The standard enables assessment of raw, process, and finished water at each stage of production or treatment, supporting proactive detection and remediation of contamination.Research and Environmental Monitoring:
Researchers and environmental health professionals use this method for studying DBP formation, treatment efficacy, and the impact of various disinfection techniques.Flexible Analysis:
In addition to its primary targets, the method allows for the optional determination of other ions such as fluoride, chloride, nitrite, nitrate, phosphate, and sulfate, facilitating comprehensive ionic profiling in water samples.
Related Standards
- U.S. EPA Method 300.1 - Determination of inorganic anions in drinking water by ion chromatography (technical equivalent).
- ASTM D1129 - Terminology relating to water.
- ASTM D1193 - Specification for reagent water.
- ASTM D2777 - Precision and bias of Committee D19 test methods.
- ASTM D3370 - Practices for sampling water from closed conduits.
- ASTM D3856 - Guide for management systems in water analysis laboratories.
- ASTM D5810 - Guide for spiking into aqueous samples.
- ASTM D5847 - Writing quality control specifications for water analysis test methods.
Practical Value
Implementing ASTM D6581-18 provides laboratories and utilities with a robust, validated, and internationally recognized procedure for routinely measuring critical oxyhalide by-products in drinking water. This standard supports risk assessment, regulatory compliance, and quality assurance, helping safeguard public health and meet both local and global water quality standards. By employing suppressed ion chromatography as specified, organizations benefit from reliable detection with precision, consistency, and compatibility with regulatory frameworks.
Buy Documents
ASTM D6581-18 - Standard Test Methods for Bromate, Bromide, Chlorate, and Chlorite in Drinking Water by Suppressed Ion Chromatography
REDLINE ASTM D6581-18 - Standard Test Methods for Bromate, Bromide, Chlorate, and Chlorite in Drinking Water by Suppressed Ion Chromatography
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D6581-18 is a standard published by ASTM International. Its full title is "Standard Test Methods for Bromate, Bromide, Chlorate, and Chlorite in Drinking Water by Suppressed Ion Chromatography". This standard covers: SIGNIFICANCE AND USE 4.1 The oxyhalides chlorite, chlorate, and bromate are inorganic disinfection by-products (DBPs) of considerable health risk concern worldwide. The occurrence of chlorite and chlorate is associated with the use of chlorine dioxide, as well as hypochlorite solutions used for drinking water disinfection. The occurrence of bromate is associated with the use of ozone for disinfection, wherein naturally occurring bromide is oxidized to bromate. Bromide is a naturally occurring precursor to the formation of bromate. SCOPE 1.1 These multi-test methods cover the determination of the oxyhalides—chlorite, bromate, and chlorate, and bromide, in raw water, finished drinking water and bottled (non-carbonated) water by chemically and electrolytically suppressed ion chromatography. The ranges tested using these test methods for each analyte were as follows: Range Sections Test Method A: Chemically Suppressed Ion Chromatography 8 to 20 Chlorite 5 to 500 µg/L Bromate 1 to 25 µg/L Bromide 5 to 250 µg/L Chlorate 5 to 500 µg/L Test Method B: Electrolytically Suppressed Ion Chromatography 21 to 31 Chlorite 20 to 1000 µg/L Bromate 1 to 30 µg/L Bromide 20 to 200 µg/L Chlorate 20 to 1000 µg/L 1.1.1 The upper limits may be extended by appropriate sample dilution or by the use of a smaller injection volume. Other ions of interest, such as fluoride, chloride, nitrite, nitrate, phosphate, and sulfate may also be determined using these test methods. However, analysis of these ions is not the object of these test methods. 1.2 It is the user’s responsibility to ensure the validity of these test methods for waters of untested matrices. 1.3 These test methods are technically equivalent with Part B of U.S. EPA Method 300.1,2 titled “The Determination of Inorganic Anions in Drinking Water by Ion Chromatography.” 1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The oxyhalides chlorite, chlorate, and bromate are inorganic disinfection by-products (DBPs) of considerable health risk concern worldwide. The occurrence of chlorite and chlorate is associated with the use of chlorine dioxide, as well as hypochlorite solutions used for drinking water disinfection. The occurrence of bromate is associated with the use of ozone for disinfection, wherein naturally occurring bromide is oxidized to bromate. Bromide is a naturally occurring precursor to the formation of bromate. SCOPE 1.1 These multi-test methods cover the determination of the oxyhalides—chlorite, bromate, and chlorate, and bromide, in raw water, finished drinking water and bottled (non-carbonated) water by chemically and electrolytically suppressed ion chromatography. The ranges tested using these test methods for each analyte were as follows: Range Sections Test Method A: Chemically Suppressed Ion Chromatography 8 to 20 Chlorite 5 to 500 µg/L Bromate 1 to 25 µg/L Bromide 5 to 250 µg/L Chlorate 5 to 500 µg/L Test Method B: Electrolytically Suppressed Ion Chromatography 21 to 31 Chlorite 20 to 1000 µg/L Bromate 1 to 30 µg/L Bromide 20 to 200 µg/L Chlorate 20 to 1000 µg/L 1.1.1 The upper limits may be extended by appropriate sample dilution or by the use of a smaller injection volume. Other ions of interest, such as fluoride, chloride, nitrite, nitrate, phosphate, and sulfate may also be determined using these test methods. However, analysis of these ions is not the object of these test methods. 1.2 It is the user’s responsibility to ensure the validity of these test methods for waters of untested matrices. 1.3 These test methods are technically equivalent with Part B of U.S. EPA Method 300.1,2 titled “The Determination of Inorganic Anions in Drinking Water by Ion Chromatography.” 1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D6581-18 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6581-18 has the following relationships with other standards: It is inter standard links to ASTM D6581-12, ASTM D1129-13(2020)e2, ASTM D2777-12, ASTM D3856-11, ASTM D5810-96(2011), ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D2777-08, ASTM D3370-07, ASTM D1129-06ae1, ASTM D1129-06a, ASTM D2777-06, ASTM D5810-96(2006), ASTM D1193-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6581-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6581 − 18
Standard Test Methods for
Bromate, Bromide, Chlorate, and Chlorite in Drinking Water
by Suppressed Ion Chromatography
This standard is issued under the fixed designation D6581; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* conversions to inch-pound units that are provided for informa-
tion only and are not considered standard.
1.1 These multi-test methods cover the determination of the
1.5 This standard does not purport to address all of the
oxyhalides—chlorite, bromate, and chlorate, and bromide, in
safety concerns, if any, associated with its use. It is the
raw water, finished drinking water and bottled (non-
responsibility of the user of this standard to establish appro-
carbonated) water by chemically and electrolytically sup-
priate safety, health, and environmental practices and deter-
pressed ion chromatography.The ranges tested using these test
mine the applicability of regulatory limitations prior to use.
methods for each analyte were as follows:
1.6 This international standard was developed in accor-
Range Sections
Test Method A: 8 to 20 dance with internationally recognized principles on standard-
Chemically Suppressed
ization established in the Decision on Principles for the
Ion Chromatography
Development of International Standards, Guides and Recom-
Chlorite 5 to 500 µg/L
Bromate 1 to 25 µg/L mendations issued by the World Trade Organization Technical
Bromide 5 to 250 µg/L
Barriers to Trade (TBT) Committee.
Chlorate 5 to 500 µg/L
2. Referenced Documents
Test Method B: 21 to 31
Electrolytically 3
2.1 ASTM Standards:
Suppressed Ion
D1129Terminology Relating to Water
Chromatography
Chlorite 20 to 1000 µg/L
D1193Specification for Reagent Water
Bromate 1 to 30 µg/L
D2777Practice for Determination of Precision and Bias of
Bromide 20 to 200 µg/L
Applicable Test Methods of Committee D19 on Water
Chlorate 20 to 1000 µg/L
D3370Practices for Sampling Water from Closed Conduits
1.1.1 The upper limits may be extended by appropriate
D3856Guide for Management Systems in Laboratories
sample dilution or by the use of a smaller injection volume.
Engaged in Analysis of Water
Otherionsofinterest,suchasfluoride,chloride,nitrite,nitrate,
D5810Guide for Spiking into Aqueous Samples
phosphate, and sulfate may also be determined using these test
D5847Practice for Writing Quality Control Specifications
methods. However, analysis of these ions is not the object of
for Standard Test Methods for Water Analysis
these test methods.
1.2 It is the user’s responsibility to ensure the validity of 3. Terminology
these test methods for waters of untested matrices.
3.1 Definitions:
1.3 These test methods are technically equivalent with Part 3.1.1 For definitions of terms used in this standard, refer to
Terminology D1129.
B of U.S. EPA Method 300.1, titled “The Determination of
InorganicAnions in Drinking Water by Ion Chromatography.”
3.2 Definitions of Terms Specific to This Standard:
3.2.1 analytical column, n—the ion exchange column used
1.4 The values stated in SI units are to be regarded as
standard. The values given in parentheses are mathematical to separate the ions of interest according to their retention
characteristics prior to detection.
3.2.2 analytical column set, n—a combination of one or
1 more guard columns, followed by one or more analytical
These test methods are under the jurisdiction of ASTM Committee D19 on
Water and are the direct responsibility of Subcommittee D19.05 on Inorganic
Constituents in Water.
Current edition approved May 1, 2018. Published May 2018. Originally For referenced ASTM standards, visit the ASTM website, www.astm.org, or
approved in 2000. Last previous edition approved in 2012 as D6581–12. DOI: contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
10.1520/D6581-18. Standards volume information, refer to the standard’s Document Summary page on
U.S. EPA 300.1, Cincinnati, OH, 1997. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6581 − 18
columns used to separate the ions of interest. All of the sufficiently high purity to permit its use without reducing the
columns in series then contribute to the overall capacity and accuracy of the determination.
resolution of the analytical column set.
5.2 Purity of Water—Unless otherwise indicated, references
3.2.3 eluent, n—theionicmobilephaseusedtotransportthe
towatershallbeunderstoodtomeanreagentwaterconforming
sample through the chromatographic system.
to Specification D1193,Type I. Other reagent water types may
be used, provided it is first ascertained that the water is of
3.2.4 guard column, n—a column used before the analytical
sufficiently high purity to permit its use without adversely
column to protect it from contaminants, such as particulates or
affecting the bias and precision of the determination.
irreversibly retained material.
3.2.5 ion chromatography, n—a form of liquid chromatog-
6. Precautions
raphy in which ionic constituents are separated by ion ex-
change then detected by an appropriate detection means, 6.1 These test methods address the determination of very
typically conductance. low concentrations of selected anions. Accordingly, every
precaution should be taken to ensure the cleanliness of sample
3.2.6 resolution, n—the ability of an analytical column to
containersaswellasothermaterialsandapparatusthatcomein
separate the method analytes under specific test conditions.
contact with the sample.
3.2.7 suppressor device, n—an ion exchange based device
that is placed between the analytical column set and the
7. Sampling and Sample Preservation
conductivity detector. Its purpose is to minimize detector
response to the ionic constituents in the eluent, in order to 7.1 Collect the sample in accordance with Practices D3370,
as applicable.
lower background conductance; and at the same time enhance
the conductivity detector response of the ions of interest.
7.2 Immediately upon taking the sample, sparge it with an
3.2.7.1 chemical suppression, n—theuseofanacidsolution
inert gas (for example, nitrogen, argon, or helium) for 5
to the suppressor in order to suppress the background conduc-
minutes to remove active gases such as chlorine dioxide or
tivity.
ozone. Add 1.00 mL of EDA Preservation Solution (see 15.3)
per 1.000 litre of sample to prevent conversion of residual
3.2.7.2 electrolytic suppressor device, n—electrolytic sup-
hypochlorite or hypobromite to chlorate or bromate. This also
pression is an ion exchange device that is placed between the
prevents metal catalyzed conversion of chlorite to chlorate.
analytical column and the conductivity detector. Its purpose is
The oxyhalides in samples preserved in this manner are stable
similar to a suppressor device, however, it does not require
for at least 14 days when stored in amber bottles at 4°C.
addition of acid. Instead the electrolytic suppressor generates
protons electrolytically and plugs into an electrical power
source on typically located on the chromatography device.
TEST METHOD A
CHEMICALLY SUPPRESSED ICON
4. Significance and Use
CHROMATOGRAPHY
4.1 The oxyhalides chlorite, chlorate, and bromate are
inorganic disinfection by-products (DBPs) of considerable
8. Scope
health risk concern worldwide. The occurrence of chlorite and
8.1 This test method covers the determination of the
chlorate is associated with the use of chlorine dioxide, as well
oxyhalides—chlorite, bromate, and chlorate, and bromide, in
as hypochlorite solutions used for drinking water disinfection.
raw water, finished drinking water and bottled (non-
The occurrence of bromate is associated with the use of ozone
carbonated) water by chemically suppressed ion chromatogra-
for disinfection, wherein naturally occurring bromide is oxi-
phy. The ranges tested using this test method for each analyte
dizedtobromate.Bromideisanaturallyoccurringprecursorto
were as follows:
the formation of bromate.
Chlorite 5 to 500 µg/L
Bromate 1 to 25 µg/L
5. Reagents and Materials
Bromide 5 to 250 µg/L
Chlorate 5 to 500 µg/L
5.1 Purity of Reagents—Reagent grade chemicals shall be
8.1.1 The upper limits may be extended by appropriate
used in all tests. Unless otherwise indicated, it is intended that
sample dilution or by the use of a smaller injection volume.
all reagents shall conform to the specifications of the Commit-
Otherionsofinterest,suchasfluoride,chloride,nitrite,nitrate,
tee onAnalytical Reagents of theAmerican Chemical Society,
phosphate, and sulfate may also be determined using this test
where such specifications are available. Other grades may be
method. However, analysis of these ions is not the object of
used, provided it is first ascertained that the reagent is of
this test method.
8.2 Itistheuser’sresponsibilitytoensurethevalidityofthis
4 test method for waters of untested matrices.
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
listed by the American Chemical Society, see Analar Standards for Laboratory
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, Hautman, D. P., and Bolyard, M., Journal of Chromatography, Vol 602, 1992,
MD. p. 65.
D6581 − 18
8.3 ThistestmethodistechnicallyequivalentwithPartBof 11. Apparatus
U.S. EPA Method 300.1, titled “The Determination of Inor-
11.1 Ion Chromatography Apparatus—Analytical system
ganic Anions in Drinking Water by Ion Chromatography.”
complete with all required accessories, including eluent pump,
injector, syringes, columns, suppressor, conductivity detector,
9. Summary of Test Method A
data system, and compressed gasses.
9.1 Oxyhalides (chlorite, bromate, and chlorate) and bro-
11.1.1 Eluent Pump—Capable of delivering 0.10 to 5.0
mide in raw water, finished drinking water and bottled water
mL/min of eluent at a pressure of up to 27 600 kPa (4000 psi).
are determined by ion chromatography. A sample (200 µL) is
11.1.2 Injection Valve—Alowdead-volumeswitchingvalve
injected into an ion chromatograph and the pumped eluent
that will allow the loading of a sample into a sample loop and
(sodium carbonate) sweeps the sample through the analytical
subsequent injection of the loop contents into the eluent
column set. Here, anions are separated from the sample matrix
stream. A loop size of up to 50 µL may be used without
according to their retention characteristics, relative to the
compromising the resolution of early eluting peaks, such as
anions in the eluent.
chlorite and bromate.
9.1.1 The separated anions in the eluent stream then pass
11.1.3 Guard Column—Anion exchange column typically
through a suppressor device, where all cations are exchanged
packed with the same material used in the analytical column.
for hydronium ions. This converts the eluent to carbonic acid,
The purpose of this column is to protect the analytical column
thus reducing the background conductivity. This process also
from particulate matter and irreversibly retained material.
converts the sample anions to their acid form, thus enhancing
11.1.4 Analytical Column—Anion exchange column ca-
their conductivity. The eluent stream then passes through a
pableofseparatingtheionsofinterestfromeachother,aswell
conductivity cell, where they are detected.Achromatographic
as from other ions which commonly occur in the sample
integrator or appropriate computer-based data system is typi-
matrix. The separation shall be at least as good as that shown
cally used for data presentation.
in Fig. 1. Conditions of the eluent may vary by column
9.2 The anions are identified based on their retention times manufacturer.
compared to known standards. Quantification is accomplished
NOTE 1—TheAnalytical Column Set (see 3.2.2) should be able to give
by measuring anion peak areas and comparing them to the
baseline resolution of all anions, even for a 50-µLinjection containing up
areas generated from known standards.
to 200 mg/L, each, of common anions, such as chloride, bicarbonate, and
sulfate.
10. Interferences
11.1.5 Suppressor Device—Asuppressor device based upon
cationexchangeprinciples.Inthistestmethod,simultaneously
10.1 Positive errors can be caused by progressive oxidation
ofresidualhypochloriteorhypobromite,orboth,inthesample regenerating suppressor device with sequential carbonate re-
moverwasused.Anequivalentsuppressordevicemaybeused
to the corresponding chlorate and bromate. Furthermore, chlo-
ritecanalsobeoxidizedtochlorate,causingnegativeerrorsfor providedthatcomparablemethoddetectionlimitsareachieved
and that adequate baseline stability is attained.
chloriteandpositiveerrorsforchlorate.Theseinterferencesare
eliminated by the sample preservation steps outlined in 15.3. 11.1.6 ConductivityDetector—Alow-volume,flowthrough,
Chloride present at >200 mg/L can interfere with bromate temperature stabilized conductivity cell equipped with a meter
determination. capable of reading from 0 to 15 000 µS/cm on a linear scale.
FIG. 1 Chromatogram of a Standard Containing Low µg/L Oxyhalides, and Bromide, in the Presence of Common Inorganic Anions
(See Table 1 for Analysis Conditions)
D6581 − 18
FIG. 2 Overlay Chromatogram Spiked and Unspiked of Low µg/L Oxyhalides, and Bromide, in Houston, TX, Drinking Water
(See Table 1 for Analysis Conditions)
11.1.7 Data System—A chromatographic integrator or
Conditions
Condition A with and without CRD-300
computer-based data system capable of graphically presenting
Column: IonPac AS23 (4 × 250 mm)
thedetectoroutputsignalversustime,aswellaspresentingthe
IonPac AG23 (4 × 50 mm)
integrated peak areas. Eluent: 4.5 mM K CO / 0.8 mM KHCO
2 3 3
Flow rate: 1.0 mL/min
Suppressor: Suppressed conductivity ASRS-300, 4-mm external water
12. Example of Chromatogram—IC Conditions—1
mode,
12.1 See Fig. 1, Fig. 2, and Table 1. CRD-300 4 mm, vacuum mode
Background: <1.5 µS
Noise: ~0.3 nS
13. Example of Chromatogram—IC Conditions—2
13.2 See Fig. 3, Fig. 4, Table 2, Table 3, and Table 4.
13.1 Acarbonateremovaldeviceisdevelopedtoremovethe
majorityofthecarbonatefromtheeluentandallowhydroxide-
14. Preparation of Apparatus
like performance with improved detection sensitivity. This
device, the CRD-300, was used with the IonPac AS23 to
14.1 Set up the ion chromatograph according to the manu-
determine bromate in a bottled mineral water samples. This
facturer’s instructions. If anAnion Self Regenerating Suppres-
data shows the improved detection sensitivity when using the
sor is used, operate the device at 100 mAin the external water
CRD-300 compared to chromatography without the CRD-300.
mode. The conductivity detector cell should be thermally
Scientists responsible for water analysis can chose the column
stabilized at 35°C.
and eluent chemistry that best meets their needs to reliably
14.2 The recommended operating conditions for the ion
determine bromate at concentrations below the common 10-
chromatograph are summarized in Table 1.
µg/L regulatory limit.
14.3 The detector ranges are variable. Normal operating
A
ranges for quantifying the low level of oxyhalides encountered
TABLE 1 Instrumentation and Operating Conditions for the
Determination of Oxyhalides and Bromide and by Ion
in treated drinking water are in the 0.2 to 2-µS/cm full scale
Chromatography, as shown in Figs. 1 and 2
range. Choose a range consistent with the concentration range
A
Ion Chromatograph: Metrohm 850 Professional IC (or equivalent)
intheexpectedsamplesandwiththeoperatingrequirementsof
A
Guard Column: Metrosep ASUPP4/5 (or equivalent)
A
the chromatographic system used.
Analytical Column: Metrosep ASUPP7 (or equivalent)
Eluent: 3.5-mM sodium carbonate
14.4 Equilibrate the chromatographic system by pumping
Flow-Rate: 0.7 mL/min
Injection Volume: 50 µL
the analysis eluent (see 15.2) through the system until a stable
A
Suppressor: Metrohm MSM-II (Tri-Chamber Micro-packed)
B
baseline is obtained (approximately 20 minutes). Typical
or equivalent coupled sequentially with MCS
Detector: Conductivity Detector stabilized at 40°C
baseline characteristics necessary to obtain the method detec-
A
A trademark of Metrohm AG, Switzerland. tion limits required for this analysis are: (1) a background
B
MCS is a carbonate suppressor device that permits use of carbonate/
conductance of 20 to 25 µS/cm and (2) a peak-to-peak (noise)
bicarbonate buffer based eluent in ion chromatography to achieve greater sensi-
variation of no greater than 5 nS/cm per minute of monitored
tivity and better detection limits.
baseline response.
D6581 − 18
NOTE 1—Peaks:
1. Fluoride (0.1 mg/L) 5. Nitrite (0.3 mg/L) 8. Nitrate (0.5 mg/L)
2. Chlorite (0.3 mg/L) 6. Chlorate (0.5 mg/L) 9. Phosphate (0.8 mg/L)
3. Bromate (0.6 mg/L) 7. Bromide (0.5 mg/L) 10. Sulfate (0.6 mg/L)
4. Chloride (0.2 mg/L)
FIG. 3 Chromatography of a Mixed Anion Standard with a CRD-300 (A) and without a CRD-300 (B)
15. Reagents and Materials 15.4 SPE Sample Pretreatment Cartridges—Chloride pres-
ent at >200 mg/L and carbonate present at >300 mg/L can
15.1 Eluent, Concentrate (90.0-mM Sodium Carbonate)
+ +
interfere with bromate determination. H form and Ag form
—Dissolve 9.540 g of sodium carbonate in 1000 mL of water.
cation exchange SPE cartridges can be used to minimize the
15.2 Eluent, Analysis (9.0-mM Sodium Carbonate)—Dilute
carbonate and chloride interferences, respectively, if required.
100.0 mL of Eluent Concentrate (see 15.1) to 1.000 L with
OnGuard-H and OnGuard-Ag cartridges have been shown to
water.
be suitable for this application. The use of these pretreatment
15.2.1 The Eluent Analysis Solution (9.0-mM sodium sar-
cartridges will effect recoveries for bromide, requiring that it
bonate)mustbepurgedfor10minuteswithheliumpriortouse
be analyzed in a separate run.
to remove dissolved gasses in order to ensure optimal system
performance. 15.5 Suppressor Regenerant Solution—If a suppressor re-
quiring chemical regeneration is used, the regenerant solution
15.3 Ethylenediamine (EDA) Preservation Solution (50.0
is prepared by cautiously adding 3.00 mL of concentrated
g/L)—Dilute 11.2 mL of ethylenediamine (99%) to 200 mL
with reagent water. Prepare this solution fresh monthly. Add
1.00 mL of this solution per 1.000 L of blank, standard or
sample to produce a final EDA concentration of 50 mg/L. Joyce, R. J., and Dhillon, H. J., Chromatography, Vol 671, 1994, p. 165.
D6581 − 18
NOTE 1—Peaks:
1. Fluoride (0.5 mg/L) 4. Chloride (50 mg/L) 6. Unknown
2. Chlorite (10, 20, and 40 µg/L) 5. Chlorate (10, 20, and 40 µg/L) 7. Sulfate (100 mg/L)
3. Bromate (10, 20, and 40 µg/L)
FIG. 4 Overlay of Chromatograms of Three Concentration Levels of Chlorite, Bromate, and Chlorate in a Mixed Anion Standard with a
CRD-300 (A) and without a CRD-300 (B)
–
TABLE 2 Calibration Report for Chlorite, Bromate, and Chlorate
15.6.1 Bromate (BrO ) Solution, Stock (1.00 mL= 1.00 mg
–
with a CRD-300 (A) and without a CRD-300 (B)
BrO )—Dissolve 1.180 g of sodium bromate (NaBrO)in
3 3
R-Square (%)
water and dilute to 1.000 L.
Peak Name Points
A
–
15.6.2 Bromide (Br ) Solution, Stock (1.00 mL = 1.00 mg
Chlorite 3 99.9961 99.9748
–
Bromate 3 100.0000 99.9986 Br )—Dissolve 1.288 g of sodium bromide (NaBr) in water
Chlorate 3 99.9995 99.9637
and dilute to 1.000 L.
–
15.6.3 Chlorate (ClO ) Solution, Stock (1.00 mL= 1.00 mg
–
C1O )—Dissolve 1.275 g of sodium chlorate (NaClO)in
3 3
sulfuricacid(sp.gr.1.84)to4.000Lofwater.IfanAnionSelf
water and dilute to 1.000 L.
Regenerating Suppressor is used, it should be operated in the –
15.6.4 Chlorite (ClO ) Solution, Stock (1.00 mL = 1.00 mg
–
external water mode.
ClO )—Dissolve 1.680 g of sodium chlorite (NaClO)in
2 2
15.6 Standard Solutions, Stock (1.00 mL = 1.00 mg)— water and dilute to 1.000 L. Note that as sodium chlorite is
Purchasecertifiedsolutionsorpreparestockstandardsolutions usuallyavailableonlyasan80%technicalgradesalt,the80%
from the following salts, as described below: purity is accounted for in the 1.680-g weight cited above. If an
D6581 − 18
TABLE 3 Determination of Bromate and Chlorate in a Bottled Mineral Water Sample
A B
Injection No. (µg/L) (µg/L)
Bromate Chlorate Bromate Chlorate
1 11.0 1.52 5.33 ND
2 10.9 1.55 6.23 ND
3 10.9 1.35 5.02 ND
4 10.1 1.91 6.25 ND
5 11.3 1.48 5.89 ND
Average 10.8 1.56 5.74 —
RSD 4.34 13.42 9.61 —
TABLE 4 Spike Recovery of Bromate, Chlorate, and Chlorite in Mineral Water Using a System with a CRD-300 (A) and without a CRD-
300 (B)
A B
Injection No. (µg/L) (µg/L)
Chlorite Bromate Chlorate Chlorite Bromate Chlorate
A
Sample ND 10.83 1.56 ND 5.74 ND
Spike 10 10 10 10 10 10
B
Measured Amount 9.88 20.51 12.02 8.58 15.30 8.50
RSD 2.39 1.60 2.45 2.39 1.60 2.45
Recovery (%) 98.8 98.5 104 85.8 97.2 85.0
A
ND = Not Detected
B
The average of five injections.
alternate purity is used, make an appropriate adjustment in the ≥0.995 for accurate results. Once the calibration curves have
weight of salt used after determining the exact percentage of been established, verification must be performed on each
NaClO , which can be done using an iodometric titration analysisday,wheneverfresheluentisprepared,andtwiceeach
2,7
procedure. batch of samples, as outlined in 20.4 and 20.5.
15.7 Reagent Blank—Add 1.00 mL of EDA Preservation
17. Procedure
Solution (see 15.3) to 1.000 L of reagent water.
17.1 Injectthereagentblank,calibrationstandardorsample
intotheeluentstreamandrecordthechromatogram.Inthecase
16. Calibration and Standardization
of a manual injector, flush an excess of the sample (minimum
16.1 Typical Range of Applicability—This test method is
of 5× loop volume) through the sample injection port using a
applicable to the determination of bromate, bromide, chlorate,
syringe prior to injection.A200-µLinjection is required when
and chlorite in raw water, finished drinking water and bottled
using a 4-mm ID column, a 50-µL injection is required when
(non-carbonated) water. The application ranges tested for each
using a 2-mm ID column, in order to achieve the required
analyte are as follows: bromate; 5–30 µg/L, bromide; 20–200
detection limits for this analysis. An example of a chromato-
µg/L, chlorite; 20–500 µg/L, and chlorate; 20–500 µg/L.
gram of low level oxyhalides and bromide is shown in Fig. 1.
16.2 Calibration Standards—For each individual calibra- An example chromatogram of low level oxyhalides and bro-
tioncurve,preparecalibrationstandards,ataminimumofthree
mide in a modest ionic strength, simulated drinking water is
concentration levels, by accurately adding measured volumes shown in Fig. 2.
of the stock standards (see 15.6) to a volumetric flask(s).Add
18. Calculation
50 mg/L of EDA (the equivalent of 1.00 mL of EDA Preser-
18.1 Comparethepeakareasfortheanionsinthesampleto
vation Solution (see 15.3) per 1.000 L of solution) to the
the calibration curves prepared in 16.3 to calculate and report
volumetric flask(s) and dilute to volume with reagent water.A
the anion concentration in µg/L:
minimum of five concentration levels is recommended if the
curve covers two orders of magnitude.
Anionconcentration, µg/L 5 A 3F (1)
16.3 Calibration Curve—To establish the calibration curve,
where:
analyze a reagent blank and the calibration standards in
A = reading from the appropriate calibration plot, in µg/L,
accordance to the procedure in Section 17, using a 200-µL
and
injection (with a 4-mm ID column) or a 50-µL injection (with
F = dilution factor if the sample was diluted prior to
a 2-mm ID column). Tabulate peak area responses against
analysis.
concentration. These results are used to prepare a calibration
18.1.1 Computing integrators and computer based chro-
curve using a linear least squares fit for each analyte. The
matographicdatasystemscanbeprogrammedtoperformthese
squared correlation coefficient of the regression (r ) should be
calculations automatically.
18.2 Report only those values that fall between the lowest
Method 4500–ClO .C in A. E. Greenberg, L. S. Clesceri, A. D. Eaton (Eds.),
and highest calibration standards. Samples exceeding the
Standard Methods for the Examination of Water and Wastewater, 18th Ed.,APHA,
Washington, DC, 1992. highest standard should be diluted and reanalyzed.
D6581 − 18
19. Precision and Bias with reagent water. The MDL sample was prepared by pi-
petting a 1.0-mL aliquot of the MDL concentrate into a clean
19.1 The precision and bias data presented in this test
volumetric flask; adding 50-mg/LEDA, and diluting to a total
method meets the requirements of Practice D2777 – 98, which
of 100 mL with reagent water.
was in place at the time of collaborative testing. Under the
allowancesmadein1.4ofPracticeD2777–13,theseprecision
19.3 All the precision and bias data presented in this test
andbiasdatadomeetexistingrequirementsforinterlaboratory
method was obtained using the IonPacAS9-HC column listed
studies of Committee D19 test methods. The full research
in Table 1.
report can be obtained from ASTM Headquarters.
19.4 The precision and bias of this test method for each
19.2 The interlaboratory study that generated the precision
analyte for reagent, drinking, and bottled water are shown in
and bias data in this test method was performed in reagent
Tables 5-8.
water, municipal drinking water, and bottled (non-carbonated)
19.5 The results of the interlaboratory study can also be
waterbytenlaboratoriesusingoneoperatoreach.Sixlevelsof
summarized as regression equations, as shown in Table 9 for
concentration were used for four analytes, producing three
reagent water and in Table 10 for a typical sample matrix of
Youden pairs. The Youden pair data was used to calculate the
drinking water.
single operator precision (S ). The analytes were supplied
o
separately as six (mixed) concentrates. The reagent water,
19.6 In addition to performing the analyses required to
municipal drinking water and bottled water were supplied by
generate the precision and bias data shown in Tables 5-8, the
the participating laboratories. Six reagent water samples, six
participating laboratories each analyzed seven replicates of an
bottled water samples, and six municipal drinking water
MDL sample. The MDLs were derived for each laboratory
samples (for a total of 18 samples) were prepared by pipetting
using the students t-test at six degrees of freedom, as follows:
1.0 mL aliquots of the concentrates labeled A1-2 (×3), B1-2
MDL 5 ~t! 3 ~S! (2)
(×3), C1-2 (×3) into volumetric flasks (18 total); adding 50
where:
mg/L EDA preservation solution, as detailed in 7.2, and
diluting to a total of 100 mL with reagent water (×6), bottled
t = students t value for a 99% confidence level and a
water (×6), and drinking water (×6), as appropriate.
standarddeviationestimatewithn-1degreesoffreedom
19.2.1 A quality control (QC) sample was supplied (as a
[t = 3.14 for seven replicates], and
concentrate) to serve as initial, and on-going, calibration S = standard deviation of the replicate analysis.
verification. A separate method detection limit (MDL) sample
19.6.1 True amounts injected, mean value determined, and
was supplied (as a concentrate) for the determination of the
pooled MDLvalues (10 laboratories × 7 replicates) are shown
pooledMDLvalues.TheQCsamplewaspreparedbypipetting
in Table 11.
a1.0-mLaliquotoftheQCconcentrateintoacleanvolumetric
flask; adding 50-mg/LEDA, and diluting to a total of 100 mL
20. Quality Control
20.1 Before this test method is applied to analyzing un-
Supporting data have been filed atASTM International Headquarters and may
known samples, the analyst should establish quality control
beobtainedbyrequestingResearchReportRR:D19-1164.ContactASTMCustomer
procedures as recommended in Guide D3856.
Service at service@astm.org.
TABLE 5 Determination of Precision and Bias for Chlorite
Amount Added Amount Found Number Retained S S Bias
o t
Water
(µg/L) (µg/L) Parts (µg/L) (µg/L) (%)
Reagent 20 19.94 8 1.40 1.25 –0.3
25 25.06 1.05 0.2
180 178.29 8 4.37 5.64 –1.0
220 214.73 6.18 –2.4
400 394.36 8 19.39 21.81 –1.4
450 440.53 8.29 –2.1
Drinking 20 19.19 8 1.52 6.58 –4.1
25 23.77 6.17 –4.9
180 174.30 8 5.06 9.29 –3.2
220 216.89 14.76 –1.4
400 398.30 8 4.42 15.56 –0.4
450 439.85 19.59 –2.3
Bottled 20 20.94 8 4.41 3.65 4.7
25 22.74 4.64 –9.0
180 177.71 8 2.95 8.76 –1.3
220 216.16 8.74 –1.7
400 390.14 8 9.53 13.65 –2.5
450 433.72 15.30 –3.6
D6581 − 18
TABLE 6 Determination of Precision and Bias for Bromate
Amount Added Amount Found Number Retained S S Bias
o t
Water
(µg/L) (µg/L) Parts (µg/L) (µg/L) (%)
Reagent 5 4.95 9 0.99 1.19 –0.9
7 7.84 1.29 12.0
10 9.98 9 0.66 0.99 –0.2
12 11.76 0.55 –2.0
20 19.56 9 2.17 2.37 –2.2
25 24.18 1.53 –3.3
Drinking 5 4.41 6 0.62 1.20 –11.8
7 6.44 0.78 –8.0
10 8.54 8 0.72 2.88 –14.6
12 10.20 2.88 –15.0
20 17.31 8 2.80 2.85 –13.4
25 20.51 4.77 –17.9
Bottled 5 4.95 6 1.09 1.51 –1.1
7 7.21 1.80 3.0
10 9.07 6 1.67 4.83 –9.3
12 10.35 3.41 –13.7
20 20.16 6 4.34 3.91 0.8
25 20.99 7.75 –16.0
TABLE 7 Determination of Precision and Bias for Bromide
Amount Added Amount Found Number Retained S S Bias
o t
Water
(µg/L) (µg/L) Parts (µg/L) (µg/L) (%)
Reagent 20 20.75 9 1.94 1.91 3.8
25 25.51 2.32 2.1
75 74.52 9 3.80 2.94 –0.6
100 99.42 4.84 –0.6
150 143.50 9 5.79 5.82 –4.3
180 176.38 5.24 –2.0
Drinking 20 20.68 8 1.30 4.39 3.4
25 25.49 3.31 2.0
75 71.89 8 4.67 5.67 –4.2
100 97.05 6.90 –3.0
150 145.81 8 1.23 8.39 –2.8
180 173.40 9.12 –3.7
Bottled 20 20.25 7 2.21 1.79 1.3
25 26.23 1.11 4.9
75 74.35 8 6.45 4.69 –0.9
100 98.49 5.00 –1.5
150 142.67 8 6.57 6.08 –4.9
180 172.71 9.24 –4.1
TABLE 8 Determination of Precision and Bias for Chlorate
Amount Added Amount Found Number Retained S S Bias
o t
Water
(µg/L) (µg/L) Parts (µg/L) (µg/L) (%)
Reagent 20 20.69 7 2.73 2.43 3.5
25 26.64 3.79 6.6
180 176.05 8 11.96 3.70 –2.2
220 215.39 7.47 –2.1
400 393.00 7 18.27 5.85 –1.7
450 443.47 16.50 –1.5
Drinking 20 19.94 9 1.81 3.95 –0.3
25 23.93 5.13 –4.3
180 175.10 9 6.92 9.05 –2.7
220 216.14 7.02 –1.8
400 396.74 9 4.74 16.55 –0.8
450 441.69 16.55 –1.8
Bottled 20 21.72 8 2.86 3.88 8.6
25 25.75 3.21 3.0
180 179.82 8 3.59 5.37 –0.1
220 217.58 9.26 –1.1
400 389.51 7 6.72 15.83 –2.6
450 443.70 10.00 –1.4
20.2 The laboratory using this test should perform an initial tion. The IDP solution contains method analytes of known
demonstration of laboratory capability. Analyze seven repli- concentration, prepared from a different source to the calibra-
cates of an initial demonstration of performance (IDP) solu- tion standards, used to fortify reagent water, which also
D6581 − 18
TABLE 9 Summary of Precision and Bias Results for Reagent Water
Chlorite Bromate Bromide Chlorate
Precision and Bias Summary Precision and Bias Summary Precision and Bias Summary Precision and Bias Summary
Number of Laboratories 10 10 10 10
Range Tested 20–450 µg/L 5–25 µg/L 20–180 µg/L 20–450 µg/L
Mean Recovery y = 0.9805x + 0.5261 y = 0.9432x + 0.6272 y = 0.9629x + 1.7475 y = 0.9809x + 0.8245
S y = 0.0465x – 1.4801 y = 0.0878x + 0.1281 y = 0.0282x + 1.3087 y = 0.0389x + 2.7278
o
S y = 0.0332x + 0.3294 y = 0.046x + 0.721 y = 0.0246x + 1.6352 y = 0.0226x + 1.8244
t
TABLE 10 Summary of Precision and Bias Results for Drinking Water
Chlorite Bromate Bromide Chlorate
Precision and Bias Summary Precision and Bias Summary Precision and Bias Summary Precision and Bias Summary
Number of Laboratories 10 10 10 10
Range Tested 20–450 µg/L 5–25 µg/L 20–180 µg/L 20–450 µg/L
Mean Recovery y = 0.9872x – 1.0243 y = 0.9432x + 0.6272 y = 0.9583x + 1.2113 y = 0.9868x – 0.7347
S y = 0.0068x + 2.2164 y = 0.1721x – 0.5532 y = –0.0022x + 2.6 y = 0.0066x + 3.0956
o
S y = 0.0289x + 5.8552 y = 0.1934x + 0.3866 y = 0.0357x + 3.1189 y = 0.03x + 3.3368
t
TABLE 11 Pooled MDL Values Obtained for This Test Method
equivalent to the lowest calibration standard) should initially
Analyte Injected Amount Mean Value Pooled MDL
be analyzed before beginning use of this method. The CVS is
Chlorite 3.0 µg/L 3.32 µg/L 2.39 µg/L a solution of method analytes of known concentration used to
Bromate 4.0 µg/L 3.98 µg/L 2.73 µg/L
fortify reagent water, which also contains a final EDAconcen-
Bromide 4.0 µg/L 3.96 µg/L 2.91 µg/L
tration of 50 mg/L(see 15.3). If the determined low level CVS
Chlorate 4.0 µg/L 3.74 µg/L 3.49 µg/L
values are not within 625% of the known amounts, the low
level CVS should be reanalyzed. If the values still fall outside
acceptable limits, a new calibration curve is required which
contains a final EDA concentration of 50 mg/L (see 15.3).
must be confirmed by a successful low level CVS before
Ideally,theIPDsolutionshouldbepreparedbyanindependent
continuing with on-going analyses.
source from reference materials. The level 3 standard used for
20.4 AcontinuingCVSshouldbeanalyzedaftereverytenth
themethodprecisionandbiasstudyisrecommendedasanIDP
field sample and an end CVS should analyzed at the end of the
solution.
sample batch (maximum of 20 samples) to verify the previ-
20.2.1 The mean and standard deviation of the seven values
should then be calculated and compared, according to Practice ously established calibration curves.After initially meeting the
D5847, to the single operator precision and recovery estab- requirementsof20.3,thelevelsselectedforthecontinuingand
lished for this test method. The upper limit for acceptable end CVS should be varied between a middle calibration level
precision and the range of acceptable recoveries are detailed andthehighestcalibrationlevelstandard.Ifthecontinuingand
below:
end CVS values are not within 615% of the known amounts,
the analyst should reanalyze the CVS. If the analyte concen-
IDP Solution Acceptable IDP
Analyte Method S
Amount Precision, n = 7
trationsstillfalloutsideacceptablelimits(615%)thatanalyte
Chlorite 180 µg/L 4.4 µg/L # 11.8 µg/L
is judged out of control, and the source of the problem should
Bromate 10 µg/L 0.66 µg/L # 1.67 µg/L
Bromide 75 µg/L 3.8 µg/L # 9.6 µg/L be identified before continuing with on-going analyses. All
Chlorate 180 µg/L 12.0 µg/L # 32.1 µg/L
samples following the last acceptable CVS should be reana-
lyzed.
Method Mean Lower Acceptable Upper Acceptable
Analyte
Recovery IDP Recovery IDP Recovery
20.5 Areagent blank (see 15.7) should be run when gener-
Chlorite 178.3 µg/L 164.6 µg/L 191.7 µg/L
Bromate 9.98 µg/L 7.37 µg/L 12.59 µg/L
ating the initial calibration curves.Ablank should also be run
Bromide 74.5 µg/L 70.8 µg/L 78.2 µg/L
witheachsamplebatch(maximumof20samples)tocheckfor
Chlorate 176.1 µg/L 171.2 µg/L 181.0 µg/L
sample or system contamination.
20.2.2 The S and mean recovery values can be calculated
o
for different IDP solution concentrations using the regression
20.6 One laboratory control sample (LCS) should be used
equations for each analyte shown in Table 9. If the values
witheachsamplebatch(maximumof20samples).TheLCSis
obtained for the IDP precision and recovery do not meet the
asolutionofmethodanalytesofknownconcentrationaddedto
criteria described above, initial demonstration of performance
a matrix which sufficiently challenges the Test Method. A
must be repeated until the results fall within these criteria.
synthetic drinking water matrix, containing fluoride at 1.0
mg/L, chloride at 50 mg/L, nitrite at 0.1 mg/L, nitrate at 10
20.3 When beginning use of this method, an initial calibra-
mg/L, phosphate at 0.1 mg/L and sulfate at 50 mg/L, spiked
tion verification standard (CVS) should be used to verify the
with the four method analytes at the level of the IDP solution
calibration standards and acceptable instrument performance.
This verification should be performed on each analysis day or would be an example of an appropriate LCS. The LCS shall
also contain 50 mg/L of EDA (the equivalent of 1.00 mL of
whenever fresh eluent has been prepared. As this method is
intended for use at trace levels, a low level CVS (that is, EDAPreservation Solution (see 15.3) per 1.000 Lof solution).
D6581 − 18
20.6.1 TheanalyterecoveriesfortheLCSshouldfallwithin
Chlorite 20 to 1000 µg/L
Bromate 1 to 30 µg/L
the control limits of x 6 3S, where x is the mean recovery and
Chlorate 20 to 1000 µg/L
(S) is the standard deviation of the mean recovery established
Bromide 20 to 200 µg/L
from the interlaboratory precision and bias study data at the
21.1.1 It should be noted that the U.S. EPA maintains that
IDP levels, as shown below:
the use of hydroxide-based separation is consistent with
Lower Recovery Upper Recovery
Analyte LCS Amount Method 300.1 (Parts A and B) for compliance monitoring.
Limit Limit
Chlorite 180 µg/L 165 µg/L 191 µg/L
22. Summary of Test Method B
Bromate 10 µg/L 8.0 µg/L 12.0 µg/L
Bromide 75 µg/L 63 µg/L 86 µg/L
22.1 Oxyhalides (chlorite, bromate, and chlorate) and bro-
Chlorate 180 µg/L 140 µg/L 219 µg/L
mide in raw water, finished drinking water and bottled water
20.7 Onematrixspike(MS)shouldberunwitheachsample
are determined by ion chromatography. A sample (200 µL) is
batch (maximum of 20 samples) to test method recovery. The
injected into an ion chromatograph and the pumped hydroxide
MS should be prepared in accordance with Guide D5810.
eluent sweeps the sample through the analytical column set.
Spikeaportionofadrinkingwater(orother)samplefromeach
Here,anionsareseparatedfromthesamplematrixaccordingto
batch with the four method analytes at the level of the IDP
their retention characteristics, relative to the anions in the
solution. The %recovery of the spike should fall within limits
eluent.
established from the interlaboratory precision and bias study
22.1.1 The separated anions in the eluent stream then pass
data (assuming a background level of zero), according to
through a suppressor device, where all cations are exchanged
Practice D5847, as shown below:
for hydronium ions. This converts the eluent to water, thus
Lower Recovery Upper Recovery
reducing the background conductivity. This process also con-
Analyte MS Amount
Limit (%) Limit (%)
vertsthesampleanionstotheiracidform,thusenhancingtheir
Chlorite 180 µg/L 90.9 % 109.1 %
Bromate 10 µg/L 73.1 % 126.9 % conductivity. The eluent stream then passes through a conduc-
Bromide 75 µg/L 80.8 % 119.2 %
tivity cell, where they are detected. An appropriate computer-
Chlorate 180 µg/L 88.3 % 111.7 %
based data system is typically used for data presentation.
20.8 One matrix duplicate (MD) should be run with each
22.2 Theanionsareidentifiedbasedontheirretentiontimes
sample batch (maximum of 20 samples) to test method
compared to known standards. Quantification is accomplished
precision. If non-detects are expected in all the samples to be
by measuring anion peak areas and comparing them to the
analyzed, a matrix spike duplicate should be run instead. The
areas generated from known standards.
precisionoftheduplicateanalysisshouldbecompared,accord-
ing to Practice D5847, to the nearest tabulated S value
o
23. Interferences
established from the interlaboratory precision and bias study
23.1 Positive errors can be caused by progressive oxidation
data for each analyte.
ofresidualhypochloriteorhypobromite,orboth,inthesample
20.9 In order to verify the quantitative values produced by
to the corresponding chlorate and bromate. Furthermore, chlo-
the test method, an Independent Reference Material (IRM),
ritecanalsobeoxidizedtochlorate,causingnegativeerrorsfor
submitted to the laboratory as a regular sample (if practical),
chloriteandpositiveerrorsforchlorate.Theseinterferencesare
should be analyzed once per quarter. The concentration of the
eliminated by the sample preservation steps outlined in 26.3.
IRM should be within the scope of the method, as defined in
Chloride present at >200 mg/L and carbonate present at >300
1.1.Thevaluesobtainedmustfallwithinthelimitsspecifiedby
mg/Lcaninterferewithbromatedetermination.Theseinterfer-
the outside source.
ences can be minimized, or eliminated, by the sample pretreat-
20.10 The laboratory may perform additional quality con- mentstepsoutlinedin26.4.Fluorideandlowmolecularweight
trolasdesiredorappropriate,forinstancetheuseofasurrogate monocarboxylic acids, present at mg/L concentrations, may
as outlined in Section 9.4.2 of U.S. EPA Method 300.1. In interfere with the quantitation of chlorite and bromate.
addition, it is recommended that a laboratory determine the
24. Apparatus
method detection limits, as discussed in 19.6, before using this
test method. 24.1 Ion Chromatography Apparatus—Analytical system
complete with all required accessories, including eluent pump,
TEST METHOD B
injector, syringes, columns, suppressor, conductivity detector,
ELECTROLYTICALLY SUPPRESSED ION
data system and compressed gasses.
CHROMATOGRAPHY
24.1.1 Eluent Pump—Capable of delivering 0.25 to 5 mL/
min of eluent at a pressure of up to 27 600 kPa (4000 psi).
21. Scope
24.1.2 Injection Valve—Alowdead-volumeswitchingvalve
21.1 This test method is technically consistent with U.S. that will allow the loading of a sample into a sample loop and
EPAMethod 300.1 (Part B), as cited above, except that it uses subsequent injection of the loop contents into the eluent
analyte separation on a hydroxide-selective anion exchange stream. A loop size of up to 250 µL may be used without
column, using a hydroxide eluent followed by electrolytically compromising the resolution of early eluting peaks, such as
suppressed conductivity detection of the analytes in a deion- chlorite and bromate.
izedwatermatrix,thusimprovingmethoddetectionlimits.The 24.1.3 Guard Column—Anion exchange column typically
range tested for each analyte were as follows: packed with the same material used in the analytical column,
D6581 − 18
to 200 mg/L, each, of common anions, such as chloride, bicarbonate, and
for example,AG19, or equivalent. The purpose of this column
sulfate.
is to protect the analytical column from particulate matter and
irreversibly retained material.
24.1.5 Suppressor Device—Asuppressordevicebasedupon
24.1.4 Analytical Column—Anion exchange column ca-
cation exchange principles. An ASRA ULTRA II (4 mm)
pableofseparatingtheionsofinterestfromeachother,aswell
electrolytic suppressor device was used that does not require
as from other ions which commonly occur in the sample
the addition of an acid but is a plug in electrolytic device. The
matrix, for example, IonPac AS19 (4-mm ID), or equivalent.
suppressed eluent (water) is simply recirculated from the
TheseparationshallbeatleastasgoodasthatshowninFig.5.
conductivity cell back to the electrolytic suppressor to back
The use of 2-mm ID AS19 column, in conjunction with a
flush the suppressor device. Alternative pumps are also typi-
50-µL sample loop, may improve the peak shape for early
cally not required.
eluting anions, such as chlorite and bromate.
NOTE 2—The Analytical Column Set (24.1.4) should be able to give
baselineresolutionofallanions,evenfora200-µLinjectioncontainingup
FIG. 5 Chromatogram of Mineral Water A Spiked with 1 µg/L Each Chlorite and Chlorate and 0.5 µg/L Bromate
D6581 − 18
24.1.6 Conductivity Detector—A low-volume, flow hydroxide by Reagent Free Ion Chromatography to generate
through,temperaturestabilizedconductivitycellequippedwith carbonate free hydroxide is also acceptable.
a meter capable of reading from 0 to 1000 µS/cm on a linear
26.2 Test Method B Eluent Conditions—10 mM hydroxide
scale.
from 0 to 10 minutes and 45 mM from 10 to 25 minutes at 1
24.1.7 Data System—A computer-based data system ca-
mL/min, 30°C (see Fig. 5).
pable of graphically presenting the detector output signal
26.3 Ethylenediamine (EDA) Preservation Solution (50.0
versus time, as well as presenting the integrated peak areas.
g/L)—Dilute 11.2 mL of ethylenediamine (99%) to 200 mL
with reagent water. Prepare this solution fresh monthly. Add
25. Preparation of Apparatus
1.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6581 − 12 D6581 − 18
Standard Test Methods for
Bromate, Bromide, Chlorate, and Chlorite in Drinking Water
by Suppressed Ion Chromatography
This standard is issued under the fixed designation D6581; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 These multi-test methods cover the determination of the oxyhalides—chlorite, bromate, and chlorate, and bromide, in raw
water, finished drinking water and bottled (non-carbonated) water by chemically and electrolytically suppressed ion chromatog-
raphy. The ranges tested using these test methods for each analyte were as follows:
Range Sections
Test Method A: 8 to 20
Chemically Suppressed
Ion Chromatography
Chlorite 5 to 500 μg/L
Bromate 1 to 25 μg/L
Bromide 5 to 250 μg/L
Chlorate 5 to 500 μg/L
Test Method B: 21 to 31
Electrolytically
Suppressed Ion
Chromatography
Chlorite 20 to 1000 μg/L
Bromate 1 to 30 μg/L
Bromide 20 to 200 μg/L
Chlorate 20 to 1000 μg/L
1.1.1 The upper limits may be extended by appropriate sample dilution or by the use of a smaller injection volume. Other ions
of interest, such as fluoride, chloride, nitrite, nitrate, phosphate, and sulfate may also be determined using these test methods.
However, analysis of these ions is not the object of these test methods.
1.2 It is the user’s responsibility to ensure the validity of these test methods for waters of untested matrices.
1.3 These test methods are technically equivalent with Part B of U.S. EPA Method 300.1, titled “The Determination of
Inorganic Anions in Drinking Water by Ion Chromatography.”
1.4 The values stated in either SI or inch-pound units are to be regarded as the standard. The values given in parentheses are
for information only.mathematical conversions to inch-pound units that are provided for information only and are not considered
standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
These test methods are under the jurisdiction of ASTM Committee D19 on Water and are the direct responsibility of Subcommittee D19.05 on Inorganic Constituents
in Water.
Current edition approved March 1, 2012May 1, 2018. Published April 2012May 2018. Originally approved in 2000. Last previous edition approved in 20082012 as
D6581 – 08.D6581 – 12. DOI: 10.1520/D6581-12.10.1520/D6581-18.
U.S. EPA 300.1, Cincinnati, OH, 1997.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6581 − 18
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Closed Conduits
D3856 Guide for Management Systems in Laboratories Engaged in Analysis of Water
D5810 Guide for Spiking into Aqueous Samples
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
3. Terminology
3.1 Definitions—Definitions: For definitions of terms used in the test methods, refer to Terminology D1129.
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 analytical column—column, n—the ion exchange column used to separate the ions of interest according to their retention
characteristics prior to detection.
3.2.2 analytical column set—set, n—a combination of one or more guard columns, followed by one or more analytical columns
used to separate the ions of interest. All of the columns in series then contribute to the overall capacity and resolution of the
analytical column set.
3.2.3 eluent—eluent, n—the ionic mobile phase used to transport the sample through the chromatographic system.
3.2.4 guard column—column, n—a column used before the analytical column to protect it from contaminants, such as
particulates or irreversibly retained material.
3.2.5 ion chromatography—chromatography, n—a form of liquid chromatography in which ionic constituents are separated by
ion exchange then detected by an appropriate detection means, typically conductance.
3.2.6 resolution—resolution, n—the ability of an analytical column to separate the method analytes under specific test
conditions.
3.2.7 suppressor device—device, n—an ion exchange based device that is placed between the analytical column set and the
conductivity detector. Its purpose is to minimize detector response to the ionic constituents in the eluent, in order to lower
background conductance; and at the same time enhance the conductivity detector response of the ions of interest.
3.2.7.1 chemical suppression—suppression, n—the use of an acid solution to the suppressor in order to suppress the background
conductivity.
3.2.7.2 electrolytic suppressor device—device, n—electrolytic suppression is an ion exchange device that is placed between the
analytical column and the conductivity detector. Its purpose is similar to a suppressor device, however, it does not require addition
of acid. Instead the electrolytic suppressor generates protons electrolytically and plugs into an electrical power source on typically
located on the chromatography device.
4. Significance and Use
4.1 The oxyhalides chlorite, chlorate, and bromate are inorganic disinfection by-products (DBPs) of considerable health risk
concern worldwide. The occurrence of chlorite and chlorate is associated with the use of chlorine dioxide, as well as hypochlorite
solutions used for drinking water disinfection. The occurrence of bromate is associated with the use of ozone for disinfection,
wherein naturally occurring bromide is oxidized to bromate. Bromide is a naturally occurring precursor to the formation of
bromate.
5. Reagents and Materials
5.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without reducing the accuracy of the determination.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
“ReagentReagent Chemicals, American Chemical Society Specifications,”Specifications, Am.American Chemical Soc.,Society, Washington, DC. For suggestions on the
testing of reagents not listed by the American Chemical Society, see “AnalarAnalar Standards for Laboratory Chemicals,”Chemicals, by BDH Ltd., Poole, Dorset, U.K.,
and the “United States Pharmacopoeia.”United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
D6581 − 18
5.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water conforming to
Specification D1193, Type I. Other reagent water types may be used, provided it is first ascertained that the water is of sufficiently
high purity to permit its use without adversely affecting the bias and precision of the determination.
6. Precautions
6.1 These test methods address the determination of very low concentrations of selected anions. Accordingly, every precaution
should be taken to ensure the cleanliness of sample containers as well as other materials and apparatus that come in contact with
the sample.
7. Sampling and Sample Preservation
7.1 Collect the sample in accordance with PracticePractices D3370, as applicable.
7.2 Immediately upon taking the sample, sparge it with an inert gas (for example, nitrogen, argon, or helium) for 5 minutes to
remove active gases such as chlorine dioxide or ozone. Add 1.00 mL of EDA Preservation Solution (see 15.3) per 1.000 litre of
sample to prevent conversion of residual hypochlorite or hypobromite to chlorate or bromate. This also prevents metal catalyzed
conversion of chlorite to chlorate. The oxyhalides in samples preserved in this manner are stable for at least 14 days when stored
in amber bottles at 4°C.
Test MethodTEST METHOD A
Chemically Suppressed Ion ChromatographyCHEMICALLY SUPPRESSED ICON CHROMATOGRAPHY
8. Scope
8.1 This test method covers the determination of the oxyhalides—chlorite, bromate, and chlorate, and bromide, in raw water,
finished drinking water and bottled (non-carbonated) water by chemically suppressed ion chromatography. The ranges tested using
this test method for each analyte were as follows:
Chlorite 5 to 500 μg/L
Bromate 1 to 25 μg/L
Bromide 5 to 250 μg/L
Chlorate 5 to 500 μg/L
8.1.1 The upper limits may be extended by appropriate sample dilution or by the use of a smaller injection volume. Other ions
of interest, such as fluoride, chloride, nitrite, nitrate, phosphate, and sulfate may also be determined using this test method.
However, analysis of these ions is not the object of this test method.
8.2 It is the user’s responsibility to ensure the validity of this test method for waters of untested matrices.
8.3 This test method is technically equivalent with Part B of U.S. EPA Method 300.1, titled “The Determination of Inorganic
Anions in Drinking Water by Ion Chromatography.”
8.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
9. Summary of Test Method A
9.1 Oxyhalides (chlorite, bromate, and chlorate) and bromide in raw water, finished drinking water and bottled water are
determined by ion chromatography. A sample (200 μL) is injected into an ion chromatograph and the pumped eluent (sodium
carbonate) sweeps the sample through the analytical column set. Here, anions are separated from the sample matrix according to
their retention characteristics, relative to the anions in the eluent.
9.1.1 The separated anions in the eluent stream then pass through a suppressor device, where all cations are exchanged for
hydronium ions. This converts the eluent to carbonic acid, thus reducing the background conductivity. This process also converts
the sample anions to their acid form, thus enhancing their conductivity. The eluent stream then passes through a conductivity cell,
where they are detected. A chromatographic integrator or appropriate computer-based data system is typically used for data
presentation.
9.2 The anions are identified based on their retention times compared to known standards. Quantification is accomplished by
measuring anion peak areas and comparing them to the areas generated from known standards.
10. Interferences
10.1 Positive errors can be caused by progressive oxidation of residual hypochlorite or hypobromite, or both, in the sample to
the corresponding chlorate and bromate. Furthermore, chlorite can also be oxidized to chlorate, causing negative errors for chlorite
Hautman, D. P., and Bolyard, M., Journal of Chromatography, Vol 602, 1992, p. 65.
D6581 − 18
and positive errors for chlorate. These interferences are eliminated by the sample preservation steps outlined in 15.3. Chloride
present at >200 mg/L can interfere with bromate determination.
11. Apparatus
11.1 Ion Chromatography Apparatus—Analytical system complete with all required accessories, including eluent pump,
injector, syringes, columns, suppressor, conductivity detector, data system, and compressed gasses.
11.1.1 Eluent Pump—Capable of delivering 0.10 to 5.0 mL/min of eluent at a pressure of up to 4000 psi (27600 kPa).27 600
kPa (4000 psi).
11.1.2 Injection Valve—A low dead-volume switching valve that will allow the loading of a sample into a sample loop and
subsequent injection of the loop contents into the eluent stream. A loop size of up to 50 μL may be used without compromising
the resolution of early eluting peaks, such as chlorite and bromate.
11.1.3 Guard Column—Anion exchange column typically packed with the same material used in the analytical column. The
purpose of this column is to protect the analytical column from particulate matter and irreversibly retained material.
11.1.4 Analytical Column—Anion exchange column capable of separating the ions of interest from each other, as well as from
other ions which commonly occur in the sample matrix. The separation shall be at least as good as that shown in Fig. 1. Conditions
of the eluent may vary by column manufacturer.
NOTE 1—The Analytical Column Set (see 3.2.2) should be able to give baseline resolution of all anions, even for a 50 μL 50-μL injection containing
up to 200 mg/L, each, of common anions, such as chloride, bicarbonate, and sulfate.
11.1.5 Suppressor Device—A suppressor device based upon cation exchange principles. In this test method, simultaneously
regenerating suppressor device with sequential carbonate remover was used. An equivalent suppressor device may be used
provided that comparable method detection limits are achieved and that adequate baseline stability is attained.
11.1.6 Conductivity Detector—A low-volume, flow through, temperature stabilized conductivity cell equipped with a meter
capable of reading from 0 to 15 000 μS/cm on a linear scale.
11.1.7 Data System—A chromatographic integrator or computer-based data system capable of graphically presenting the
detector output signal versus time, as well as presenting the integrated peak areas.
12. Example of Chromatogram—IC Conditions—1
12.1 See Fig. 1, Fig. 2, and Table 1.
13. Example of Chromatogram—IC Conditions—2
13.1 A carbonate removal device is developed to remove the majority of the carbonate from the eluent and allow hydroxide-like
performance with improved detection sensitivity. This device, the CRD-300, was used with the IonPac AS23 to determine bromate
in a bottled mineral water samples. This data shows the improved detection sensitivity when using the CRD-300 compared to
chromatography without the CRD-300. Scientists responsible for water analysis can chose the column and eluent chemistry that
best meets their needs to reliably determine bromate at concentrations below the common 10 μg/L 10-μg/L regulatory limit.
FIG. 1 Chromatogram of a Standard Containing Low μg/L Oxyhalides, and Bromide, in the Presence of Common Inorganic Anions
(See Table 1 for Analysis Conditions)
D6581 − 18
FIG. 2 Overlay Chromatogram Spiked and Unspiked of Low μg/L Oxyhalides, and Bromide, in Houston, TX, Drinking Water
(See Table 1 for Analysis Conditions)
A
TABLE 1 Instrumentation and Operating Conditions for the
Determination of Oxyhalides and Bromide and by Ion
Chromatography, as shown in Figs. 1 and 2
A
Ion Chromatograph: Metrohm 850 Professional IC (or equivalent)
A
Guard Column: Metrosep ASUPP4/5 (or equivalent)
A
Analytical Column: Metrosep ASUPP7 (or equivalent)
Eluent: 3.5 mM Sodium carbonate
Eluent: 3.5-mM sodium carbonate
Flow-Rate: 0.7 mL/min
Injection Volume: 50 μL
A
Suppressor: Metrohm MSM-II (Tri-Chamber Micro-packed)
B
or equivalent coupled sequentially with MCS
Detector: Conductivity Detector stabilized at 40°C
A
A trademark of Metrohm AG, Switzerland.
B
MCS is a carbonate suppressor device that permits use of carbonate/
bicarbonate buffer based eluent in ion chromatography to achieve greater sensi-
tivity and better detection limits.
Conditions
Condition A with and without CRD-300
Column: IonPac AS23 (4 × 250 mm)
IonPac AG23 (4 × 50 mm)
Eluent: 4.5 mM K CO / 0.8 mM KHCO
2 3 3
Flow rate: 1.0 mL/min
Suppressor: Suppressed conductivity ASRS-300, 4 mm External water mode,
CRD-300 4 mm, Vacuum mode
Suppressor: Suppressed conductivity ASRS-300, 4-mm external water mode,
CRD-300 4 mm, vacuum mode
Background: <1.5 μS
Noise: ~0.3 nS
13.2 See Fig. 3, Fig. 4, Table 2, Table 3, and Table 4.
14. Preparation of Apparatus
14.1 Set up the ion chromatograph according to the manufacturer’s instructions. If an Anion Self Regenerating Suppressor is
used, operate the device at 100 mA in the external water mode. The conductivity detector cell should be thermally stabilized at
35°C.
14.2 The recommended operating conditions for the ion chromatograph are summarized in Table 1.
14.3 The detector ranges are variable. Normal operating ranges for quantifying the low level of oxyhalides encountered in
treated drinking water are in the 0.2 to 2 μS/cm 2-μS/cm full scale range. Choose a range consistent with the concentration range
in the expected samples and with the operating requirements of the chromatographic system used.
14.4 Equilibrate the chromatographic system by pumping the analysis eluent (see 15.2) through the system until a stable
baseline is obtained (approximately 20 minutes). Typical baseline characteristics necessary to obtain the method detection limits
D6581 − 18
NOTE 1—Peaks:
1. Fluoride (0.1 mg/L) 5. Nitrite (0.3 mg/L) 8. Nitrate (0.5 mg/L)
2. Chlorite (0.3 mg/L) 6. Chlorate (0.5 mg/L) 9. Phosphate (0.8 mg/L)
3. Bromate (0.6 mg/L) 7. Bromide (0.5 mg/L) 10. Sulfate (0.6 mg/L)
4. Chloride (0.2 mg/L)
FIG. 3 Chromatography of a Mixed Anion Standard with a CRD-300 (A) and without a CRD-300 (B)
required for this analysis are: (1) a background conductance of 20 to 25 μS/cm and (2) a peak-to-peak (noise) variation of no
greater than 5 nS/cm per minute of monitored baseline response.
15. Reagents and Materials
15.1 Eluent, Concentrate (90.0 mM (90.0-mM Sodium Carbonate) —Dissolve 9.540 g of sodium carbonate in 1000 mL of water.
15.2 Eluent, Analysis (9.0 mM (9.0-mM Sodium Carbonate)—Dilute 100.0 mL of Eluent Concentrate (see 15.1) to 1.000 L with
water.
15.2.1 The Eluent Analysis solution (9.0 mM Sodium Carbonate) Solution (9.0-mM sodium sarbonate) must be purged for 10
minutes with helium prior to use to remove dissolved gasses in order to ensure optimal system performance.
15.3 Ethylenediamine (EDA) Preservation Solution (50.0 g/L)—Dilute 11.2 mL of ethylenediamine (99 %) to 200 mL with
reagent water. Prepare this solution fresh monthly. Add 1.00 mL of this solution per 1.000 L of blank, standard or sample to produce
a final EDA concentration of 50 mg/L.
D6581 − 18
NOTE 1—Peaks:
1. Fluoride (0.5 mg/L) 4. Chloride (50 mg/L) 6. Unknown
2. Chlorite (10, 20, and 40 μg/L) 5. Chlorate (10, 20, and 40 μg/L) 7. Sulfate (100 mg/L)
3. Bromate (10, 20, and 40 μg/L)
FIG. 4 Overlay of Chromatograms of Three Concentration Levels of Chlorite, Bromate, and Chlorate in a Mixed Anion Standard with a
CRD-300 (A) and without a CRD-300 (B)
TABLE 2 Calibration Report for Chlorite, Bromate, and Chlorate
with a CRD-300 (A) and without a CRD-300 (B)
R-Square (%)
Peak Name Points
A
Chlorite 3 99.9961 99.9748
Bromate 3 100.0000 99.9986
Chlorate 3 99.9995 99.9637
D6581 − 18
TABLE 3 Determination of Bromate and Chlorate in a Bottled Mineral Water Sample
A B
Injection No. (μg/L) (μg/L)
Bromate Chlorate Bromate Chlorate
1 11.0 1.52 5.33 ND
2 10.9 1.55 6.23 ND
3 10.9 1.35 5.02 ND
4 10.1 1.91 6.25 ND
5 11.3 1.48 5.89 ND
Average 10.8 1.56 5.74 —
RSD 4.34 13.42 9.61 —
TABLE 4 Spike Recovery of Bromate, Chlorate, and Chlorite in Mineral Water Using a System with a CRD-300 (A) and without a CRD-
300 (B)
A B
Injection No. (μg/L) (μg/L)
Chlorite Bromate Chlorate Chlorite Bromate Chlorate
A
Sample ND 10.83 1.56 ND 5.74 ND
Spike 10 10 10 10 10 10
B
Measured Amount 9.88 20.51 12.02 8.58 15.30 8.50
RSD 2.39 1.60 2.45 2.39 1.60 2.45
Recovery (%) 98.8 98.5 104 85.8 97.2 85.0
A
ND = Not Detected
B
The average of five injections.
15.4 SPE Sample Pretreatment Cartridges—Chloride present at >200 mg/L and carbonate present at >300 mg/L can interfere
+ +
with bromate determination. H form and Ag form cation exchange SPE cartridges can be used to minimize the carbonate and
chloride interferences, respectively, if required. OnGuard-H and OnGuard-Ag cartridges have been shown to be suitable for this
application. The use of these pretreatment cartridges will effect recoveries for bromide, requiring that it be analyzed in a separate
run.
15.5 Suppressor Regenerant Solution—If a suppressor requiring chemical regeneration is used, the regenerant solution is
prepared by cautiously adding 3.00 mL of concentrated sulfuric acid (sp. gr. 1.84) to 4.000 L of water. If an Anion Self
Regenerating Suppressor is used, it should be operated in the external water mode.
15.6 Standard Solutions, Stock (1.00 mL = 1.00 mg)—Purchase certified solutions or prepare stock standard solutions from the
following salts, as described below:
—– —–
15.6.1 Bromate (BrO ) Solution, Stock (1.00 mL = 1.00 mg BrO )—Dissolve 1.180 g of sodium bromate (NaBrO ) in
3 3 3
water and dilute to 1.000 L.
—– —–
15.6.2 Bromide (Br ) Solution, Stock (1.00 mL = 1.00 mg Br )—Dissolve 1.288 g of sodium bromide (NaBr) in water and
dilute to 1.000 L.
—– —–
15.6.3 Chlorate (ClO ) Solution, Stock (1.00 mL = 1.00 mg C1O )—Dissolve 1.275 g of sodium chlorate (NaClO ) in
3 3 3
water and dilute to 1.000 L.
—– —–
15.6.4 Chlorite (ClO ) Solution, Stock (1.00 mL = 1.00 mg ClO )—Dissolve 1.680 g of sodium chlorite (NaClO ) in water
2 2 2
and dilute to 1.000 L. Note that as sodium chlorite is usually available only as an 80 % technical grade salt, the 80 % purity is
accounted for in the 1.680 g 1.680-g weight cited above. If an alternate purity is used, make an appropriate adjustment in the weight
2,7
of salt used after determining the exact percentage of NaClO , which can be done using an iodometric titration procedure.
15.7 Reagent Blank—Add 1.00 mL of EDA Preservation Solution (see 15.3) to 1.000 L of reagent water.
16. Calibration and Standardization
16.1 Typical Range of Applicability—This test method is applicable to the determination of bromate, bromide, chlorate, and
chlorite in raw water, finished drinking water and bottled (non-carbonated) water. The application ranges tested for each analyte
are as follows: bromate; 5–30 μg/L, bromide; 20–200 μg/L, chlorite; 20–500 μg/L, and chlorate; 20–500 μg/L.
16.2 Calibration Standards—For each individual calibration curve, prepare calibration standards, at a minimum of three
concentration levels, by accurately adding measured volumes of the stock standards (see 15.6) to a volumetric flask(s). Add 50
mg/L of EDA (the equivalent of 1.00 mL of EDA Preservation Solution (see 15.3) per 1.000 L of solution) to the volumetric flask(s)
and dilute to volume with reagent water. A minimum of five concentration levels is recommended if the curve covers two orders
of magnitude.
Joyce, R. J., and Dhillon, H. J., Chromatography, Vol 671, 1994, p. 165.
Method 4500–ClO .C in A. E. Greenberg, L. S. Clesceri, A. D. Eaton (Eds.), Standard Methods for the Examination of Water and Wastewater, 18th Ed., APHA,
Washington, DC, 1992.
D6581 − 18
16.3 Calibration Curve—To establish the calibration curve, analyze a reagent blank and the calibration standards in accordance
to the procedure in Section 17, using a 200 μL 200-μL injection (with a 4 mm 4-mm ID column) or a 50 μL 50-μL injection (with
a 2 mm 2-mm ID column). Tabulate peak area responses against concentration. These results are used to prepare a calibration curve
using a linear least squares fit for each analyte. The squared correlation coefficient of the regression (r ) should be ≥ 0.995 for
accurate results. Once the calibration curves have been established, verification must be performed on each analysis day, whenever
fresh eluent is prepared, and twice each batch of samples, as outlined in 20.4 and 20.5.
17. Procedure
17.1 Inject the reagent blank, calibration standard or sample into the eluent stream and record the chromatogram. In the case
of a manual injector, flush an excess of the sample (minimum of 5x5× loop volume) through the sample injection port using a
syringe prior to injection. A 200-μL injection is required when using a 4 mm 4-mm ID column, a 50 μL 50-μL injection is required
when using a 2 mm 2-mm ID column, in order to achieve the required detection limits for this analysis. An example of a
chromatogram of low level oxyhalides and bromide is shown in Fig. 1. An example chromatogram of low level oxyhalides and
bromide in a modest ionic strength, simulated drinking water is shown in Fig. 2.
18. Calculation
18.1 Compare the peak areas for the anions in the sample to the calibration curves prepared in 16.3 to calculate and report the
anion concentration in μg/L:
Anion concentration, µg/L5 A 3F (1)
where:
A = reading from the appropriate calibration plot, in μg/L, and
F = dilution factor if the sample was diluted prior to analysis.
18.1.1 Computing integrators and computer based chromatographic data systems can be programmed to perform these
calculations automatically.
18.2 Report only those values that fall between the lowest and highest calibration standards. Samples exceeding the highest
standard should be diluted and reanalyzed.
19. Precision and Bias
19.1 The precision and bias data presented in this test method meets the requirements of Practice D2777 – 98, which was in
place at the time of collaborative testing. Under the allowances made in 1.4 of Practice D2777 – 06,13, these precision and bias
data do meet existing requirements for interlaboratory studies of Committee D19 test methods. The full research report can be
obtained from ASTM Headquarters.
19.2 The interlaboratory study that generated the precision and bias data in this test method was performed in reagent water,
municipal drinking water, and bottled (non-carbonated) water by ten laboratories using one operator each. Six levels of
concentration were used for four analytes, producing three Youden pairs. The Youden pair data was used to calculate the single
operator precision (S ). The analytes were supplied separately as six (mixed) concentrates. The reagent water, municipal drinking
o
water and bottled water were supplied by the participating laboratories. Six reagent water samples, six bottled water samples, and
six municipal drinking water samples (for a total of 18 samples) were prepared by pipetting 1.0 mL aliquots of the concentrates
labeled A1-2 (×3), B1-2 (×3), C1-2 (×3) into volumetric flasks (18 total); adding 50 mg/L EDA preservation solution, as detailed
in 7.2, and diluting to a total of 100 mL with reagent water (×6), bottled water (×6), and drinking water (×6), as appropriate.
19.2.1 A quality control (QC) sample was supplied (as a concentrate) to serve as initial, and on-going, calibration verification.
A separate method detection limit (MDL) sample was supplied (as a concentrate) for the determination of the pooled MDL values.
The QC sample was prepared by pipetting a 1.0 mL 1.0-mL aliquot of the QC concentrate into a clean volumetric flask; adding
50 mg/L 50-mg/L EDA, and diluting to a total of 100 mL with reagent water. The MDL sample was prepared by pipetting a 1.0
mL 1.0-mL aliquot of the MDL concentrate into a clean volumetric flask; adding 50 mg/L 50-mg/L EDA, and diluting to a total
of 100 mL with reagent water.
19.3 All the precision and bias data presented in this test method was obtained using the IonPac AS9-HC column listed in Table
1.
19.4 The precision and bias of this test method for each analyte for reagent, drinking, and bottled water are shown in Tables
5-8.
19.5 The results of the interlaboratory study can also be summarized as regression equations, as shown in Table 9 for reagent
water and in Table 10 for a typical sample matrix of drinking water.
Supporting data have been filed at ASTM International Headquarters and may be obtained by requesting Research Report RR:D19-1164. Contact ASTM Customer
Service at service@astm.org.
D6581 − 18
TABLE 5 Determination of Precision and Bias for Chlorite
Amount Added Amount Found Number Retained S S Bias
o t
Water
(μg/L) (μg/L) Parts (μg/L) (μg/L) (%)
Reagent 20 19.94 8 1.40 1.25 –0.3
25 25.06 1.05 0.2
180 178.29 8 4.37 5.64 –1.0
220 214.73 6.18 –2.4
400 394.36 8 19.39 21.81 –1.4
450 440.53 8.29 –2.1
Drinking 20 19.19 8 1.52 6.58 –4.1
25 23.77 6.17 –4.9
180 174.30 8 5.06 9.29 –3.2
220 216.89 14.76 –1.4
400 398.30 8 4.42 15.56 –0.4
450 439.85 19.59 –2.3
Bottled 20 20.94 8 4.41 3.65 4.7
25 22.74 4.64 –9.0
180 177.71 8 2.95 8.76 –1.3
220 216.16 8.74 –1.7
400 390.14 8 9.53 13.65 –2.5
450 433.72 15.30 –3.6
TABLE 6 Determination of Precision and Bias for Bromate
Amount Added Amount Found Number Retained S S Bias
o t
Water
(μg/L) (μg/L) Parts (μg/L) (μg/L) (%)
Reagent 5 4.95 9 0.99 1.19 –0.9
7 7.84 1.29 12.0
10 9.98 9 0.66 0.99 –0.2
12 11.76 0.55 –2.0
20 19.56 9 2.17 2.37 –2.2
25 24.18 1.53 –3.3
Drinking 5 4.41 6 0.62 1.20 –11.8
7 6.44 0.78 –8.0
10 8.54 8 0.72 2.88 –14.6
12 10.20 2.88 –15.0
20 17.31 8 2.80 2.85 –13.4
25 20.51 4.77 –17.9
Bottled 5 4.95 6 1.09 1.51 –1.1
7 7.21 1.80 3.0
10 9.07 6 1.67 4.83 –9.3
12 10.35 3.41 –13.7
20 20.16 6 4.34 3.91 0.8
25 20.99 7.75 –16.0
TABLE 7 Determination of Precision and Bias for Bromide
Amount Added Amount Found Number Retained S S Bias
o t
Water
(μg/L) (μg/L) Parts (μg/L) (μg/L) (%)
Reagent 20 20.75 9 1.94 1.91 3.8
25 25.51 2.32 2.1
75 74.52 9 3.80 2.94 –0.6
100 99.42 4.84 –0.6
150 143.50 9 5.79 5.82 –4.3
180 176.38 5.24 –2.0
Drinking 20 20.68 8 1.30 4.39 3.4
25 25.49 3.31 2.0
75 71.89 8 4.67 5.67 –4.2
100 97.05 6.90 –3.0
150 145.81 8 1.23 8.39 –2.8
180 173.40 9.12 –3.7
Bottled 20 20.25 7 2.21 1.79 1.3
25 26.23 1.11 4.9
75 74.35 8 6.45 4.69 –0.9
100 98.49 5.00 –1.5
150 142.67 8 6.57 6.08 –4.9
180 172.71 9.24 –4.1
19.6 In addition to performing the analyses required to generate the precision and bias data shown in Tables 5-8, the
participating laboratories each analyzed seven replicates of an MDL sample. The MDLs were derived for each laboratory using
the students t-test at six degrees of freedom, as follows:
D6581 − 18
TABLE 8 Determination of Precision and Bias for Chlorate
Amount Added Amount Found Number Retained S S Bias
o t
Water
(μg/L) (μg/L) Parts (μg/L) (μg/L) (%)
Reagent 20 20.69 7 2.73 2.43 3.5
25 26.64 3.79 6.6
180 176.05 8 11.96 3.70 –2.2
220 215.39 7.47 –2.1
400 393.00 7 18.27 5.85 –1.7
450 443.47 16.50 –1.5
Drinking 20 19.94 9 1.81 3.95 –0.3
25 23.93 5.13 –4.3
180 175.10 9 6.92 9.05 –2.7
220 216.14 7.02 –1.8
400 396.74 9 4.74 16.55 –0.8
450 441.69 16.55 –1.8
Bottled 20 21.72 8 2.86 3.88 8.6
25 25.75 3.21 3.0
180 179.82 8 3.59 5.37 –0.1
220 217.58 9.26 –1.1
400 389.51 7 6.72 15.83 –2.6
450 443.70 10.00 –1.4
TABLE 9 Summary of Precision and Bias Results for Reagent Water
Chlorite Bromate Bromide Chlorate
Precision and Bias Summary Precision and Bias Summary Precision and Bias Summary Precision and Bias Summary
Number of Laboratories 10 10 10 10
Range Tested 20–450 μg/L 5–25 μg/L 20–180 μg/L 20–450 μg/L
Mean Recovery y = 0.9805x + 0.5261 y = 0.9432x + 0.6272 y = 0.9629x + 1.7475 y = 0.9809x + 0.8245
S y = 0.0465x – 1.4801 y = 0.0878x + 0.1281 y = 0.0282x + 1.3087 y = 0.0389x + 2.7278
o
S y = 0.0332x + 0.3294 y = 0.046x + 0.721 y = 0.0246x + 1.6352 y = 0.0226x + 1.8244
t
TABLE 10 Summary of Precision and Bias Results for Drinking Water
Chlorite Bromate Bromide Chlorate
Precision and Bias Summary Precision and Bias Summary Precision and Bias Summary Precision and Bias Summary
Number of Laboratories 10 10 10 10
Range Tested 20–450 μg/L 5–25 μg/L 20–180 μg/L 20–450 μg/L
Mean Recovery y = 0.9872x – 1.0243 y = 0.9432x + 0.6272 y = 0.9583x + 1.2113 y = 0.9868x – 0.7347
S y = 0.0068x + 2.2164 y = 0.1721x – 0.5532 y = –0.0022x + 2.6 y = 0.0066x + 3.0956
o
S y = 0.0289x + 5.8552 y = 0.1934x + 0.3866 y = 0.0357x + 3.1189 y = 0.03x + 3.3368
t
MDL 5 t 3 S (2)
~ ! ~ !
where:
t = students t value for a 99 % confidence level and a standard deviation estimate with n-1 degrees of freedom [t = 3.14 for seven
replicates], and
S = standard deviation of the replicate analysis.
19.6.1 True amounts injected, mean value determined, and pooled MDL values (10 laboratories × 7 replicates) are shown in
Table 11.
20. Quality Control
20.1 Before this test method is applied to analyzing unknown samples, the analyst should establish quality control procedures
as recommended in Guide D3856.
TABLE 11 Pooled MDL Values Obtained for This Test Method
Analyte Injected Amount Mean Value Pooled MDL
Chlorite 3.0 μg/L 3.32 μg/L 2.39 μg/L
Bromate 4.0 μg/L 3.98 μg/L 2.73 μg/L
Bromide 4.0 μg/L 3.96 μg/L 2.91 μg/L
Chlorate 4.0 μg/L 3.74 μg/L 3.49 μg/L
D6581 − 18
20.2 The laboratory using this test should perform an initial demonstration of laboratory capability. Analyze seven replicates
of an Initial Demonstrationinitial demonstration of Performanceperformance (IDP) solution. The IDP solution contains method
analytes of known concentration, prepared from a different source to the calibration standards, used to fortify reagent water, which
also contains a final EDA concentration of 50 mg/L (see 15.3). Ideally, the IPD solution should be prepared by an independent
source from reference materials. The level 3 standard used for the method precision and bias study is recommended as an IDP
solution.
20.2.1 The mean and standard deviation of the seven values should then be calculated and compared, according to Practice
D5847, to the single operator precision and recovery established for this test method. The upper limit for acceptable precision and
the range of acceptable recoveries are detailed below:
IDP Solution Acceptable IDP
Analyte Method S
Amount Precision, n = 7
Chlorite 180 μg/L 4.4 μg/L # 11.8 μg/L
Bromate 10 μg/L 0.66 μg/L # 1.67 μg/L
Bromide 75 μg/L 3.8 μg/L # 9.6 μg/L
Chlorate 180 μg/L 12.0 μg/L # 32.1 μg/L
Method Mean Lower Acceptable Upper Acceptable
Analyte
Recovery IDP Recovery IDP Recovery
Chlorite 178.3 μg/L 164.6 μg/L 191.7 μg/L
Bromate 9.98 μg/L 7.37 μg/L 12.59 μg/L
Bromide 74.5 μg/L 70.8 μg/L 78.2 μg/L
Chlorate 176.1 μg/L 171.2 μg/L 181.0 μg/L
20.2.2 The S and mean recovery values can be calculated for different IDP solution concentrations using the regression
o
equations for each analyte shown in Table 9. If the values obtained for the IDP precision and recovery do not meet the criteria
described above, initial demonstration of performance must be repeated until the results fall within these criteria.
20.3 When beginning use of this method, an initial Calibration Verification Standardcalibration verification standard (CVS)
should be used to verify the calibration standards and acceptable instrument performance. This verification should be performed
on each analysis day or whenever fresh eluent has been prepared. As this method is intended for use at trace levels, a low level
CVS (that is, equivalent to the lowest calibration standard) should initially be analyzed before beginning use of this method. The
CVS is a solution of method analytes of known concentration used to fortify reagent water, which also contains a final EDA
concentration of 50 mg/L (see 15.3). If the determined low level CVS values are not within 625 % of the known amounts, the
low level CVS should be reanalyzed. If the values still fall outside acceptable limits, a new calibration curve is required which must
be confirmed by a successful low level CVS before continuing with on-going analyses.
20.4 A continuing CVS should be analyzed after every tenth field sample and an end CVS should analyzed at the end of the
sample batch (maximum of 20 samples) to verify the previously established calibration curves. After initially meeting the
requirements of 20.3, the levels selected for the continuing and end CVS should be varied between a middle calibration level and
the highest calibration level standard. If the continuing and end CVS values are not within 615 % of the known amounts, the
analyst should reanalyze the CVS. If the analyte concentrations still fall outside acceptable limits (615 %) that analyte is judged
out of control, and the source of the problem should be identified before continuing with on-going analyses. All samples following
the last acceptable CVS should be reanalyzed.
20.5 A reagent blank (see 15.7) should be run when generating the initial calibration curves. A blank should also be run with
each sample batch (maximum of 20 samples) to check for sample or system contamination.
20.6 One Laboratory Control Samplelaboratory control sample (LCS) should be used with each sample batch (maximum of 20
samples). The LCS is a solution of method analytes of known concentration added to a matrix which sufficiently challenges the
Test Method. A synthetic drinking water matrix, containing fluoride at 1.0 mg/L, chloride at 50 mg/L, nitrite at 0.1 mg/L, nitrate
at 10 mg/L, phosphate at 0.1 mg/L and sulfate at 50 mg/L, spiked with the four method analytes at the level of the IDP solution
would be an example of an appropriate LCS. The LCS shall also contain 50 mg/L of EDA (the equivalent of 1.00 mL of EDA
Preservation Solution (see 15.3) per 1.000 L of solution).
20.6.1 The analyte recoveries for the LCS should fall within the control limits of x 6 3S, where x is the mean recovery and
(S) is the standard deviation of the mean recovery established from the interlaboratory precision and bias study data at the IDP
levels, as shown below:
Lower Recovery Upper Recovery
Analyte LCS Amount
Limit Limit
Chlorite 180 μg/L 165 μg/L 191 μg/L
Bromate 10 μg/L 8.0 μg/L 12.0 μg/L
Bromide 75 μg/L 63 μg/L 86 μg/L
Chlorate 180 μg/L 140 μg/L 219 μg/L
D6581 − 18
20.7 One Matrix Spikematrix spike (MS) should be run with each sample batch (maximum of 20 samples) to test method
recovery. The MS should be prepared in accordance with Guide D5810. Spike a portion of a drinking water (or other) sample from
each batch with the four method analytes at the level of the IDP solution. The % recovery of the spike should fall within limits
established from the interlaboratory precision and bias study data (assuming a background level of zero), according to Practice
D5847, as shown below:
Lower Recovery Upper Recovery
Analyte MS Amount
Limit (%) Limit (%)
Chlorite 180 μg/L 90.9 % 109.1 %
Bromate 10 μg/L 73.1 % 126.9 %
Bromide 75 μg/L 80.8 % 119.2 %
Chlorate 180 μg/L 88.3 % 111.7 %
20.8 One Matrix Duplicatematrix duplicate (MD) should be run with each sample batch (maximum of 20 samples) to test
method precision. If non-detects are expected in all the samples to be analyzed, a Matrix Spike Duplicatematrix spike duplicate
should be run instead. The precision of the duplicate analysis should be compared, according to Practice D5847, to the nearest
tabulated S value established from the interlaboratory precision and bias study data for each analyte.
o
20.9 In order to verify the quantitative values produced by the test method, an Independent Reference Material (IRM), submitted
to the laboratory as a regular sample (if practical), should be analyzed once per quarter. The concentration of the IRM should be
within the scope of the method, as defined in 1.1. The values obtained must fall within the limits specified by the outside source.
20.10 The laboratory may perform additional quality control as desired or appropriate, for instance the use of a surrogate as
outlined in Section 9.4.2 of U.S. EPA Method 300.1. In addition, it is recommended that a laboratory determine the method
detection limits, as discussed in 19.6, before using this test method.
Test MethodTEST METHOD B
Electrolytically Suppressed Ion ChromatographyELECTROLYTICALLY SUPPRESSED ION CHROMATOGRAPHY
21. Scope
21.1 This test method is technically consistent with USU.S. EPA Method 300.1 (Part B), as cited above, except that it uses
analyte separation on a hydroxide-selective anion exchange column, using a hydroxide eluent followed by electrolytically
suppressed conductivity detection of the analytes in a deionized water matrix, thus improving method detection limits. The range
tested for each analyte were as follows:
Chlorite 20 to 1,000 μg/L
Chlorite 20 to 1000 μg/L
Bromate 1 to 30 μg/L
Chlorate 20 to 1,000 μg/L
Chlorate 20 to 1000 μg/L
Bromide 20 to 200 μg/L
21.1.1 It should be noted that the U.S. EPA maintains that the use of hydroxide-based separation is consistent with Method 300.1
(Parts A and B) for compliance monitoring.
22. Summary of Test Method B
22.1 Oxyhalides (chlorite, bromate, and chlorate) and bromide in raw water, finished drinking water and bottled water are
determined by ion chromatography. A sample (200 μL) is injected into an ion chromatograph and the pumped hydroxide eluent
sweeps the sample through the analytical column set. Here, anions are separated from the sample matrix according to their
retention characteristics, relative to the anions in the eluent.
22.1.1 The separated anions in the eluent stream then pass through a suppressor device, where all cations are exchanged for
hydronium ions. This converts the eluent to water, thus reducing the background conductivity. This process also converts the
sample anions to their acid form, thus enhancing their conductivity. The eluent stream then passes through a conductivity cell,
where they are detected. An appropriate computer-based data system is typically used for data presentation.
22.2 The anions are identified based on their retention times compared to known standards. Quantification is accomplished by
measuring anion peak areas and comparing them to the areas generated from known standards.
23. Interferences
23.1 Positive errors can be caused by progressive oxidation of residual hypochlorite or hypobromite, or both, in the sample to
the corresponding chlorate and bromate. Furthermore, chlorite can also be oxidized to chlorate, causing negative errors for chlorite
and positive errors for chlorate. These interferences are eliminated by the sample preservation steps outlined in 26.3. Chloride
present at >200 mg/L and carbonate present at >300 mg/L can interfere with bromate determination. These interferences can be
minimized, or eliminated, by the sample pretreatment steps outlined in 26.4. Fluoride and low molecular weight monocarboxylic
acids, present at mg/L concentrations, may interfere with the quantitation of chlorite and bromate.
D6581 − 18
24. Apparatus
24.1 Ion Chromatography Apparatus—Analytical system complete with all required accessories, including eluent pump,
injector, syringes, columns, suppressor, conductivity detector, data system and compressed gasses.
24.1.1 Eluent Pump—Capable of delivering 0.25 to 5 mL/min of eluent at a pressure of up to 4000 psi.27 600 kPa (4000 psi).
24.1.2 Injection Valve—A low dead-volume switching valve that will allow the loading of a sample into a sample loop and
subsequent injection of the loop contents into the eluent stream. A loop size of up to 250 μL may be used without compromising
the resolution of early eluting peaks, such as chlorite and bromate.
24.1.3 Guard Column—Anion exchange column typically packed with the same material used in the analytical column, for
example, AG19, or equivalent. The purpose of this column is to protect the analytical column from particulate m
...



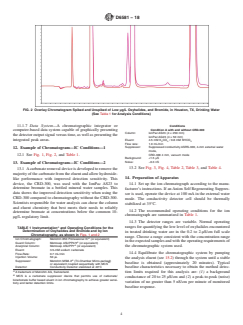




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...