ASTM D7757-22
(Test Method)Standard Test Method for Silicon in Gasoline and Related Products by Monochromatic Wavelength Dispersive X-ray Fluorescence Spectrometry
Standard Test Method for Silicon in Gasoline and Related Products by Monochromatic Wavelength Dispersive X-ray Fluorescence Spectrometry
SIGNIFICANCE AND USE
5.1 This test method provides rapid and precise measurement of total silicon in naphthas, gasoline, gasoline-ethanol blends, RFG, ethanol and ethanol-fuel blends, and toluene with minimum sample preparation. Typical analysis time is 5 min to 10 min per sample.
5.2 Excitation by monochromatic X-rays reduces background, simplifies matrix correction, and increases the signal/background ratio compared to polychromatic excitation used in conventional WDXRF techniques.3
5.3 Silicone oil defoamer can be added to coker feedstocks to minimize foaming in the coker. Residual silicon in the coker naphtha can adversely affect downstream catalytic processing of the naphtha. This test method provides a means to determine the silicon content of the naphtha.
5.4 Silicon contamination of gasoline, gasoline-ethanol blends, denatured ethanol, and their blends has led to fouled vehicle components (for example, spark plugs, exhaust oxygen sensors, catalytic converters) requiring parts replacement and repairs. Finished gasoline, gasoline-ethanol blends, and ethanol-fuel blends can come into contact with silicon a number of ways. Waste hydrocarbon solvents such as toluene can be added to gasoline. Such solvents can contain soluble silicon compounds. Silicon-based antifoam agents can be used in ethanol plants, which then pass silicon on to the finished ethanol-fuel blend. This test method can be used to determine if gasoline, gasoline-ethanol blends, and ethanol-fuel blends meet specifications with respect to silicon content of the fuel, and for resolution of customer problems.
5.5 Some silicon compounds covered by this test method are significantly more volatile than the silicon compounds typically used for the preparation of the calibration standards. Volatile compounds may not meet the stated precision from this test method because of selective loss of light materials during the analysis.
SCOPE
1.1 This test method covers the determination of total silicon by monochromatic, wavelength-dispersive X-ray fluorescence (MWDXRF) spectrometry in naphthas, gasoline, gasoline-ethanol blends, reformulated gasoline (RFG), ethanol and ethanol-fuel blends, and toluene at concentrations of 3 mg/kg to 100 mg/kg. The precision of this test method was determined by an interlaboratory study using representative samples of the liquids described in 1.1 and 1.2. The pooled limit of quantitation (PLOQ) was estimated to be 3 mg/kg.
Note 1: Volatile samples such as high-vapor-pressure gasolines or light hydrocarbons might not meet the stated precision because of the evaporation of light components during the analysis.
Note 2: Aromatic compounds such as toluene are under the jurisdiction of Committee D16 on Aromatic Hydrocarbons and Related Chemicals. However, toluene can be a contributor to silicon contamination in gasoline (see 5.4), thus its inclusion in this test method.
1.2 Gasoline samples containing ethanol and other oxygenates may be analyzed with this test method provided the matrix of the calibration standards is either matched to the sample matrices or the matrix correction described in Annex A1 is applied to the results. The conditions for matrix matching and matrix correction are provided Section 6, Interferences.
1.3 Samples with silicon concentrations above 100 mg/kg can be analyzed after dilution with appropriate solvent. The precision and bias of silicon determinations on diluted samples have not been determined and may not be the same as shown for neat samples (Section 17).
1.4 A fundamental assumption in this test method is that the standard and sample matrices are well matched, or that the matrix differences are accounted for (see 14.5). Matrix mismatch can be caused by C/H ratio differences between samples and standards or by the presence of other interfering heteroatoms; observe the cautions and recommendations in Section 6.
1.5 The values stated in SI units are to be regarded as standard...
General Information
- Status
- Published
- Publication Date
- 31-Oct-2022
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.03 - Elemental Analysis
Relations
- Effective Date
- 01-Mar-2024
- Refers
ASTM D4175-23a - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 15-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Refers
ASTM D4175-23e1 - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 01-Jul-2023
- Effective Date
- 01-Dec-2019
- Effective Date
- 15-Sep-2019
- Effective Date
- 01-Jul-2019
- Effective Date
- 01-May-2019
- Effective Date
- 01-Mar-2019
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Dec-2018
- Effective Date
- 01-Dec-2018
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Jul-2018
Overview
ASTM D7757-22 sets the standard test method for determining total silicon in gasoline, gasoline-ethanol blends, naphthas, reformulated gasoline (RFG), ethanol, ethanol-fuel blends, and toluene using monochromatic wavelength dispersive X-ray fluorescence spectrometry (MWDXRF). The method provides rapid, precise analysis with minimal sample preparation, supporting accurate silicon measurement at concentrations from 3 mg/kg up to 100 mg/kg. This test is essential for monitoring silicon contamination that can negatively impact fuel quality and automotive components.
Key Topics
- Rapid, Precise Silicon Measurement: Analysis can be completed within 5 to 10 minutes per sample, making it suitable for high-throughput environments.
- Applicable Fuels and Solvents: The method covers naphthas, gasoline, gasoline-ethanol blends, RFG, ethanol, ethanol-fuel blends, and toluene.
- Monochromatic X-ray Excitation: This technology reduces background interference, improves the signal-to-noise ratio, and simplifies matrix corrections compared to conventional WDXRF methods.
- Matrix Matching and Corrections: Proper matrix matching or correction is crucial for accurate results, especially when testing oxygenate-rich fuels or when sample composition differs significantly from calibration standards.
- Detection Range: The test method quantifies silicon content within 3 mg/kg to 100 mg/kg, with capabilities for higher concentrations through sample dilution.
- Quality Assurance: The method includes detailed guidance for calibration, drift correction, and use of quality control samples to ensure consistent, reliable measurements.
Applications
- Fuel Quality Control: Routine use in refineries and blending facilities to ensure that gasoline, gasoline-ethanol blends, and ethanol-fuel blends meet regulatory specifications for silicon content.
- Contamination Troubleshooting: Identifies silicon contamination sources that can cause engine and vehicle component failures, such as spark plug fouling or catalytic converter damage.
- Process Monitoring: Evaluates the impact of silicon-based antifoam agents or waste solvents (like toluene), which can introduce silicon to fuels.
- Downstream Protection: Prevents adverse effects of residual silicon from coker units on downstream catalytic processing by controlling silicon in naphthas.
- Compliance: Assists laboratories in demonstrating compliance with fuel specifications and in resolving customer or regulatory concerns regarding silicon content.
- Research and Development: Supports development of new fuel formulations and quality assurance plans that minimize silicon risks.
Related Standards
- ASTM D4057: Manual Sampling of Petroleum and Petroleum Products
- ASTM D4175: Terminology for Petroleum Products, Liquid Fuels, and Lubricants
- ASTM D4177: Automatic Sampling of Petroleum and Petroleum Products
- ASTM D4806: Specification for Denatured Fuel Ethanol for Blending with Gasolines
- ASTM D5798: Specification for Ethanol Fuel Blends for Flexible-Fuel Engines
- ASTM D6299: Statistical Quality Assurance and Control Charting in Laboratories
- ASTM D6300: Precision and Bias Data for Test Methods
- ASTM D7343: Optimization and Validation of X-ray Fluorescence Spectrometry Methods
Practical Value
The ASTM D7757-22 standard is critical for laboratories, fuel producers, and quality control specialists concerned with maintaining the performance, reliability, and compliance of modern fuels. Its adoption helps minimize vehicle maintenance costs, supports effective troubleshooting of silicon-related issues, and ensures adherence to international fuel quality standards. The use of MWDXRF provides a precise, robust, and efficient tool for managing silicon in complex hydrocarbon matrices, making ASTM D7757-22 an essential resource for fuel testing and quality management programs.
Buy Documents
ASTM D7757-22 - Standard Test Method for Silicon in Gasoline and Related Products by Monochromatic Wavelength Dispersive X-ray Fluorescence Spectrometry
REDLINE ASTM D7757-22 - Standard Test Method for Silicon in Gasoline and Related Products by Monochromatic Wavelength Dispersive X-ray Fluorescence Spectrometry
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D7757-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Silicon in Gasoline and Related Products by Monochromatic Wavelength Dispersive X-ray Fluorescence Spectrometry". This standard covers: SIGNIFICANCE AND USE 5.1 This test method provides rapid and precise measurement of total silicon in naphthas, gasoline, gasoline-ethanol blends, RFG, ethanol and ethanol-fuel blends, and toluene with minimum sample preparation. Typical analysis time is 5 min to 10 min per sample. 5.2 Excitation by monochromatic X-rays reduces background, simplifies matrix correction, and increases the signal/background ratio compared to polychromatic excitation used in conventional WDXRF techniques.3 5.3 Silicone oil defoamer can be added to coker feedstocks to minimize foaming in the coker. Residual silicon in the coker naphtha can adversely affect downstream catalytic processing of the naphtha. This test method provides a means to determine the silicon content of the naphtha. 5.4 Silicon contamination of gasoline, gasoline-ethanol blends, denatured ethanol, and their blends has led to fouled vehicle components (for example, spark plugs, exhaust oxygen sensors, catalytic converters) requiring parts replacement and repairs. Finished gasoline, gasoline-ethanol blends, and ethanol-fuel blends can come into contact with silicon a number of ways. Waste hydrocarbon solvents such as toluene can be added to gasoline. Such solvents can contain soluble silicon compounds. Silicon-based antifoam agents can be used in ethanol plants, which then pass silicon on to the finished ethanol-fuel blend. This test method can be used to determine if gasoline, gasoline-ethanol blends, and ethanol-fuel blends meet specifications with respect to silicon content of the fuel, and for resolution of customer problems. 5.5 Some silicon compounds covered by this test method are significantly more volatile than the silicon compounds typically used for the preparation of the calibration standards. Volatile compounds may not meet the stated precision from this test method because of selective loss of light materials during the analysis. SCOPE 1.1 This test method covers the determination of total silicon by monochromatic, wavelength-dispersive X-ray fluorescence (MWDXRF) spectrometry in naphthas, gasoline, gasoline-ethanol blends, reformulated gasoline (RFG), ethanol and ethanol-fuel blends, and toluene at concentrations of 3 mg/kg to 100 mg/kg. The precision of this test method was determined by an interlaboratory study using representative samples of the liquids described in 1.1 and 1.2. The pooled limit of quantitation (PLOQ) was estimated to be 3 mg/kg. Note 1: Volatile samples such as high-vapor-pressure gasolines or light hydrocarbons might not meet the stated precision because of the evaporation of light components during the analysis. Note 2: Aromatic compounds such as toluene are under the jurisdiction of Committee D16 on Aromatic Hydrocarbons and Related Chemicals. However, toluene can be a contributor to silicon contamination in gasoline (see 5.4), thus its inclusion in this test method. 1.2 Gasoline samples containing ethanol and other oxygenates may be analyzed with this test method provided the matrix of the calibration standards is either matched to the sample matrices or the matrix correction described in Annex A1 is applied to the results. The conditions for matrix matching and matrix correction are provided Section 6, Interferences. 1.3 Samples with silicon concentrations above 100 mg/kg can be analyzed after dilution with appropriate solvent. The precision and bias of silicon determinations on diluted samples have not been determined and may not be the same as shown for neat samples (Section 17). 1.4 A fundamental assumption in this test method is that the standard and sample matrices are well matched, or that the matrix differences are accounted for (see 14.5). Matrix mismatch can be caused by C/H ratio differences between samples and standards or by the presence of other interfering heteroatoms; observe the cautions and recommendations in Section 6. 1.5 The values stated in SI units are to be regarded as standard...
SIGNIFICANCE AND USE 5.1 This test method provides rapid and precise measurement of total silicon in naphthas, gasoline, gasoline-ethanol blends, RFG, ethanol and ethanol-fuel blends, and toluene with minimum sample preparation. Typical analysis time is 5 min to 10 min per sample. 5.2 Excitation by monochromatic X-rays reduces background, simplifies matrix correction, and increases the signal/background ratio compared to polychromatic excitation used in conventional WDXRF techniques.3 5.3 Silicone oil defoamer can be added to coker feedstocks to minimize foaming in the coker. Residual silicon in the coker naphtha can adversely affect downstream catalytic processing of the naphtha. This test method provides a means to determine the silicon content of the naphtha. 5.4 Silicon contamination of gasoline, gasoline-ethanol blends, denatured ethanol, and their blends has led to fouled vehicle components (for example, spark plugs, exhaust oxygen sensors, catalytic converters) requiring parts replacement and repairs. Finished gasoline, gasoline-ethanol blends, and ethanol-fuel blends can come into contact with silicon a number of ways. Waste hydrocarbon solvents such as toluene can be added to gasoline. Such solvents can contain soluble silicon compounds. Silicon-based antifoam agents can be used in ethanol plants, which then pass silicon on to the finished ethanol-fuel blend. This test method can be used to determine if gasoline, gasoline-ethanol blends, and ethanol-fuel blends meet specifications with respect to silicon content of the fuel, and for resolution of customer problems. 5.5 Some silicon compounds covered by this test method are significantly more volatile than the silicon compounds typically used for the preparation of the calibration standards. Volatile compounds may not meet the stated precision from this test method because of selective loss of light materials during the analysis. SCOPE 1.1 This test method covers the determination of total silicon by monochromatic, wavelength-dispersive X-ray fluorescence (MWDXRF) spectrometry in naphthas, gasoline, gasoline-ethanol blends, reformulated gasoline (RFG), ethanol and ethanol-fuel blends, and toluene at concentrations of 3 mg/kg to 100 mg/kg. The precision of this test method was determined by an interlaboratory study using representative samples of the liquids described in 1.1 and 1.2. The pooled limit of quantitation (PLOQ) was estimated to be 3 mg/kg. Note 1: Volatile samples such as high-vapor-pressure gasolines or light hydrocarbons might not meet the stated precision because of the evaporation of light components during the analysis. Note 2: Aromatic compounds such as toluene are under the jurisdiction of Committee D16 on Aromatic Hydrocarbons and Related Chemicals. However, toluene can be a contributor to silicon contamination in gasoline (see 5.4), thus its inclusion in this test method. 1.2 Gasoline samples containing ethanol and other oxygenates may be analyzed with this test method provided the matrix of the calibration standards is either matched to the sample matrices or the matrix correction described in Annex A1 is applied to the results. The conditions for matrix matching and matrix correction are provided Section 6, Interferences. 1.3 Samples with silicon concentrations above 100 mg/kg can be analyzed after dilution with appropriate solvent. The precision and bias of silicon determinations on diluted samples have not been determined and may not be the same as shown for neat samples (Section 17). 1.4 A fundamental assumption in this test method is that the standard and sample matrices are well matched, or that the matrix differences are accounted for (see 14.5). Matrix mismatch can be caused by C/H ratio differences between samples and standards or by the presence of other interfering heteroatoms; observe the cautions and recommendations in Section 6. 1.5 The values stated in SI units are to be regarded as standard...
ASTM D7757-22 is classified under the following ICS (International Classification for Standards) categories: 75.160.20 - Liquid fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7757-22 has the following relationships with other standards: It is inter standard links to ASTM D6300-24, ASTM D4175-23a, ASTM D6300-23a, ASTM D6299-23a, ASTM D4175-23e1, ASTM D6300-19a, ASTM D4806-19a, ASTM D5798-19b, ASTM D5798-19a, ASTM D4806-19, ASTM D5798-19, ASTM D4806-18a, ASTM D5798-18a, ASTM D5798-18, ASTM D4806-18. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7757-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7757 − 22
Standard Test Method for
Silicon in Gasoline and Related Products by Monochromatic
Wavelength Dispersive X-ray Fluorescence Spectrometry
This standard is issued under the fixed designation D7757; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 1.5 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this
1.1 This test method covers the determination of total
standard.
silicon by monochromatic, wavelength-dispersive X-ray fluo-
1.6 This standard does not purport to address all of the
rescence (MWDXRF) spectrometry in naphthas, gasoline,
safety concerns, if any, associated with its use. It is the
gasoline-ethanol blends, reformulated gasoline (RFG), ethanol
responsibility of the user of this standard to establish appro-
and ethanol-fuel blends, and toluene at concentrations of
priate safety, health, and environmental practices and deter-
3 mg⁄kg to 100 mg⁄kg. The precision of this test method was
mine the applicability of regulatory limitations prior to use.
determined by an interlaboratory study using representative
1.7 This international standard was developed in accor-
samples of the liquids described in 1.1 and 1.2. The pooled
dance with internationally recognized principles on standard-
limit of quantitation (PLOQ) was estimated to be 3 mg⁄kg.
ization established in the Decision on Principles for the
NOTE 1—Volatile samples such as high-vapor-pressure gasolines or
Development of International Standards, Guides and Recom-
light hydrocarbons might not meet the stated precision because of the
mendations issued by the World Trade Organization Technical
evaporation of light components during the analysis.
Barriers to Trade (TBT) Committee.
NOTE 2—Aromatic compounds such as toluene are under the jurisdic-
tion of Committee D16 on Aromatic Hydrocarbons and Related Chemi-
2. Referenced Documents
cals. However, toluene can be a contributor to silicon contamination in
gasoline (see 5.4), thus its inclusion in this test method. 2
2.1 ASTM Standards:
D4057 Practice for Manual Sampling of Petroleum and
1.2 Gasoline samples containing ethanol and other oxygen-
Petroleum Products
ates may be analyzed with this test method provided the matrix
D4175 Terminology Relating to Petroleum Products, Liquid
of the calibration standards is either matched to the sample
Fuels, and Lubricants
matrices or the matrix correction described in Annex A1 is
D4177 Practice for Automatic Sampling of Petroleum and
applied to the results. The conditions for matrix matching and
Petroleum Products
matrix correction are provided Section 6, Interferences.
D4806 Specification for Denatured Fuel Ethanol for Blend-
1.3 Samples with silicon concentrations above 100 mg⁄kg
ing with Gasolines for Use as Automotive Spark-Ignition
can be analyzed after dilution with appropriate solvent. The
Engine Fuel
precision and bias of silicon determinations on diluted samples
D5798 Specification for Ethanol Fuel Blends for Flexible-
have not been determined and may not be the same as shown
Fuel Automotive Spark-Ignition Engines
for neat samples (Section 17).
D6299 Practice for Applying Statistical Quality Assurance
and Control Charting Techniques to Evaluate Analytical
1.4 Afundamental assumption in this test method is that the
Measurement System Performance
standard and sample matrices are well matched, or that the
D6300 Practice for Determination of Precision and Bias
matrix differences are accounted for (see 14.5). Matrix mis-
Data for Use in Test Methods for Petroleum Products,
match can be caused by C/H ratio differences between samples
Liquid Fuels, and Lubricants
and standards or by the presence of other interfering heteroa-
D7343 Practice for Optimization, Sample Handling,
toms; observe the cautions and recommendations in Section 6.
Calibration, and Validation of X-ray Fluorescence Spec-
trometry Methods for Elemental Analysis of Petroleum
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.03 on Elemental Analysis. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 1, 2022. Published December 2022. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2012. Last previous edition approved in 2017 as D7757 – 17. Standards volume information, refer to the standard’s Document Summary page on
DOI:10.1520/D7757-22. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7757 − 22
Products and Lubricants 5.4 Silicon contamination of gasoline, gasoline-ethanol
blends, denatured ethanol, and their blends has led to fouled
3. Terminology vehicle components (for example, spark plugs, exhaust oxygen
sensors, catalytic converters) requiring parts replacement and
3.1 Definitions:
repairs. Finished gasoline, gasoline-ethanol blends, and
3.1.1 For definitions of terms used in this test method, refer
ethanol-fuel blends can come into contact with silicon a
to Terminology D4175.
number of ways. Waste hydrocarbon solvents such as toluene
can be added to gasoline. Such solvents can contain soluble
4. Summary of Test Method
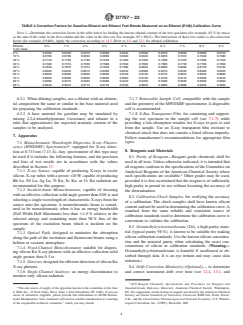
silicon compounds. Silicon-based antifoam agents can be used
4.1 A monochromatic X-ray beam with a wavelength suit-
in ethanol plants, which then pass silicon on to the finished
able to excite the K-shell electrons of silicon is focused onto a
ethanol-fuel blend. This test method can be used to determine
test specimen contained in a sample cell (see Fig. 1). The
if gasoline, gasoline-ethanol blends, and ethanol-fuel blends
fluorescent Kα radiation at 0.713 nm (7.13 Å) emitted by
meet specifications with respect to silicon content of the fuel,
silicon is collected by a fixed monochromator (analyzer). The
and for resolution of customer problems.
intensity (counts per second) of the silicon X-rays is measured
5.5 Some silicon compounds covered by this test method
using a suitable detector and converted to the concentration of
are significantly more volatile than the silicon compounds
silicon (mg/kg) in a test specimen using a calibration equation.
typically used for the preparation of the calibration standards.
Volatile compounds may not meet the stated precision from
5. Significance and Use
this test method because of selective loss of light materials
5.1 This test method provides rapid and precise measure-
during the analysis.
ment of total silicon in naphthas, gasoline, gasoline-ethanol
blends,RFG,ethanolandethanol-fuelblends,andtoluenewith
6. Interferences
minimum sample preparation.Typical analysis time is 5 min to
6.1 Differences between the elemental composition of test
10 min per sample.
samples and the calibration standards can result in biased
5.2 Excitation by monochromatic X-rays reduces
silicon determinations. For fuels within the scope of this test
background, simplifies matrix correction, and increases the
method, the only important elements contributing to bias
signal/background ratio compared to polychromatic excitation
resulting from differences in the matrices of calibrants and test
used in conventional WDXRF techniques.
samples are hydrogen, carbon, and oxygen. A matrix-
correction factor (C) may be used to correct this bias; the
5.3 Silicone oil defoamer can be added to coker feedstocks
calculation is described in Annex A1. For general analytical
to minimize foaming in the coker. Residual silicon in the coker
purposes, the matrices of test samples and the calibrants are
naphtha can adversely affect downstream catalytic processing
consideredtobematchedwhenthecalculatedcorrectionfactor
ofthenaphtha.Thistestmethodprovidesameanstodetermine
C is within 0.95 to 1.05. No matrix correction is required
the silicon content of the naphtha.
within this range. A matrix correction is required when the
valueofCisoutsidetherangeof0.95to1.05.Formosttesting,
matrix correction can be avoided with a proper choice of
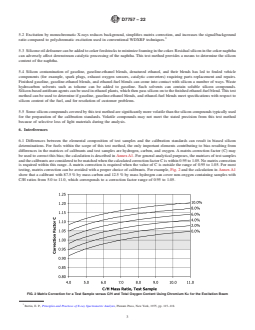
calibrants.Forexample,Fig.2andthecalculationinAnnexA1
Bertin, E. P., Principles and Practices of X-ray SpectrometricAnalysis, Plenum
Press, New York, 1975, pp. 115–118.
FIG. 1 Schematic of the MWDXRF Analyzer
D7757 − 22
FIG. 2 Matrix Correction for a Test Sample versus C/H and Total Oxygen Content Using Chromium Kα for the Excitation Beam
show that a calibrant with 87.5 % by mass carbon and 12.5 % 6.2.1 For test samples with high oxygenate content, such as
by mass hydrogen can cover non-oxygen containing samples
denatured fuel ethanol and ethanol fuel blends (see Specifica-
with C/H ratios from 5.0 to 11.0, which corresponds to a
tionsD4806andD5798),ethanol-basedcalibrantsmaybeused
correction factor range of 0.95 to 1.05.
provided the correction factors as described in 6.1 are applied
to the results. Table 1 and Table 2 show the correction factor
6.2 Fuels containing large quantities of ethanol, such as
that should be applied to the measurement results of the
ethanol fuel blends, denatured fuel ethanol, and gasoline-
gasoline-ethanol and ethanol fuel blends if they are measured
ethanol blends (see Specifications D4806 and D5798), can
have a high oxygen content leading to significant absorption of using either an isooctane or ethanol calibration curve.
silicon Kα radiation and low silicon results. Such fuels may be
NOTE 3—Alcohol based calibration standards may be preferred for test
analyzed using this test method provided either that correction
samples containing a high oxygenate content.
factors(seeTable1andTable2)areappliedtotheresultsorby
using calibration standards that are matrix matched to the test 6.3 To minimize any bias in the results, use calibration
standardspreparedfromsilicon-freebasematerialsofthesame
sample. For gasoline samples with oxygenates, up to 3.1 % by
mass oxygen can be tolerated for test samples with the same or similar elemental composition as the test samples.
C/H ratio as the calibrants.
TABLE 1 Correction Factors for Gasoline-Ethanol and Ethanol Fuel Blends Measured on an Isooctane Calibration Curve
NOTE 1—Determine the correction factor in the table below by finding the known ethanol content of the test specimen (for example, 15 % by mass)
as the sum of the value in the first column and the value in the first row (for example, 15 = 10+5). The intersection of these two values is the correction
factor (for example, 1.0844). Apply the correction according to 14.5. Refer to 8.7 and 12.1 for isooctane calibration.
Ethanol, 0% 1% 2% 3% 4% 5% 6% 7% 8% 9%
% by mass
0 % 1.0000 1.0056 1.0112 1.0169 1.0225 1.0281 1.0337 1.0394 1.0450 1.0506
10 % 1.0562 1.0619 1.0675 1.0731 1.0787 1.0844 1.0900 1.0956 1.1012 1.1069
20 % 1.1125 1.1181 1.1237 1.1294 1.1350 1.1406 1.1462 1.1519 1.1575 1.1631
30 % 1.1687 1.1744 1.1800 1.1856 1.1912 1.1969 1.2025 1.2081 1.2137 1.2194
40 % 1.2250 1.2306 1.2362 1.2419 1.2475 1.2531 1.2587 1.2644 1.2700 1.2756
50 % 1.2812 1.2868 1.2925 1.2981 1.3037 1.3093 1.3150 1.3206 1.3262 1.3318
60 % 1.3375 1.3431 1.3487 1.3543 1.3600 1.3656 1.3712 1.3768 1.3825 1.3881
70 % 1.3937 1.3993 1.4050 1.4106 1.4162 1.4218 1.4275 1.4331 1.4387 1.4443
80 % 1.4500 1.4556 1.4612 1.4668 1.4725 1.4781 1.4837 1.4893 1.4950 1.5006
90 % 1.5062 1.5118 1.5175 1.5231 1.5287 1.5343 1.5400 1.5456 1.5512 1.5568
D7757 − 22
TABLE 2 Correction Factors for Gasoline-Ethanol and Ethanol Fuel Blends Measured on an Ethanol (E100) Calibration Curve
NOTE 1—Determine the correction factor in the table below by finding the known ethanol content of the test specimen (for example, 85 % by mass)
as the sum of the value in the first column and the value in the first row (for example, 85 = 80+5). The intersection of these two values is the correction
factor (for example, 0.9460). Apply the correction according to 14.5. Refer to 8.6 and 12.1 for ethanol calibration.
Ethanol, 0% 1% 2% 3% 4% 5% 6% 7% 8% 9%
% by mass
0 % 0.6400 0.6436 0.6472 0.6508 0.6544 0.6580 0.6616 0.6652 0.6688 0.6724
10 % 0.6760 0.6796 0.6832 0.6868 0.6904 0.6940 0.6976 0.7012 0.7048 0.7084
20 % 0.7120 0.7156 0.7192 0.7228 0.7264 0.7300 0.7336 0.7372 0.7408 0.7444
30 % 0.7480 0.7516 0.7552 0.7588 0.7624 0.7660 0.7696 0.7732 0.7768 0.7804
40 % 0.7840 0.7876 0.7912 0.7948 0.7984 0.8020 0.8056 0.8092 0.8128 0.8164
50 % 0.8200 0.8236 0.8272 0.8308 0.8344 0.8380 0.8416 0.8452 0.8488 0.8524
60 % 0.8560 0.8596 0.8632 0.8668 0.8704 0.8740 0.8776 0.8812 0.8848 0.8884
70 % 0.8920 0.8956 0.8992 0.9028 0.9064 0.9100 0.9136 0.9172 0.9208 0.9244
80 % 0.9280 0.9316 0.9352 0.9388 0.9424 0.9460 0.9496 0.9532 0.9568 0.9604
90 % 0.9640 0.9676 0.9712 0.9748 0.9784 0.9820 0.9856 0.9892 0.9928 0.9964
6.3.1 When diluting samples, use a diluent with an elemen- 7.1.7 Removable Sample Cell, compatible with the sample
tal composition the same or similar to the base material used and the geometry of the MWDXRF spectrometer.Adisposable
for preparing the calibration standards. cell is recommended.
6.3.2 A base material for gasoline may be simulated by 7.1.8 X-Ray Transparent Film, for containing and support-
mixing 2,2,4-trimethylpentane (isooctane) and toluene in a ing the test specimen in the sample cell (see 7.1.7) while
ratio that approximates the expected aromatic content of the providing a low-absorption window for X-rays to pass to and
samples to be analyzed. from the sample. Use an X-ray transparent film resistant to
chemical attack that does not contain a listed silicon impurity.
7. Apparatus
Follow manufacturer’s recommendations for appropriate film
types.
7.1 Monochromatic Wavelength Dispersive X-ray Fluores-
cence (MWDXRF) Spectrometer , equipped for X-ray detec-
8. Reagents and Materials
tion at 0.713 nm (7.13 Å). Any spectrometer of this type may
be used if it includes the following features, and the precision 8.1 Purity of Reagents—Reagent grade chemicals shall be
and bias of test results are in accordance with the values
used in all tests. Unless otherwise indicated, it is intended that
described in Section 17.
all reagents conform to the specifications of the Committee on
7.1.1 X-ray Source, capable of producing X-rays to excite Analytical Reagents of the American Chemical Society where
silicon. X-ray tubes with a power >20 W capable of producing
such specifications are available. Other grades may be used,
Rh Lα,PdLα,AgLα,TiKα,ScKα,orCrKα radiation are provided it is first ascertained that the reagent is of sufficiently
recommended for this purpose.
high purity to permit its use without lessening the accuracy of
7.1.2 Incident-beam Monochromator, capable of focusing the determination.
with an effective collection solid angle greater than 0.05 sr and
8.2 Calibration-Check Samples, for verifying the accuracy
selecting a single wavelength of characteristic X-rays from the
of a calibration. The check samples shall have known silicon
source onto the specimen. A monochromatic beam is consid-
content and not be used in determining the calibration curve.A
ered to be monochromatic when it has an energy bandwidth
standard from the same reliable and consistent source of
(Full Width Half Maximum) less than 61.5 % relative to the
calibration standards used to determine the calibration curve is
selected energy and containing more than 98 % flux of the
convenient to validate the calibration.
spectrum of the excitation beam which is incident on the
8.3 Octamethylcyclotetrasiloxane (D4), a high-purity mate-
sample.
rial (typical purity 98 %), is known to be suitable for making
7.1.3 Optical Path, designed to minimize the absorption
silicon calibration standards. Use the known silicon concentra-
along the path of the excitation and fluorescent beams using a
tion and the material purity when calculating the exact con-
helium or vacuum atmosphere.
centrations of silicon in calibration standards. (Warning—
7.1.4 Fixed-Channel Monochromator, suitable for dispers-
Octamethylcyclotetrasiloxane is harmful if swallowed or ab-
ing silicon Kα X-ray photons with an effective collection solid
sorbed through skin. It is an eye irritant and may cause skin
angle greater than 0.3 sr.
irritation.)
7.1.5 Detector, designed for efficient detection of silicon Kα
X-ray photons.
8.4 Drift Correction Monitor(s) (Optional)—, to determine
7.1.6 Single-Channel Analyzer, an energy discriminator to
and correct instrument drift over time (see 12.4, 13.1, and
monitor only silicon radiation.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
The sole source of supply of the apparatus known to the committee at this time Standard-Grade Reference Materials, American Chemical Society, Washington,
is XOS, Inc., 15 Tech Valley Drive, Suite 1, East Greenbush, NY 12061. If you are DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
aware of alternative suppliers, please provide this information to ASTM Interna- Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
tional Headquarters.Your comments will receive careful consideration at a meeting U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
of the responsible technical committee, which you may attend. copeial Convention, Inc. (USPC), Rockville, MD.
D7757 − 22
14.1). Various forms of stable (with respect to repeated 9.2 Consult current health and safety regulations such as
exposure to X-rays) silicon-containing materials are suitable OSHA, suppliers’ Material Safety Data Sheets and local
drift correction monitors; for example, liquid petroleum, solid, regulations for all materials used in this test method.
pressed powder, metal alloy, and fused glass. The count rate
10. Sampling and Sample Handling
displayed by the monitor sample, in combination with a
convenient count time (T), shall be sufficient to give a relative
10.1 Sample fuel according to the procedures in Practices
standard deviation (RSD) of less than 1 % (see Appendix X1).
D4057 or D4177.
8.4.1 Calibration standards may be used as drift correction
10.2 Use the utmost care in sampling and handling gasoline
monitors. Because it is desirable to discard test specimens after
to prevent evaporation of light ends which could change the
each determination, a lower cost material is suggested for use
concentration of silicon in the sample. Store gasoline in a leak
over time. Any stable material meeting the recommendations
tight container at 0 °C to 4 °C until ready for analysis. If
of 8.4 or 8.4.1 may be used for monitoring of drift on a given
possible, maintain at this temperature throughout any transfer
day when samples are being analyzed.
and handling processes. Allow samples maintained at 0 °C to
NOTE 4—The effect of drift correction on the precision and bias of this 4 °C to come to room temperature before testing, and expose
test method has not been studied.
these materials to ambient conditions only as long as necessary
to obtain a sample for analysis.Analyze test specimens as soon
8.4.2 Drift correction may be done automatically if the
as possible after sub-sampling from bulk container. Do not
instrument embodies this option, although the calculation may
allow bulk container to remain uncovered any longer than is
be readily done by conventional methods of data reduction and
needed to obtain desired sub-samples.
processing.
10.3 For specimen preparation, see 11.2.
8.5 Quality-Control (QC) Samples, for use in establishing
10.3.1 Because impurities and thickness variations can oc-
and monitoring the stability and precision of an analytical
cur in commercially available transparent films and vary from
measurement system (see Section 16). Use homogeneous
lot to lot, use calibration-check samples (see 8.2) to verify
materials, similar to samples of interest and available in
calibration integrity after starting each new batch of film or if
sufficient quantity to be analyzed regularly for a long period of
the type and thickness of the window film is changed.
time.
8.5.1 Verification of system control through the use of QC 10.4 When reusable sample cells are used, thoroughly clean
and dry cells before each use.
samples and control charting is highly recommended.
10.4.1 Disposable sample cells shall not be reused.
NOTE 5—Suitable QC samples can be prepared by combining retains of
typical samples.
11. Preparation of Apparatus and Specimens for Analysis
8.6 Ethanol, use a high purity grade and account for its
11.1 Analyzer Preparation—Ensure that the MWDXRF
silicon content when calculating the silicon concentrations of
analyzer has been installed and put into operation according to
the calibration standards. (Warning—Ethanol is flammable
manufacturer’s instructions. Allow sufficient time for instru-
and harmful if swallowed or inhaled. It is an eye irritant and
ment electronics to stabilize. Perform any instrument checkout
may cause skin irritation.)
procedures required. When possible, the instrument should be
8.7 2,2,4-Trimethylpentane (Isooctane), use a high purity run continuously to maintain optimum stability.
grade and account for its silicon content when calculating the
11.1.1 Use the count time (T) recommended by the instru-
silicon concentration of the calibration standards. (Warning— ment manufacturer for the lowest silicon concentration ex-
Isooctane is flammable and harmful if swallowed or inhaled. It
pected. The typical time for each measurement is five to ten
is an eye irritant and may cause skin irritation.) minutes.
11.1.2 Alternatively, determine T expected for a desired
8.8 Toluene, use a high purity grade and account for its
count precision by following the procedure in Appendix X1.
silicon content when calculating the silicon concentration of
11.2 Specimen Preparation—Prepare a specimen of a test
the calibration standards. (Warning—Toluene is flammable
sample or a calibration standard as follows:
and harmful if swallowed or inhaled. It is an eye irritant and
11.2.1 Carefully transfer a sufficient portion of the liquid to
may cause skin irritation.)
fill a sample cell above a minimum depth beyond which
8.9 Helium,(forunitsthatrequireheliumpermanufacturer),
additional liquid does not affect the count rate. In a XRF
minimum purity 99.9 %, for use as an optical path.
sample cup, filling the sample cell to two thirds of the cell’s
depth is generally adequate.
9. Hazards
11.2.2 Fit an unused piece of
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7757 − 17 D7757 − 22
Standard Test Method for
Silicon in Gasoline and Related Products by Monochromatic
Wavelength Dispersive X-ray Fluorescence Spectrometry
This standard is issued under the fixed designation D7757; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of total silicon by monochromatic, wavelength-dispersive X-ray fluorescence
(MWDXRF) spectrometry in naphthas, gasoline, gasoline-ethanol blends, reformulated gasoline (RFG), ethanol and ethanol-fuel
blends, and toluene at concentrations of 3 mg ⁄kg to 100 mg ⁄kg. The precision of this test method was determined by an
interlaboratory study using representative samples of the liquids described in 1.1 and 1.2. The pooled limit of quantitation (PLOQ)
was estimated to be 3 mg ⁄kg.
NOTE 1—Volatile samples such as high-vapor-pressure gasolines or light hydrocarbons might not meet the stated precision because of the evaporation
of light components during the analysis.
NOTE 2—Aromatic compounds such as toluene are under the jurisdiction of Committee D16 on Aromatic Hydrocarbons and Related Chemicals. However,
toluene can be a contributor to silicon contamination in gasoline (see 4.45.4), thus its inclusion in this test method.
1.2 Gasoline samples containing ethanol and other oxygenates may be analyzed with this test method provided the matrix of the
calibration standards is either matched to the sample matrices or the matrix correction described in Annex A1 is applied to the
results. The conditions for matrix matching and matrix correction are provided Section 56, Interferences.
1.3 Samples with silicon concentrations above 100 mg ⁄kg can be analyzed after dilution with appropriate solvent. The precision
and bias of silicon determinations on diluted samples have not been determined and may not be the same as shown for neat samples
(Section 1617).
1.4 A fundamental assumption in this test method is that the standard and sample matrices are well matched, or that the matrix
differences are accounted for (see 13.514.5). Matrix mismatch can be caused by C/H ratio differences between samples and
standards or by the presence of other interfering heteroatoms; observe the cautions and recommendations in Section 56.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.03 on Elemental Analysis.
Current edition approved July 1, 2017Nov. 1, 2022. Published July 2017December 2022. Originally approved in 2012. Last previous edition approved in 20122017 as
D7757 – 12.D7757 – 17. DOI:10.1520/D7757-17.DOI:10.1520/D7757-22.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7757 − 22
2. Referenced Documents
2.1 ASTM Standards:
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4175 Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants
D4177 Practice for Automatic Sampling of Petroleum and Petroleum Products
D4806 Specification for Denatured Fuel Ethanol for Blending with Gasolines for Use as Automotive Spark-Ignition Engine Fuel
D5798 Specification for Ethanol Fuel Blends for Flexible-Fuel Automotive Spark-Ignition Engines
D6299 Practice for Applying Statistical Quality Assurance and Control Charting Techniques to Evaluate Analytical Measure-
ment System Performance
D6300 Practice for Determination of Precision and Bias Data for Use in Test Methods for Petroleum Products, Liquid Fuels, and
Lubricants
D7343 Practice for Optimization, Sample Handling, Calibration, and Validation of X-ray Fluorescence Spectrometry Methods
for Elemental Analysis of Petroleum Products and Lubricants
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this test method, refer to Terminology D4175.
4. Summary of Test Method
4.1 A monochromatic X-ray beam with a wavelength suitable to excite the K-shell electrons of silicon is focused onto a test
specimen contained in a sample cell (see Fig. 1). The fluorescent Kα radiation at 0.713 nm (7.13 Å) emitted by silicon is collected
by a fixed monochromator (analyzer). The intensity (counts per second) of the silicon X-rays is measured using a suitable detector
and converted to the concentration of silicon (mg/kg) in a test specimen using a calibration equation.
5. Significance and Use
5.1 This test method provides rapid and precise measurement of total silicon in naphthas, gasoline, gasoline-ethanol blends, RFG,
ethanol and ethanol-fuel blends, and toluene with minimum sample preparation. Typical analysis time is 5 min to 10 min per
sample.
FIG. 1 Schematic of the MWDXRF Analyzer
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
D7757 − 22
5.2 Excitation by monochromatic X-rays reduces background, simplifies matrix correction, and increases the signal/background
ratio compared to polychromatic excitation used in conventional WDXRF techniques.
5.3 Silicone oil defoamer can be added to coker feedstocks to minimize foaming in the coker. Residual silicon in the coker naphtha
can adversely affect downstream catalytic processing of the naphtha. This test method provides a means to determine the silicon
content of the naphtha.
5.4 Silicon contamination of gasoline, gasoline-ethanol blends, denatured ethanol, and their blends has led to fouled vehicle
components (for example, spark plugs, exhaust oxygen sensors, catalytic converters) requiring parts replacement and repairs.
Finished gasoline, gasoline-ethanol blends, and ethanol-fuel blends can come into contact with silicon a number of ways. Waste
hydrocarbon solvents such as toluene can be added to gasoline. Such solvents can contain soluble silicon compounds.
Silicon-based antifoam agents can be used in ethanol plants, which then pass silicon on to the finished ethanol-fuel blend. This test
method can be used to determine if gasoline, gasoline-ethanol blends, and ethanol-fuel blends meet specifications with respect to
silicon content of the fuel, and for resolution of customer problems.
5.5 Some silicon compounds covered by this test method are significantly more volatile than the silicon compounds typically used
for the preparation of the calibration standards. Volatile compounds may not meet the stated precision from this test method
because of selective loss of light materials during the analysis.
6. Interferences
6.1 Differences between the elemental composition of test samples and the calibration standards can result in biased silicon
determinations. For fuels within the scope of this test method, the only important elements contributing to bias resulting from
differences in the matrices of calibrants and test samples are hydrogen, carbon, and oxygen. A matrix-correction factor (C) may
be used to correct this bias; the calculation is described in Annex A1. For general analytical purposes, the matrices of test samples
and the calibrants are considered to be matched when the calculated correction factor C is within 0.95 to 1.05. No matrix correction
is required within this range. A matrix correction is required when the value of C is outside the range of 0.95 to 1.05. For most
testing, matrix correction can be avoided with a proper choice of calibrants. For example, Fig. 2 and the calculation in Annex A1
show that a calibrant with 87.5 % by mass carbon and 12.5 % by mass hydrogen can cover non-oxygen containing samples with
C/H ratios from 5.0 to 11.0, which corresponds to a correction factor range of 0.95 to 1.05.
FIG. 2 Matrix Correction for a Test Sample versus C/H and Total Oxygen Content Using Chromium Kα for the Excitation Beam
Bertin, E. P., Principles and Practices of X-ray Spectrometric Analysis, Plenum Press, New York, 1975, pp. 115–118.
D7757 − 22
6.2 Fuels containing large quantities of ethanol, such as ethanol fuel blends, denatured fuel ethanol, and gasoline-ethanol blends
(see Specifications D4806 and D5798), can have a high oxygen content leading to significant absorption of silicon Kα radiation
and low silicon results. Such fuels may be analyzed using this test method provided either that correction factors (see Table 1 and
Table 2) are applied to the results or by using calibration standards that are matrix matched to the test sample. For gasoline samples
with oxygenates, up to 3.1 % by mass oxygen can be tolerated for test samples with the same C/H ratio as the calibrants.
6.2.1 For test samples with high oxygenate content, such as denatured fuel ethanol and ethanol fuel blends (see Specifications
D4806 and D5798), ethanol-based calibrants may be used provided the correction factors as described in 5.16.1 are applied to the
results. Table 1 and Table 2 show the correction factor that should be applied to the measurement results of the gasoline-ethanol
and ethanol fuel blends if they are measured using either an isooctane or ethanol calibration curve.
NOTE 3—Alcohol based calibration standards may be preferred for test samples containing a high oxygenate content.
6.3 To minimize any bias in the results, use calibration standards prepared from silicon-free base materials of the same or similar
elemental composition as the test samples.
6.3.1 When diluting samples, use a diluent with an elemental composition the same or similar to the base material used for
preparing the calibration standards.
6.3.2 A base material for gasoline may be simulated by mixing 2,2,4-trimethylpentane (isooctane) and toluene in a ratio that
approximates the expected aromatic content of the samples to be analyzed.
7. Apparatus
7.1 Monochromatic Wavelength Dispersive X-ray Fluorescence (MWDXRF) Spectrometer , equipped for X-ray detection at
0.713 nm (7.13 Å). Any spectrometer of this type may be used if it includes the following features, and the precision and bias of
test results are in accordance with the values described in Section 1617.
7.1.1 X-ray Source, capable of producing X-rays to excite silicon. X-ray tubes with a power >20 W capable of producing Rh Lα,
Pd Lα, Ag Lα, Ti Kα, Sc Kα, or Cr Kα radiation are recommended for this purpose.
7.1.2 Incident-beam Monochromator, capable of focusing with an effective collection solid angle greater than 0.05 sr and selecting
a single wavelength of characteristic X-rays from the source onto the specimen. A monochromatic beam is considered to be
monochromatic when it has an energy bandwidth (Full Width Half Maximum) less than 61.5 % relative to the selected energy and
containing more than 98 % flux of the spectrum of the excitation beam which is incident on the sample.
TABLE 1 Correction Factors for Gasoline-Ethanol and Ethanol Fuel Blends Measured on an Isooctane Calibration Curve
NOTE 1—Determine the correction factor in the table below by finding the known ethanol content of the test specimen (for example, 15 % by mass)
as the sum of the value in the first column and the value in the first row (for example, 15 = 10+5). The intersection of these two values is the correction
factor (for example, 1.0844). Apply the correction according to 13.514.5. Refer to 7.78.7 and 11.112.1 for isooctane calibration.
Ethanol, 0 % 1 % 2 % 3 % 4 % 5 % 6 % 7 % 8 % 9 %
% by mass
0 % 1.0000 1.0056 1.0112 1.0169 1.0225 1.0281 1.0337 1.0394 1.0450 1.0506
10 % 1.0562 1.0619 1.0675 1.0731 1.0787 1.0844 1.0900 1.0956 1.1012 1.1069
20 % 1.1125 1.1181 1.1237 1.1294 1.1350 1.1406 1.1462 1.1519 1.1575 1.1631
30 % 1.1687 1.1744 1.1800 1.1856 1.1912 1.1969 1.2025 1.2081 1.2137 1.2194
40 % 1.2250 1.2306 1.2362 1.2419 1.2475 1.2531 1.2587 1.2644 1.2700 1.2756
50 % 1.2812 1.2868 1.2925 1.2981 1.3037 1.3093 1.3150 1.3206 1.3262 1.3318
60 % 1.3375 1.3431 1.3487 1.3543 1.3600 1.3656 1.3712 1.3768 1.3825 1.3881
70 % 1.3937 1.3993 1.4050 1.4106 1.4162 1.4218 1.4275 1.4331 1.4387 1.4443
80 % 1.4500 1.4556 1.4612 1.4668 1.4725 1.4781 1.4837 1.4893 1.4950 1.5006
90 % 1.5062 1.5118 1.5175 1.5231 1.5287 1.5343 1.5400 1.5456 1.5512 1.5568
The sole source of supply of the apparatus known to the committee at this time is XOS, Inc., 15 Tech Valley Drive, Suite 1, East Greenbush, NY 12061. If you are aware
of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible
technical committee, which you may attend.
D7757 − 22
TABLE 2 Correction Factors for Gasoline-Ethanol and Ethanol Fuel Blends Measured on an Ethanol (E100) Calibration Curve
NOTE 1—Determine the correction factor in the table below by finding the known ethanol content of the test specimen (for example, 85 % by mass)
as the sum of the value in the first column and the value in the first row (for example, 85 = 80+5). The intersection of these two values is the correction
factor (for example, 0.9460). Apply the correction according to 13.514.5. Refer to 7.68.6 and 11.112.1 for ethanol calibration.
Ethanol, 0 % 1 % 2 % 3 % 4 % 5 % 6 % 7 % 8 % 9 %
% by mass
0 % 0.6400 0.6436 0.6472 0.6508 0.6544 0.6580 0.6616 0.6652 0.6688 0.6724
10 % 0.6760 0.6796 0.6832 0.6868 0.6904 0.6940 0.6976 0.7012 0.7048 0.7084
20 % 0.7120 0.7156 0.7192 0.7228 0.7264 0.7300 0.7336 0.7372 0.7408 0.7444
30 % 0.7480 0.7516 0.7552 0.7588 0.7624 0.7660 0.7696 0.7732 0.7768 0.7804
40 % 0.7840 0.7876 0.7912 0.7948 0.7984 0.8020 0.8056 0.8092 0.8128 0.8164
50 % 0.8200 0.8236 0.8272 0.8308 0.8344 0.8380 0.8416 0.8452 0.8488 0.8524
60 % 0.8560 0.8596 0.8632 0.8668 0.8704 0.8740 0.8776 0.8812 0.8848 0.8884
70 % 0.8920 0.8956 0.8992 0.9028 0.9064 0.9100 0.9136 0.9172 0.9208 0.9244
80 % 0.9280 0.9316 0.9352 0.9388 0.9424 0.9460 0.9496 0.9532 0.9568 0.9604
90 % 0.9640 0.9676 0.9712 0.9748 0.9784 0.9820 0.9856 0.9892 0.9928 0.9964
7.1.3 Optical Path, designed to minimize the absorption along the path of the excitation and fluorescent beams using a helium or
vacuum atmosphere.
7.1.4 Fixed-Channel Monochromator, suitable for dispersing silicon Kα X-ray photons with an effective collection solid angle
greater than 0.3 sr.
7.1.5 Detector, designed for efficient detection of silicon Kα X-ray photons.
7.1.6 Single-Channel Analyzer, an energy discriminator to monitor only silicon radiation.
7.1.7 Removable Sample Cell, compatible with the sample and the geometry of the MWDXRF spectrometer. A disposable cell is
recommended.
7.1.8 X-Ray Transparent Film, for containing and supporting the test specimen in the sample cell (see 6.1.77.1.7) while providing
a low-absorption window for X-rays to pass to and from the sample. Use an X-ray transparent film resistant to chemical attack
that does not contain a listed silicon impurity. Follow manufacturer’s recommendations for appropriate film types.
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society where such
specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity
to permit its use without lessening the accuracy of the determination.
8.2 Calibration-Check Samples, for verifying the accuracy of a calibration. The check samples shall have known silicon content
and not be used in determining the calibration curve. A standard from the same reliable and consistent source of calibration
standards used to determine the calibration curve is convenient to validate the calibration.
8.3 Octamethylcyclotetrasiloxane (D4), a high-purity material (typical purity 98 %), is known to be suitable for making silicon
calibration standards. Use the known silicon concentration and the material purity when calculating the exact concentrations of
silicon in calibration standards. (Warning— Octamethylcyclotetrasiloxane is harmful if swallowed or absorbed through skin. It
is an eye irritant and may cause skin irritation.)
8.4 Drift Correction Monitor(s) (Optional)—, to determine and correct instrument drift over time (see 11.412.4, 12.113.1, and
13.114.1). Various forms of stable (with respect to repeated exposure to X-rays) silicon-containing materials are suitable drift
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For Suggestionssuggestions on the testing of reagents not listed by the American Chemical Society, see
AnnualAnalar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial
Convention, Inc. (USPC), Rockville, MD.
D7757 − 22
correction monitors; for example, liquid petroleum, solid, pressed powder, metal alloy, and fused glass. The count rate displayed
by the monitor sample, in combination with a convenient count time (T), shall be sufficient to give a relative standard deviation
(RSD) of less than 1 % (see Appendix X1).
8.4.1 Calibration standards may be used as drift correction monitors. Because it is desirable to discard test specimens after each
determination, a lower cost material is suggested for use over time. Any stable material meeting the recommendations of 7.48.4
or 7.4.18.4.1 may be used for monitoring of drift on a given day when samples are being analyzed.
D7757 − 22
NOTE 4—The effect of drift correction on the precision and bias of this test method has not been studied.
8.4.2 Drift correction may be done automatically if the instrument embodies this option, although the calculation may be readily
done by conventional methods of data reduction and processing.
8.5 Quality-Control (QC) Samples, for use in establishing and monitoring the stability and precision of an analytical measurement
system (see Section 1516). Use homogeneous materials, similar to samples of interest and available in sufficient quantity to be
analyzed regularly for a long period of time.
8.5.1 Verification of system control through the use of QC samples and control charting is highly recommended.
NOTE 5—Suitable QC samples can be prepared by combining retains of typical samples.
8.6 Ethanol, use a high purity grade and account for its silicon content when calculating the silicon concentrations of the
calibration standards. (Warning—Ethanol is flammable and harmful if swallowed or inhaled. It is an eye irritant and may cause
skin irritation.)
8.7 2,2,4-Trimethylpentane (Isooctane), use a high purity grade and account for its silicon content when calculating the silicon
concentration of the calibration standards. (Warning—Isooctane is flammable and harmful if swallowed or inhaled. It is an eye
irritant and may cause skin irritation.)
8.8 Toluene, use a high purity grade and account for its silicon content when calculating the silicon concentration of the calibration
standards. (Warning—Toluene is flammable and harmful if swallowed or inhaled. It is an eye irritant and may cause skin
irritation.)
8.9 Helium, (for units that require helium per manufacturer), minimum purity 99.9 %, for use as an optical path.
9. Hazards
9.1 (Warning—Exposure to excessive quantities of X-ray radiation is injurious to health. The operator needs to take appropriate
actions to avoid exposing any part of his/her body, not only to primary X-rays, but also to secondary or scattered radiation that
might be present. The X-ray spectrometer should be operated in accordance with local and national regulations governing the use
of ionizing radiation.)
9.2 Consult current health and safety regulations such as OSHA, suppliers’ Material Safety Data Sheets and local regulations for
all materials used in this test method.
10. Sampling and Sample Handling
10.1 Sample fuel according to the procedures in Practices D4057 or D4177.
10.2 Use the utmost care in sampling and handling gasoline to prevent evaporation of light ends which could change the
concentration of silicon in the sample. Store gasoline in a leak tight container at 0 °C to 4 °C until ready for analysis. If possible,
maintain at this temperature throughout any transfer and handling processes. Allow samples maintained at 0 °C to 4 °C to come
to room temperature before testing, and expose these materials to ambient conditions only as long as necessary to obtain a sample
for analysis. Analyze test specimens as soon as possible after sub-sampling from bulk container. Do not allow bulk container to
remain uncovered any longer than is needed to obtain desired sub-samples.
10.3 For specimen preparation, see 10.211.2.
10.3.1 Because impurities and thickness variations can occur in commercially available transparent films and vary from lot to lot,
use calibration-check samples (see 7.28.2) to verify calibration integrity after starting each new batch of film or if the type and
thickness of the window film is changed.
D7757 − 22
10.4 When reusable sample cells are used, thoroughly clean and dry cells before each use.
10.4.1 Disposable sample cells shall not be reused.
11. Preparation of Apparatus and Specimens for Analysis
11.1 Analyzer Preparation—Ensure that the MWDXRF analyzer has been installed and put into operation according to
manufacturer’s instructions. Allow sufficient time for instrument electronics to stabilize. Perform any instrument checkout
procedures required. When possible, the instrument should be run continuously to maintain optimum stability.
11.1.1 Use the count time (T) recommended by the instrument manufacturer for the lowest silicon concentration expected. The
typical time for each measurement is five to ten minutes.
11.1.2 Alternatively, determine T expected for a desired count precision by following the procedure in Appendix X1.
11.2 Specimen Preparation—Prepare a specimen of a test sample or a calibration standard as follows:
11.2.1 Carefully transfer a sufficient portion of the liquid to fill a sample cell above a minimum depth beyond which additional
liquid does not
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...