ASTM D6729-20
(Test Method)Standard Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100 Metre Capillary High Resolution Gas Chromatography
Standard Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100 Metre Capillary High Resolution Gas Chromatography
SIGNIFICANCE AND USE
5.1 Knowledge of the specified individual component composition (speciation) of gasoline fuels and blending stocks is useful for refinery quality control and product specification. Process control and product specification compliance for many individual hydrocarbons may be determined through the use of this test method.
SCOPE
1.1 This test method covers the determination of individual hydrocarbon components of spark-ignition engine fuels and their mixtures containing oxygenate blends (MTBE, ETBE, ethanol, and so forth) with boiling ranges up to 225 °C. Other light liquid hydrocarbon mixtures typically encountered in petroleum refining operations, such as blending stocks (naphthas, reformates, alkylates, and so forth) may also be analyzed; however, statistical data was obtained only with blended spark-ignition engine fuels.
1.2 Based on the cooperative study results, individual component concentrations and precision are determined in the range of 0.01 % mass to approximately 30 % mass. The procedure may be applicable to higher and lower concentrations for the individual components; however, the user must verify the accuracy if the procedure is used for components with concentrations outside the specified ranges.
1.3 The test method also determines methanol, ethanol, t-butanol, methyl t-butyl ether (MTBE), ethyl t-butyl ether (ETBE), t-amyl methyl ether (TAME) in spark ignition engine fuels in the concentration range of 1 % mass to 30 % mass. However, the cooperative study data provided sufficient statistical data for MTBE only.
1.4 Although a majority of the individual hydrocarbons present are determined, some co-elution of compounds is encountered. If this test method is utilized to estimate bulk hydrocarbon group-type composition (PONA) the user of such data should be cautioned that some error will be encountered due to co-elution and a lack of identification of all components present. Samples containing significant amounts of olefinic or naphthenic (for example, virgin naphthas), or both, constituents above n-octane may reflect significant errors in PONA type groupings. Based on the gasoline samples in the interlaboratory cooperative study, this procedure is applicable to samples containing less than 25 % mass of olefins. However, some interfering coelution with the olefins above C7 is possible, particularly if blending components or their higher boiling cuts such as those derived from fluid catalytic cracking (FCC) are analyzed, and the total olefin content may not be accurate. Caution should also be exercised when analyzing olefin-free samples using this test method as some of the paraffins may be reported as olefins since analysis is based purely on retention times of the eluting components.
1.4.1 Total olefins in the samples may be obtained or confirmed, or both, if necessary, by Test Method D1319 (percent volume) or other test methods, such as those based on multidimensional PONA type of instruments (Test Method D6839).
1.5 If water is or is suspected of being present, its concentration may be determined, if desired, by the use of Test Method D1744, or equivalent. Other compounds containing oxygen, sulfur, nitrogen, and so forth, may also be present, and may co-elute with the hydrocarbons. If determination of these specific compounds is required, it is recommended that test methods for these specific materials be used, such as Test Methods D4815 and D5599 for oxygenates, and D5623 for sulfur compounds, or equivalent.
1.6 Annex A1 of this test method compares results of the test procedure with other test methods for selected components, including olefins, and several group types for several interlaboratory cooperative study samples. Although benzene, toluene, and several oxygenates are determined, when doubtful as to the analytical results of these components, confirmatory analyses can be obtained by using specific test methods.
1.7 The values stated in SI units ar...
General Information
- Status
- Published
- Publication Date
- 31-May-2020
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.04.0L - Gas Chromatography Methods
Relations
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-May-2017
- Effective Date
- 20-Apr-2016
- Effective Date
- 01-Dec-2015
- Effective Date
- 01-Jun-2015
- Effective Date
- 01-Oct-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Oct-2010
- Effective Date
- 01-Oct-2009
- Effective Date
- 15-Apr-2009
- Effective Date
- 15-Oct-2008
- Effective Date
- 01-Nov-2007
Overview
ASTM D6729-20 defines the standard test method for the determination of individual hydrocarbon components in spark-ignition engine fuels, including oxygenate blends, using 100 metre capillary high resolution gas chromatography. This standard, issued by ASTM International, enables detailed analysis of gasoline and related refinery streams-such as naphthas and reformates-to support quality control, product specification, and regulatory compliance processes. The method is broadly recognized in the petroleum industry for its role in characterizing the detailed chemical composition of modern gasoline formulations.
Key Topics
Scope and Applicability
- Applies to spark-ignition engine fuels and their mixtures, including those containing oxygenates (MTBE, ETBE, ethanol, TAME, etc.) with boiling ranges up to 225 °C.
- Can be used for other refinery hydrocarbon mixtures (e.g., naphthas, reformates, alkylates), although method precision is validated for blended gasolines.
- Capable of quantifying individual hydrocarbons in the 0.01% to 30% mass range, with verification needed for concentrations outside this range.
- Suitable for determining certain oxygenates in the 1% to 30% mass concentration, with robust statistical data primarily for MTBE.
Gas Chromatography Approach
- Utilizes a 100 metre fused silica capillary column with high resolution, optimizing separation of individual components.
- Separation is based on retention times, and components are quantified using flame ionization detection (FID).
Reporting and Cautions
- Results are typically expressed as percent mass of each component.
- Some co-elutions occur, which may impact group-type (PONA: Paraffins, Olefins, Naphthenes, Aromatics) analysis, especially for samples with higher olefin or naphthene content.
- Interfering compounds (such as other oxygen, sulfur, or nitrogen species) should be analyzed with alternative ASTM test methods if present.
Applications
Refinery Quality Control
- Ensures manufactured gasoline meets necessary quality and compositional specifications.
- Facilitates monitoring and controlling refinery streams, blending operations, and finished product distribution.
Oxygenate and Hydrocarbon Speciation
- Allows for detailed hydrocarbon profiling in reformulated gasolines, especially important with increased use of oxygenate additives for emissions reduction.
- Supports compliance with regional and international fuel standards that specify maximum allowable concentrations of certain components (e.g., benzene, olefins, oxygenates).
Regulatory and Research Uses
- Used in verifying compositional requirements for fuels under emissions and air-quality regulations.
- Provides valuable data for comparative analysis and method validation within laboratory and research environments.
Process Optimization
- Guiding process adjustments for optimal blending, reducing production losses or off-spec batches.
Related Standards
ASTM D1319
Test Method for Hydrocarbon Types in Liquid Petroleum Products by Fluorescent Indicator Adsorption. Offers supplementary olefin content analysis where more precise measurement is required.ASTM D1744
Test Method for Water in Liquid Petroleum Products by Karl Fischer Reagent. Supports water determination if present in samples.ASTM D4815 and D5599
Methods for determination of various oxygenates in gasoline by gas chromatography, enabling identification of compounds that may co-elute in D6729 analysis.ASTM D5623
Test Method for Sulfur Compounds in Light Petroleum Liquids, addressing sulfur species that may interfere in the chromatographic analysis.ASTM D6839
Method for determining hydrocarbon types, oxygenated compounds, and benzene in spark-ignition fuels using multidimensional GC techniques.ASTM E355
Practice for Gas Chromatography Terms and Relationships, providing definitional support for method users.
Conclusion
ASTM D6729-20 is a cornerstone analytical standard in the petroleum industry, supporting detailed compositional analysis of gasoline and related fuels. Laboratories and refineries rely on this high-resolution capillary GC method for quality assurance, regulatory compliance, and optimal process control, ensuring fuel products are safe, efficient, and environmentally compliant. Understanding the scope, application limits, and related analytical standards is key to using this method effectively.
Keywords: capillary gas chromatography, ASTM D6729, gasoline speciation, refinery quality control, oxygenate analysis, individual hydrocarbon determination, petroleum standards, PONA analysis, FID detection.
Buy Documents
ASTM D6729-20 - Standard Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100 Metre Capillary High Resolution Gas Chromatography
REDLINE ASTM D6729-20 - Standard Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100 Metre Capillary High Resolution Gas Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D6729-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100 Metre Capillary High Resolution Gas Chromatography". This standard covers: SIGNIFICANCE AND USE 5.1 Knowledge of the specified individual component composition (speciation) of gasoline fuels and blending stocks is useful for refinery quality control and product specification. Process control and product specification compliance for many individual hydrocarbons may be determined through the use of this test method. SCOPE 1.1 This test method covers the determination of individual hydrocarbon components of spark-ignition engine fuels and their mixtures containing oxygenate blends (MTBE, ETBE, ethanol, and so forth) with boiling ranges up to 225 °C. Other light liquid hydrocarbon mixtures typically encountered in petroleum refining operations, such as blending stocks (naphthas, reformates, alkylates, and so forth) may also be analyzed; however, statistical data was obtained only with blended spark-ignition engine fuels. 1.2 Based on the cooperative study results, individual component concentrations and precision are determined in the range of 0.01 % mass to approximately 30 % mass. The procedure may be applicable to higher and lower concentrations for the individual components; however, the user must verify the accuracy if the procedure is used for components with concentrations outside the specified ranges. 1.3 The test method also determines methanol, ethanol, t-butanol, methyl t-butyl ether (MTBE), ethyl t-butyl ether (ETBE), t-amyl methyl ether (TAME) in spark ignition engine fuels in the concentration range of 1 % mass to 30 % mass. However, the cooperative study data provided sufficient statistical data for MTBE only. 1.4 Although a majority of the individual hydrocarbons present are determined, some co-elution of compounds is encountered. If this test method is utilized to estimate bulk hydrocarbon group-type composition (PONA) the user of such data should be cautioned that some error will be encountered due to co-elution and a lack of identification of all components present. Samples containing significant amounts of olefinic or naphthenic (for example, virgin naphthas), or both, constituents above n-octane may reflect significant errors in PONA type groupings. Based on the gasoline samples in the interlaboratory cooperative study, this procedure is applicable to samples containing less than 25 % mass of olefins. However, some interfering coelution with the olefins above C7 is possible, particularly if blending components or their higher boiling cuts such as those derived from fluid catalytic cracking (FCC) are analyzed, and the total olefin content may not be accurate. Caution should also be exercised when analyzing olefin-free samples using this test method as some of the paraffins may be reported as olefins since analysis is based purely on retention times of the eluting components. 1.4.1 Total olefins in the samples may be obtained or confirmed, or both, if necessary, by Test Method D1319 (percent volume) or other test methods, such as those based on multidimensional PONA type of instruments (Test Method D6839). 1.5 If water is or is suspected of being present, its concentration may be determined, if desired, by the use of Test Method D1744, or equivalent. Other compounds containing oxygen, sulfur, nitrogen, and so forth, may also be present, and may co-elute with the hydrocarbons. If determination of these specific compounds is required, it is recommended that test methods for these specific materials be used, such as Test Methods D4815 and D5599 for oxygenates, and D5623 for sulfur compounds, or equivalent. 1.6 Annex A1 of this test method compares results of the test procedure with other test methods for selected components, including olefins, and several group types for several interlaboratory cooperative study samples. Although benzene, toluene, and several oxygenates are determined, when doubtful as to the analytical results of these components, confirmatory analyses can be obtained by using specific test methods. 1.7 The values stated in SI units ar...
SIGNIFICANCE AND USE 5.1 Knowledge of the specified individual component composition (speciation) of gasoline fuels and blending stocks is useful for refinery quality control and product specification. Process control and product specification compliance for many individual hydrocarbons may be determined through the use of this test method. SCOPE 1.1 This test method covers the determination of individual hydrocarbon components of spark-ignition engine fuels and their mixtures containing oxygenate blends (MTBE, ETBE, ethanol, and so forth) with boiling ranges up to 225 °C. Other light liquid hydrocarbon mixtures typically encountered in petroleum refining operations, such as blending stocks (naphthas, reformates, alkylates, and so forth) may also be analyzed; however, statistical data was obtained only with blended spark-ignition engine fuels. 1.2 Based on the cooperative study results, individual component concentrations and precision are determined in the range of 0.01 % mass to approximately 30 % mass. The procedure may be applicable to higher and lower concentrations for the individual components; however, the user must verify the accuracy if the procedure is used for components with concentrations outside the specified ranges. 1.3 The test method also determines methanol, ethanol, t-butanol, methyl t-butyl ether (MTBE), ethyl t-butyl ether (ETBE), t-amyl methyl ether (TAME) in spark ignition engine fuels in the concentration range of 1 % mass to 30 % mass. However, the cooperative study data provided sufficient statistical data for MTBE only. 1.4 Although a majority of the individual hydrocarbons present are determined, some co-elution of compounds is encountered. If this test method is utilized to estimate bulk hydrocarbon group-type composition (PONA) the user of such data should be cautioned that some error will be encountered due to co-elution and a lack of identification of all components present. Samples containing significant amounts of olefinic or naphthenic (for example, virgin naphthas), or both, constituents above n-octane may reflect significant errors in PONA type groupings. Based on the gasoline samples in the interlaboratory cooperative study, this procedure is applicable to samples containing less than 25 % mass of olefins. However, some interfering coelution with the olefins above C7 is possible, particularly if blending components or their higher boiling cuts such as those derived from fluid catalytic cracking (FCC) are analyzed, and the total olefin content may not be accurate. Caution should also be exercised when analyzing olefin-free samples using this test method as some of the paraffins may be reported as olefins since analysis is based purely on retention times of the eluting components. 1.4.1 Total olefins in the samples may be obtained or confirmed, or both, if necessary, by Test Method D1319 (percent volume) or other test methods, such as those based on multidimensional PONA type of instruments (Test Method D6839). 1.5 If water is or is suspected of being present, its concentration may be determined, if desired, by the use of Test Method D1744, or equivalent. Other compounds containing oxygen, sulfur, nitrogen, and so forth, may also be present, and may co-elute with the hydrocarbons. If determination of these specific compounds is required, it is recommended that test methods for these specific materials be used, such as Test Methods D4815 and D5599 for oxygenates, and D5623 for sulfur compounds, or equivalent. 1.6 Annex A1 of this test method compares results of the test procedure with other test methods for selected components, including olefins, and several group types for several interlaboratory cooperative study samples. Although benzene, toluene, and several oxygenates are determined, when doubtful as to the analytical results of these components, confirmatory analyses can be obtained by using specific test methods. 1.7 The values stated in SI units ar...
ASTM D6729-20 is classified under the following ICS (International Classification for Standards) categories: 75.160.20 - Liquid fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6729-20 has the following relationships with other standards: It is inter standard links to ASTM D5623-24, ASTM D5599-17, ASTM D6839-16, ASTM D6839-15, ASTM D5599-15, ASTM D1319-14, ASTM D5623-94(2014), ASTM D1744-13, ASTM D6839-13, ASTM D1319-13, ASTM D5599-00(2010), ASTM D4815-09, ASTM D5623-94(2009), ASTM D1319-08, ASTM D6839-02(2007). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6729-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6729 − 20
Standard Test Method for
Determination of Individual Components in Spark Ignition
Engine Fuels by 100 Metre Capillary High Resolution Gas
Chromatography
This standard is issued under the fixed designation D6729; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* laboratory cooperative study, this procedure is applicable to
samples containing less than 25 % mass of olefins. However,
1.1 This test method covers the determination of individual
some interfering coelution with the olefins above C is
hydrocarbon components of spark-ignition engine fuels and
possible, particularly if blending components or their higher
their mixtures containing oxygenate blends (MTBE, ETBE,
boiling cuts such as those derived from fluid catalytic cracking
ethanol, and so forth) with boiling ranges up to 225 °C. Other
(FCC) are analyzed, and the total olefin content may not be
light liquid hydrocarbon mixtures typically encountered in
accurate. Caution should also be exercised when analyzing
petroleum refining operations, such as blending stocks
olefin-free samples using this test method as some of the
(naphthas, reformates, alkylates, and so forth) may also be
paraffins may be reported as olefins since analysis is based
analyzed; however, statistical data was obtained only with
purely on retention times of the eluting components.
blended spark-ignition engine fuels.
1.4.1 Total olefins in the samples may be obtained or
1.2 Based on the cooperative study results, individual com-
confirmed, or both, if necessary, by Test Method D1319
ponent concentrations and precision are determined in the
(percent volume) or other test methods, such as those based on
range of 0.01 % mass to approximately 30 % mass. The
multidimensional PONA type of instruments (Test Method
procedure may be applicable to higher and lower concentra-
D6839).
tions for the individual components; however, the user must
1.5 If water is or is suspected of being present, its concen-
verify the accuracy if the procedure is used for components
tration may be determined, if desired, by the use of Test
with concentrations outside the specified ranges.
Method D1744, or equivalent. Other compounds containing
1.3 The test method also determines methanol, ethanol,
oxygen, sulfur, nitrogen, and so forth, may also be present, and
t-butanol, methyl t-butyl ether (MTBE), ethyl t-butyl ether
may co-elute with the hydrocarbons. If determination of these
(ETBE), t-amyl methyl ether (TAME) in spark ignition engine
specific compounds is required, it is recommended that test
fuels in the concentration range of 1 % mass to 30 % mass.
methods for these specific materials be used, such as Test
However, the cooperative study data provided sufficient statis-
Methods D4815 and D5599 for oxygenates, and D5623 for
tical data for MTBE only.
sulfur compounds, or equivalent.
1.4 Although a majority of the individual hydrocarbons
1.6 Annex A1 of this test method compares results of the
present are determined, some co-elution of compounds is
test procedure with other test methods for selected
encountered. If this test method is utilized to estimate bulk
components, including olefins, and several group types for
hydrocarbon group-type composition (PONA) the user of such
several interlaboratory cooperative study samples. Although
data should be cautioned that some error will be encountered
benzene,toluene,andseveraloxygenatesaredetermined,when
due to co-elution and a lack of identification of all components
doubtful as to the analytical results of these components,
present. Samples containing significant amounts of olefinic or
confirmatory analyses can be obtained by using specific test
naphthenic (for example, virgin naphthas), or both, constitu-
methods.
ents above n-octane may reflect significant errors in PONA
1.7 The values stated in SI units are to be regarded as
type groupings. Based on the gasoline samples in the inter-
standard. No other units of measurement are included in this
standard.
1.8 This standard does not purport to address all of the
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
safety concerns, if any, associated with its use. It is the
Subcommittee D02.04.0L on Gas Chromatography Methods.
responsibility of the user of this standard to establish appro-
Current edition approved June 1, 2020. Published October 2020. Originally
priate safety, health, and environmental practices and deter-
approved in 2001. Last previous edition approved in 2014 as D6729 – 14. DOI:
10.1520/D6729-20. mine the applicability of regulatory limitations prior to use.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6729 − 20
1.9 This international standard was developed in accor- 5. Significance and Use
dance with internationally recognized principles on standard-
5.1 Knowledge of the specified individual component com-
ization established in the Decision on Principles for the
position (speciation) of gasoline fuels and blending stocks is
Development of International Standards, Guides and Recom-
useful for refinery quality control and product specification.
mendations issued by the World Trade Organization Technical
Process control and product specification compliance for many
Barriers to Trade (TBT) Committee.
individual hydrocarbons may be determined through the use of
this test method.
2. Referenced Documents
6. Apparatus
2.1 ASTM Standards:
D1319 Test Method for Hydrocarbon Types in Liquid Petro-
6.1 Gas Chromatograph, a gas chromatograph equipped
leum Products by Fluorescent Indicator Adsorption
with cryogenic column oven cooling and capable of producing
D1744 Test Method for Determination of Water in Liquid
repeatable oven ramps from 0 °C to at least 300 °C is required.
Petroleum Products by Karl Fischer Reagent (Withdrawn
The following features are useful during the sample analysis
2016)
phase: electronic flow readout, electronic sample split-ratio
D4815 Test Method for Determination of MTBE, ETBE,
readout,andelectronicpneumaticcontrolofflow.Thoughtheir
TAME, DIPE, tertiary-Amyl Alcohol and C to C Alco-
1 4 use is not required, careful review of this test method will
hols in Gasoline by Gas Chromatography
demonstrate the usefulness of a gas chromatograph equipped
D5599 Test Method for Determination of Oxygenates in
with these features. These features will replace the need to
Gasoline by Gas Chromatography and Oxygen Selective
carry out the manual calculations that must be performed as
Flame Ionization Detection
listed in 8.1 and 8.2.
D5623 Test Method for Sulfur Compounds in Light Petro-
6.2 Inlet—a capillary split/splitless inlet system operated in
leum Liquids by Gas Chromatography and Sulfur Selec-
thesplitmodeisrecommended.Itmustbeoperatedinitslinear
tive Detection
range. Refer to 8.4 to determine the proper split ratio.
D6839 Test Method for Hydrocarbon Types, Oxygenated
6.2.1 Carrier Gas Pneumatic Control—Constant carrier gas
Compounds, and Benzene in Spark Ignition Engine Fuels
pressurecontrolwasusedbyallcooperativestudyparticipants.
by Gas Chromatography
This may be either direct pressure to the inlet (injector) or by
E355 Practice for Gas ChromatographyTerms and Relation-
using a total flow/back pressure system.
ships
6.2.2 Pneumatic Operation of the Chromatograph—Theuse
of constant pressure was the mode of operating the gas
3. Terminology
chromatography used by the participants in the interlaboratory
3.1 Definitions—This test method makes reference to many
cooperative study. Other carrier gas control methods such as
common gas chromatographic procedures, terms, and relation-
constant flow (pressure programming) may be used, but this
ships. Detailed definitions can be found in Practice E355.
may change the chromatography elution pattern unless the
temperature programming profile is also adjusted to compen-
4. Summary of Test Method sate for the flow differences.
6.2.3 Temperature Control—The injector operated in the
4.1 Representative samples of the petroleum liquid are
split mode shall be heated by a separate heating zone and
introduced into a gas chromatograph equipped with an open
heated to temperatures of 200 °C to 275 °C.
tubular (capillary) column coated with the specified stationary
6.3 Column,afusedsilicacapillarycolumn,100minlength
phase. Helium carrier gas transports the vaporized sample
by 0.25 mm inside diameter, coated with a 0.5 µm film of
through the column, in which it is partitioned into individual
bonded dimethylpolysiloxane. The column must meet the
components which are sensed with a flame ionization detector
resolution requirements expressed in 8.3. Columns from two
as they elute from the end of the column. The detector signal
different commercial sources were used in the interlaboratory
is recorded digitally by way of an integrator or integrating
cooperative study.
computer. Each eluting component is identified by comparing
its retention time to that established by analyzing reference
6.4 Data System, a computer based chromatography data
standards or samples under identical conditions. The concen-
system capable of accurately and repeatedly measuring the
tration of each component in mass percent is determined by
retention time and areas of eluting peaks. The system shall be
normalization of the peak areas after correction of selected
abletoacquiredataatarateofatleast10Hz.Althoughitisnot
components with detector response factors. The unknown
mandatory, a data system which calculates column resolution
components are reported individually and as a summary total.
(R) is extremely useful as it will replace the need to carry out
the manual calculations which must be performed as listed in
8.3.
6.4.1 Electronic Integrators, shall be capable of storing up
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
to 400 components in the peak table and shall be able to
Standards volume information, refer to the standard’s Document Summary page on
acquire the data at 10 Hz or faster speeds. They shall be
the ASTM website.
capable of integrating peaks having peak widths at half height
The last approved version of this historical standard is referenced on
www.astm.org. which are 1.0s wide. The integrator must be capable of
D6729 − 20
displaying the integration mode of partially resolved peaks. In 8.1.3 The typical retention times for methane and linear gas
addition, these integrators should be able to download a velocity for helium are 6.5 cm⁄s to 6.8 cm⁄s and 24 cm⁄s to
commonly readable format of data (that is, ASCII) to a 26 cm⁄s, respectively.
computer in order to facilitate data processing.
8.2 Setting the Split Ratio—If the gas chromatograph is
6.5 Sample Introduction—Sample introduction by way of a
equipped with an electronic split-ratio readout device, set the
valve, automatic injection device, robotic arm or other auto-
split ratio to a sample split of 200:1. If the gas chromatograph
matic means is highly recommended. An automatic sample
is not equipped with an electronic split-ratio readout device,
introduction device is essential to the reproducibility of the
one must first calculate column flow rate and then proceed to
analysis. Manual injections are not recommended. All of the
calculating split ratio using Eq 2 and 3.
reproducibility data reported by this test method for the
60π r L T 2 P 2 P
~ ! ~ ! ~ !
ref i o
samples analyzed were gathered using automatic injection column flow rate 5 F 5 (2)
2 2
~T!3~P !~P 2 P !µ
ref i o
devices.
where:
6.6 Flame Ionization Detector (FID)—The gas chromato-
F = flow rate as calculated by using the equation,
graph should possess a FID having a sensitivity of 0.005 cou-
r = column radius, cm,
lombs⁄g for n-butane.The linear dynamic range of the detector
6 L = column length, cm,
should be 10 or better. The detector is heated to 300 °C.
P = inlet pressure,
i
P = outlet pressure,
7. Reagents and Materials o
P = reference pressure, 1 atm,
ref
7.1 Calibrating Standard Mixture—A spark ignition engine
T = temperature of the column oven,
fuelstandardofknowncompositionandconcentrationbymass
T = temperature at the column outlet, and
ref
can be used. In order to corroborate the identification of the
µ = linear velocity, cm/s.
sample, a typical chromatogram (Fig. 1) was obtained from
split vent flow1F
reference sample ARC96OX.
split ratio 5 S 5 (3)
F
7.2 Gas Chromatograph Gases—All of the following gases
8.2.1 The column flow rate is calculated by the use of Eq 2.
shall have a purity of 99.999 % (V⁄V) or greater.
Use the results obtained from Eq 3 to adjust the split flow until
NOTE1—Warning:Gasesarecompressed.Someareflammableandall
a split flow of approximately 200:1 is achieved.
gases are under high pressure.
8.3 Evaluation of Column Performance:
7.2.1 Helium—The test data was developed with helium as
8.3.1 Prior to using the column described in Table 1,
the carrier gas. It is possible that other carrier gases may be
measure the resolution of the column under the conditions of
usedforthistestmethod.Atthistime,nodataisavailablefrom
Table 2. Check that the resolution for the following pairs of
this test method with other carrier gases.
components is obtained using Eq 4 to calculate the resolution
7.2.2 Air, Hydrogen and Make-up Gas (Helium or
of a pair of components:
Nitrogen), shall have a purity of 99.999 % (V/V) or greater.
2 t 2 t
~ !
R2 R1
8. Instrument Check Out Prior to Analysis
R 5 (4)
1.699 ~W 1W !
h1 h2
8.1 Setting:
where:
8.1.1 Linear Gas Velocity—If the gas chromatograph is
R = resolution,
equippedwithanelectronicflowreadoutdevice,settheflowto
t = retention time of the first member of the pair,
1.8 mL/min. This is achieved by setting the carrier gas flow R2
t = retention time of the second member of the pair,
R1
rate by injection of cm/s methane or natural gas at 35 °C.
W = peak width at half height of the first member of the
h1
Ensure that the retention time is 7.00 min 6 0.05 min. This
pair, and
corresponds to a linear velocity of 25 cm/s to 26 cm/s. This is
W = peakwidthathalfheightofthesecondmemberofthe
h2
equivalent to retention times of methane at 0 °C ranging from
pair.
6.5 min to 6.8 min.
8.1.2 If the gas chromatograph is not equipped with an
8.3.1.1 Column resolution should be checked frequently by
electronic flow readout device, calculate the linear gas velocity examining the resolution of these compounds.
in cm/s using Eq 1.
8.3.2 Evaluation of the Baseline—Carry out a blank base-
line run utilizing no solvent injection, by setting the GC in
column length cm
~ !
linear gas velocity 5 V 5 (1)
accordance with the conditions of Table 1.
retention time of methane~s!
8.3.3 Subtract the baseline from a sample chromatogram
and verify that the residual signal at the beginning of the
Reference spark ignition sample No. ARC 960X obtained from the Alberta
chromatogram does not differ from the end of the chromato-
Research Council, Edmonton, Alberta, Canada. Other samples are available from
suppliers. gram by more than 2 %.
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
8.4 Evaluation of Splitter Linearity—Using the reference
gasoline sample, inject this sample according to the schedule
listed in Table 3.
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
8.4.1 Select from the chromatogram about 10 to 15 9. Procedure
components, which have concentrations in the range of 0.01 %
9.1 Settheoperatingconditionsofthegaschromatographas
mass to 30 % mass. Tabulate for each split ratio the concen-
shown in Table 1. These conditions will elute all components
trations of the 10 to 15 components. Verify that for each
up to and including pentadecane (nC ).
component selected, its concentration does not vary by more
than 3 %.
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
9.2 All of the parameters in Table 1 can be marginally column resolution (R) performance requirements listed in
changed to optimize for sample types and optimize for char- Table 2 should not be compromised.
acteristics of each gas chromatographic system. The final
boiling point of samples should not exceed nC and the
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
9.3 Obtain a representative sample following the guidelines 9.4 Preparation/Storage:
of Practice D4057 and any other applicable guidelines. Take 9.4.1 Samples Stored in Vials—Cool the original sample to
precautions to minimize the loss of light ends from volatile less than 4 °C prior to taking a sample aliquot or prior to filling
samples. The sample container may be cooled prior to transfer the sample vials. The sample aliquot container, or the vial, or
of sample into it. Cool the sample to less than 4 °C, maintain both, can also be cooled prior to the transfer of the original
atthatapproximatetemperatureuntiltheautosamplerisloaded sample. Syringes may also be cooled along with the sample for
and analysis begins. manual injections.
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
9.4.2 Samples Stored in Pressurized Containers—It is rec- 9.5 It is recommended that a quality assurance (QA) sample
ommended that they be kept away from direct heat or light. No similar to the reference material gasoline sample be run at
other sample preparations are necessary for samples stored in regular intervals (see Fig. 1). An interval of once per week or
pressurized containers. Avoid storage at temperatures greater after every 15 samples is recommended. The quantitation
than 25 °C. Store pressure containers in accordance with the results use statistical quality control charts can track benzene.
manufacturer’s instructions. Other components of interest in the reference sample can be
D6729 − 20
TABLE 1 Chromatographic Operating Conditions, Column Requirements and Data Acquisition Requirements
Chromatographic Conditions Requirements
Injector settings
Injector temperature, °C 250
Split ratio 175:1 - 275:1
Liner deactivated glass
Injection volume, µL 0.2–0.5
Detector settings
A
FID detector temperature, °C 300–350
Gas flows
B
Hydrogen, mL/min 30–40
Air, mL/min 300–450
Nitrogen make up, mL/min 30
Column oven settings
Initial temperature, °C 0
Initial time, min 15
1st ramp rate, °C/min 1
Final temperature, °C 50
Final time, min 0
2nd ramp rate, °C/min 2
Final temperature, °C 130
Final time, min 0
3rd ramp rate, °C/min 4
Final temperature, °C 270
C
Final time, min 0
Column Requirements
Length, m 100
Inside diameter, mm 0.25
Liquid phase 100 % dimethylpolysiloxane
Film thickness, µ 0.5
Pressure, psig 40–50
Flow, mL/min 1.7–2.0
Linear gas velocity, cm/s 24.5
Data acquisition, Hz 10–20
Total analysis time, min 140–150
A
Set to 25 °C to 50°C above the highest column temperature.
B
Values to be set as recommended by instrument manufacturer.
C
Final temperature or time may be adjusted to ensure complete elution of the sample components.
TABLE 2 Resolution Performance Requirements
be overloaded, and peaks can shift in retention time. Observe
Concentration of the peak pattern so that proper identification is made by
Component Minimum
Each
comparison with the reference material.
Pair Resolution
Component, W/W
10.2 Consistency in peak identification can be achieved by
Benzene 1.0 0.5 %–0.5 %
1-Methyl-cyclopentene
using software (data handling software, spreadsheet software,
m-Xylene 0.4 2.0 %–2.0 %
and so forth). Alternatively, a retention index system can be
p-Xylene
used.
n-Tridecane 1.0 0.5 %–0.5 %
1-Methylnaphthalene
log T 2 log T
~ ! ~ !
i n
~R1! 5 100n1100 (5)
F G
i
log~TN! 2 log~T !
n
TABLE 3 Injection Schedule
where:
Injection Volume, Injection Temperature,
(R1) = retention index of component I bracketed by the
Split Ratio
i
µL °C
N-paraffin, n in its lower boundary and N-paraffin N
100:1 0.1 250
in its upper boundary,
200:1 0.5 250
T = adjusted retention time of component i (retention
300:1 1.0 250
i
time of component i minus the retention time of
methane),
tracked in a similar manner. By monitoring these components T = adjusted retention time of N-paraffin n, and
n
TN = retention time of N-paraffin N.
over an extended period of time, the performance of the
column and the chromatographic system can be determined.
10.3 Determine the hydrocarbon response factors by using
the following equation.
10. Data Analysis
MW 1
i
10.1 Compound Identification—Prepare a table listing all of
RRF 5 3 (6)
CH4
N MW
c CH4
the retention times of the components in the sample. Compare
the retention time of each peak with that of the reference
gasoline. Pay particular attention to the fact that columns can Sevcik, J., Detectors in Gas Chromatography, Elsevier, NY, 1976, p. 94.
D6729 − 20
A
TABLE 4 Predominant Compounds and Identified Coeluting Compounds
NOTE 1—The response factor of the predominant compound will be used for the analyte and this analyte will be used for the calculations.
Peak Number Predominant Coeluting
(from Annex A1) Compound Compound(s)
164 3,3-dimethylpentane 5-methyl-1-hexene
186 2-methylhexane C -olefin
278 2,5-dimethylhexane C -olefin
286 3,3-dimethylhexane C -olefin
B
304 toluene 2,3,3-trimethylpentane
324 1,1,2-trimethylcyclopentane C -triolefin
326 C -diolefin C -paraffin
8 8
492 4-methyloctane C -olefin
796 1,2,3,4-tetramethylbenzene C -aromatic
A
This is not an exhaustive list. Due to the possibility of coeluting peaks in other areas, the user is cautioned in the interpretation of the data.
B
In most alkylated gasolines, a split may occur between toluene and 233 TMC5.
TABLE 5 Response Factors of Oxygenated Compounds
Relative Response Factors
Analytes
RRF C = 1.000 RRF CH = 1.000
7 4
Methanol 2.996 2.672
Ethanol 2.087 1.862
t-Butanol (TBA) 1.302 1.161
Methyl-t-butyl ether (MTBE) 1.577 1.407
Ethyl-t-butyl ether (ETBE) 1.407 1.255
t-Amyl methyl ether (TAME) 1.356 1.210
where: 11. Oxygenates
RRF = relative response factor of each component with
11.1 A cooperative study for linearity was performed for
CH4
respect to methane (RRF =1.000),
methanol, ethanol, t-butanol, methyl-t-butyl ether (MTBE),
CH4
MW = molecular weight of the component, i,
i ethyl-t-butylether(ETBE),andt-amylmethylether(TAME)in
N = number of carbon atoms in the molecule, and
c concentrationrangesfrom1.0 %massupto30 %mass(Annex
MW = molecular weight of methane (16.04276).
CH4
A2). The average relative response factors for the oxygenates
were calculated from the study and are listed in Table A2.1.
10.4 Convert the acquired areas to corrected areas by
They have been incorporated into the IHA Method. The
multiplying each area by its corresponding relative response
percent standard deviation of these relative response values
factor as indicated in the following equation.
was as high as 7 %. MTBE was the only oxygenate that was
A 5 A RRF (7)
~ ! ~ !
c I I
i
present in sufficient number of samples to meet the ASTM
where: requirements for round robin testing in accordance with
RR:D02-1007. Therefore the statistical data for MTBE should
(A ) = corrected area,
c i
be taken from Table A1.2.
A = acquired area for an individual component, and
i
RRF = relative response factor (weight basis).
12. Expression of Results
10.4.1 The percent mass (% W) is calculated as follows:
12.1 Report the concentration of each components as per-
~A ! cent (m/m) to the nearest 0.001 % mass.
C i
% W 5 3100 (8)
i i5n
12.2 The data for individual components may by grouped
A
( c
i
i51
bysummingtheconcentrationofcompoundsineachparticular
group type such as paraffin, isoparaffin, olefin, aromatic,
where:
naphthene, oxygenates, and unknowns. Commercially avail-
%W = percent mass of the component i in the mixture,
able software may be used to provide this function, as well as
and
the calculation of other properties of petroleum liquids.
i5n
= summation of all the corrected areas for the com-
A ponents analyzed.
( 13. Precision and Bias
c
i
i51
13.1 The repeatability and reproducibility precision esti-
mates are quoted in Annex A1.
10.4.1.1 The subscript i indicates that the operations are
carried out for each individual component in the matrix. Supporting data describing the interlaboratory cooperative study to determine
precision and bias has been filed at ASTM International Headquarters and may be
10.5 In the case of unidentified components, utilize a
obtained by requesting RR: RR:D02-1519. Contact ASTM Customer Service at
relative response factor of 0.800 (relative to methane). service@astm.org.
D6729 − 20
13.2 Precision Statement Outline—(>Analyte Qualification 13.3.4 r : upper 95 % confidence limit of r ,
max est
Process):
13.3.5 R ,R ,R : same as above except for
min est max
13.2.1 For each analyte to qualify for a precision statement,
reproducibility,
it must be present in at least six samples, and detected by at
13.3.6 C : lower concentration limit that rest, R is
min est
least six laboratories, at least once, in accordance with
applicable, and
RR:D02-1007 requirements.
13.3.7 C : upper concentration limit that rest, R is
max est
13.2.2 The(repeatabilitystandarddeviation)/meanvaluefor
applicable.
each analyte/sample combination must be less than or equal to
0.1, in accordance with LOQ requirements which, while not a
13.4 The summaries for the paraffins, isoparaffins, C
standard, is what CS94 is recommending.
benzene, and oxygenates follow the same procedure that was
used for the analytes and are listed in Table A1.3.
13.3 A brief explanation of headers in Table A1.2 follows:
13.3.1 ID: self explanatory,
13.5 Bias—The bias of this test method cannot be deter-
13.3.2 r : lower 95 % confidence limit of r ,
min est
mined since an appropriate standard reference material is not
13.3.3 r : repeatability estimate in percentage of
est
available.
concentration,
14. Keywords
Supporting data have been filed at ASTM International Headquarters and may
14.1 gas chromatograph; gasoline; individual hydrocarbon
beobtainedbyrequestingResearchReportRR:D02-1007.ContactASTMCustomer
Service at service@astm.org. analysis; oxygenated fuels; spark-ignition engine fuels
ANNEXES
(Mandatory Information)
A1. HYDROCARBON DATA
A1.1 TableA1.1presentsthecomponentretentiontimesand A1.4 The precision statement for the olefins and cyclopar-
properties. affins is determined by taking the square root of the value
determined in the summary; multiply by the coefficient (r )
coef
A1.2 Table A1.2 represents the repeatability and reproduc-
for repeatability and the coefficient (R ) for the reproduc-
coef
ibility precision estimates prepared by statisticians of CS94 in
ibility.
accordance with RR:D02-1519. The analyte qualification pro-
Name r r r R R R C C
min coef max min coef max min max
cess for precision statements is outlined as follows:
Cyclo- 0.0726 0.08 0.098 0.286 0.384 0.586 2 10
paraffins
A1.2.1 For each analyte to qualify for a precision statement,
Olefins 0.1555 0.18 0.21 0.382 0.555 1.012 2 25
it must be present in at least six samples, and detected by at
least six laboratories, at least once, in accordance with
A1.5 The precision for the aromatics does not depend on
RR:D02-1007 requirements.
level and is stated below in mass percent.
A1.2.2 The (repeatability standard deviation)/mean value Name r r% r R R% R C C
min max min max min max
Aromatics 0.8549 0.98 1.155 2.151 2.706 3.651 15 50
foreachanalyte/samplecombinationmustbelessthanorequal
to 0.1, in accordance with LOQ requirements which, while not
A1.6 The summaries for the paraffins, isoparaffins,
a standard, is what CS94 is recommending.
C benzene and oxygenates follow the same procedure that was
used for the analytes. The statistics for the grouping are shown
A1.3 Summary for Oxygenates: Warning—The statistical
in Table A1.3 as an indication of reproducibility and repeat-
data could be done on the oxygenates but there was not an
ability of reporting the results as a group summary. However,
equal number of all oxygenates in the round robin. MTBE was
there is a possibility that significant error could occur due to
the largest contributor to the statistical results. The number of
co-elution of peaks, the presence of significant amounts of
samples that contained each oxygenate is as follows:
olefinic or naphthenic constituents, or both, above octane and
Oxygenate No. of Approximate
Type Samples Concentration Range the percent unknown in the sample. If more accurate summary
Ethanol 2 1 %, and 12 %
results are needed that are not covered by the above precision
t-butanol 2 0.20 %, and 1.0 %
statement, for some or all of the above families of components,
MTBE 6 1, 2, 4, 4, 8 and 16 %
ETBE 1 0.50 %
please consider another ASTM test method.
TAME 1 15.00 %
D6729 − 20
TABLE A1.1 Component Retention Times and Properties
NOTE1—ThenamesusedarefromseveralothertablesandchangeshavebeenmadewheretheGCMSdidnotagreewiththepeaknameoritsretention
time.
NOTE 2—n-propanol will coelute with 3M-1-C5=.
NOTE 3—MTBE will coelute with 23DN-1C4=.
NOTE 4—MSBE will coelute with 1-hexene.
NOTE 5—ETBE will coelute with 23DM-13C4= =.
NOTE 6—isobutanol will coelute with 44DM-1-c5=.
NOTE 7—233TM pentane will coelute with toluene when the ratio with toluene is greater than 5.0:1.
NOTE 8—The coeluting olefins in Notes 2-6 will usually be below 1000 ppm.
NOTE 9—In some instances the chemical group is known, but the chemical structure is not known (for example, C -olefin; the position of the double
bond is not known).
NOTE 10—Relative response factors for six of the major oxygenated compounds have been determined by using the average results from seven
laboratories analyzing six samples in duplicate. These same samples were used to determine linearity of methanol, ethanol, t-butanol, MTBE, ETBE and
TAME from a concentration level ranging from 1 % mass up to 30 % mass.
Peak No. Compound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
1 Methane 6.74 16.04 1.000
2 Ethene 7.10 28.05 0.874
3 Ethane 7.21 30.07 0.937
4 Propene 7.41 42.05 0.874
5 Propane 7.87 44.11 0.916
6 Isobutane 8.26 58.12 0.906
7 Methanol 8.64 32.03 2.672
8 Isobutene 8.95 56.11 0.874
9 1-butene 8.99 56.11 0.874
10 1,3-butadiene 9.17 54.09 0.843
12 N-butane 9.28 58.12 0.906
14 Trans-2-butene 9.70 56.11 0.874
16 2,2-dimethylpropane 9.82 72.15 0.899
18 Cis-2-butene 10.33 56.11 0.874
20 1,2-butadiene 10.88 54.09 0.843
22 Ethanol 11.39 46.07 1.862
24 3-methyl-1-butene 12.21 70.13 0.874
26 Isopentane 13.57 72.15 0.899
28 1,4-pentadiene 14.25 68.12 0.849
30 2-Butyne (dimethylacetylene) 14.57 54.09 0.843
32 1-pentene 15.03 70.13 0.874
34 Isopropanol 15.28 60.11 1.950
36 2-methyl-1-butene 15.76 70.13 0.874
38 N-pentane 16.24 72.15 0.899
40 2-methyl-1,3-butadiene 16.73 68.12 0.849
42 Trans-2-pentene 17.23 70.13 0.874
44 3,3-dimethyl-1-butene 17.86 84.16 0.874
46 Cis-2-pentene 18.17 70.13 0.874
48 Tert-butanol (TBA) 18.51 74.12 1.161
50 2-methyl-2-butene 18.76 70.13 0.874
52 Trans-1,3-pentadiene 19.12 68.12 0.849
54 3-methyl-1,2-butadiene 19.48 68.12 0.849
56 Cyclopentadiene 19.76 66.10 0.824
58 Cis-1,3-pentadiene 20.25 68.12 0.849
60 1,2-pentadiene 20.51 68.12 0.849
62 2,2-dimethylbutane 20.69 86.18 0.895
64 Cyclopentene 23.16 68.12 0.849
66 4-methyl-1-pentene 24.30 84.16 0.874
68 3-methyl-1-pentene 24.38 84.16 0.874
70 n-propanol 24.68 60.11 1.770
72 Cyclopentane 24.86 70.13 0.874
74 2,3-dimethylbutane 25.57 86.18 0.895
76 2,3-dimethyl-1-butene 25.99 84.16 0.874
78 Methyl tert-butyl ether (MTBE) 26.18 88.09 1.407
80 Cis-4-methyl-2-pentene 26.48 84.16 0.874
82 2-methylpentane 26.66 86.18 0.895
84 Trans-4-methyl-2-pentene 27.09 84.16 0.874
86 Methyl ethyl ketone (MEK) 28.00 72.06 1.570
88 3-methylpentane 29.15 86.18 0.895
90 C -olefin 29.61 84.16 0.874
92 2-methyl-1-pentene 30.29 84.16 0.874
94 1-hexene 30.52 84.16 0.874
96 Methyl sec-butyl ether (MSBE) 30.66 88.09 1.550
D6729 − 20
TABLE A1.1 Continued
Peak No. Compound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
98 C -olefin 30.94 84.16 0.874
100 2-butanol 31.56 74.12 1.600
102 2ethyl-1-butene 32.47 84.16 0.874
104 N-hexane 32.75 86.18 0.895
106 Cis-3-hexene 33.41 84.16 0.874
108 Di-isopropyl ether (DIPE) 33.58 102.00 1.600
110 Trans-3-hexene+hexadiene 33.86 84.16 0.874
112 2-methyl-2-pentene 34.33 84.16 0.874
114 3-methylcyclopentene 34.57 82.10 0.853
116 Trans-3-methyl-2-pentene 34.71 84.16 0.874
118 Cis-2-hexene 35.62 84.16 0.874
120 3,3-dimethyl-1-pentene 36.04 98.19 0.874
122 Cis-3-methyl-2-pentene 36.92 84.16 0.874
124 Ethyl tert-butyl ether (ETBE) 37.07 102.18 1.255
126 2,3-dimethyl-1,3-butadiene 37.19 82.10 0.853
128 Methylcyclopentane 37.40 84.16 0.874
130 2,2-dimethylpentane 37.60 100.21 0.892
132 4,4-dimethyl-1-pentene 37.91 98.19 0.874
134 Isobutanol 38.06 74.12 1.500
136 2,3-dimethyl-2-butene 38.30 84.16 0.874
138 2,4-dimethylpentane 38.99 100.21 0.892
140 1,3,5-hexatriene 39.31 80.06 0.832
142 2,2,3-trimethylbutane 39.48 100.21 0.892
144 Methylcyclopentadiene 40.17 80.06 0.832
146 C -olefin 40.30 98.19 0.874
148 C -olefin 40.68 98.19 0.874
150 C -diolefin 41.20 96.18 0.856
152 4-methylcyclopentene 41.44 82.10 0.853
154 Methylenecyclopentane 42.08 82.10 0.853
156 Benzene 42.30 78.05 0.812
158 1-methyl-1-cyclopentene 42.46 82.10 0.853
160 C -olefin 43.06 98.19 0.874
162 Cis-2-methyl-3-hexene 43.37 98.19 0.874
164 3,3-dimethylpentane+5-methyl- 43.81 100.21 0.892
1-hexene
166 Cyclohexane 44.07 84.16 0.874
168 Trans-2-methyl-3-hexene 44.82 98.19 0.874
170 3,3-dimethyl-1,4-pentadiene 45.44 96.18 0.856
172 N-butanol 45.58 74.12 1.500
174 Dimethylcyclopentadiene 45.69 94.17 0.838
176 t,2-ethyl-3-methyl-1-butene 45.97 98.19 0.874
178 4-methyl-1-hexene 46.27 98.19 0.874
180 C -olefin 46.55 98.19 0.874
182 3-methyl-1-hexene 46.78 98.19 0.874
184 4-methyl-2-hexene 46.92 98.19 0.874
186 2-methylhexane+C -olefin 47.29 100.21 0.892
188 2,3-dimethylpentane 47.51 100.21 0.892
190 Cyclohexene 47.65 82.10 0.853
192 Tert-amyl methyl ether (TAME) 48.10 102.18 1.210
194 C -olefin 48.46 98.19 0.874
196 C -olefin 48.64 98.19 0.874
198 3-methylhexane 49.05 100.21 0.892
200 C -olefin 49.47 98.19 0.874
202 C -olefin 49.62 98.19 0.874
204 Trans-1,3-dimethylcyclopentane 49.83 98.19 0.874
206 Cis-1,3-dimethylcyclopentane 50.40 98.19 0.874
208 Trans-1,2-dimethylcyclopentane 51.01 98.19 0.874
210 3-ethylpentane 51.21 100.10 0.892
212 C -olefin 51.43 98.19 0.874
214 2,2,4-trimethylpentane 51.61 114.23 0.890
216 C -olefin 51.75 98.19 0.874
218 1-heptene 52.05 98.19 0.874
220 C -olefin 52.18 98.19 0.874
222 2,3-dimethyl-1,3-pentadiene 52.69 96.18 0.856
224 C -diolefin 53.00 96.18 0.856
226 C -olefin 53.36 98.19 0.874
228 C -diolefin 53.81 96.18 0.856
230 C -diolefin 54.13 96.18 0.856
232 C -olefin 54.28 98.19 0.874
234 N-heptane 54.59 100.21 0.892
236 Cis-3-heptene 54.81 98.19 0.874
238 2-methyl-2-hexene 55.10 98.19 0.874
240 Cis-3-methyl-3-hexene 55.35 98.19 0.874
242 Trans-3-heptene 55.72 98.19 0.874
244 3-ethyl-2-pentene 55.88 96.19 0.874
D6729 − 20
TABLE A1.1 Continued
Peak No. Compound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
246 1,5-dimethylcyclopentene 56.06 96.18 0.856
248 Cis-3-methyl-2-hexene 56.58 98.19 0.874
250 C -diolefin+C -triolefin 57.01 96.18 0.856
7 7
252 2,3-dimethyl-2-pentene 57.35 98.19 0.874
254 3-ethylpentene 57.57 98.19 0.874
256 Methylcyclohexane 57.79 98.19 0.874
258 C -olefin 58.28 98.19 0.874
260 1,1,3-trimethylcyclopentane 58.79 112.22 0.874
262 2,2-dimethylhexane 59.29 114.10 0.890
264 2,3,4-trimethyl-1,4-pentadiene 59.45 110.21 0.859
266 3,3-dimethyl-1,5-hexadiene 59.79 110.21 0.859
268 C -olefin 60.12 112.22 0.874
270 Ethylcyclopentane 60.60 98.19 0.874
272 3-methylcyclohexene 60.99 96.18 0.856
274 Methylcyclohexadiene 61.14 94.17 0.838
276 2,2,3-trimethylpentane 61.22 114.10 0.890
278 2,5-dimethylhexane+C -olefin 61.59 114.23 0.890
280 2,4-dimethylhexane 61.91 114.23 0.890
282 C -triolefin+C -olefin 62.28 112.24 0.856
7 8
284 Trans,cis-1,2,4- 62.68 112.22 0.874
trimethylcyclopentane
286 3,3-dimethylhexane+C -olefin 63.13 114.23 0.890
288 C -triolefin+C -olefin 63.39 112.24 0.856
7 8
290 C -olefin 63.69 112.22 0.874
292 Trans,cis-1,2,3- 64.27 112.22 0.874
trimethylcyclopentane
294 C -olefin 64.52 112.22 0.874
296 C -olefin 64.73 112.22 0.874
298 C -olefin 64.82 112.22 0.874
300 2,3,4-trimethylpentane 64.94 114.23 0.890
302 C -diolefin 65.25 96.18 0.856
304 Toluene 65.50 92.06 0.821
306 2,3,3-trimethylpentane 65.76 114.23 0.890
308 C -olefin 65.90 112.22 0.874
310 C -diolefin 66.12 110.21 0.859
312 C -olefin 66.48 112.22 0.874
314 C -olefin 66.65 112.22 0.874
316 C -olefin 67.08 112.22 0.874
318 C -olefin 67.30 112.22 0.874
320 2,3-dimethylhexane 67.47 114.23 0.890
322 2-methyl-3-ethylpentane 67.71 114.23 0.890
324 1,1,2-trimethylcyclopentane+C - 68.04 112.22 0.874
triolefin
326 C -diolefin+C -paraffin 68.31 114.23 0.859
8 8
328 C -olefin 68.41 112.22 0.874
330 C -olefin 68.64 112.22 0.874
332 2-methylheptane 68.86 114.23 0.890
334 4-methylheptane 69.11 114.23 0.890
336 C -diolefin+C -olefin 69.41 112.22 0.874
8 7
338 C -olefin 69.70 112.22 0.874
340 Cis-1,3-dimethylcyclohexane 69.91 112.22 0.874
342 Trans-1,4-dimethylcyclohexane 70.01 112.22 0.874
344 3-methylheptane 70.23 114.23 0.890
346 3-ethylhexane 70.38 114.23 0.890
348 C -diolefin 70.51 110.21 0.874
350 C -olefin 70.72 112.22 0.874
352 C -olefin 70.92 112.22 0.874
354 1,1-dimethylcyclohexane 71.18 112.22 0.874
356 C -olefin 71.43 112.22 0.874
358 C -olefin 71.70 112.22 0.874
360 Cis-1-ethyl-3- 72.10 112.22 0.874
methylcyclopentane
362 2,2,5-trimethylhexane 72.23 128.26 0.888
364 Trans-1-ethyl-3- 72.46 112.22 0.874
methylcyclopentane
366 Trans-1-ethyl-2- 72.68 112.22 0.874
methylcyclopentane
368 1-methyl-1-ethylcyclopentane 72.96 112.22 0.874
370 1-octene 73.16 112.22 0.874
372 C -olefin 73.26 112.22 0.874
374 Trans-1,2-dimethylcyclohexane 73.36 112.22 0.874
376 C -olefin 73.48 112.22 0.874
378 C -olefin 73.68 112.22 0.874
380 C -olefin 74.08 112.11 0.874
382 C -olefins 74.45 112.22 0.874
D6729 − 20
TABLE A1.1 Continued
Peak No. Compound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
384 Trans-1,3-dmcyclohexane 74.66 112.22 0.874
386 Cis-1,4-dmcycloHexane 74.79 112.22 0.874
388 N-octane 74.98 114.23 0.890
390 C -olefin 75.33 112.22 0.874
392 C -olefin 75.49 112.22 0.874
394 Trans-2-octene 75.62 112.22 0.874
396 Isopropylcyclopentane 75.72 112.22 0.874
398 C -olefin 75.85 126.24 0.874
400 C -olefin 75.89 126.24 0.874
402 C -olefin 75.90 126.24 0.874
404 C -olefin 76.08 126.24 0.874
406 2,2,4-trimethylhexane 76.31 128.26 0.888
408 2,4,4-trimethylhexane 76.62 128.26 0.888
410 C -olefins 76.86 126.24 0.874
412 2,3,5-trimethylhexane 77.29 128.26 0.888
414 Cis-2-octene 77.53 112.22 0.874
416 2,2,3,4-tetramethylpentane 77.77 128.26 0.888
418 2,2-dimethylheptane 78.02 128.26 0.888
420 Cis-1,2-dimethylcyclohexane 78.36 112.22 0.874
422 2,4-dimethylheptane 78.74 128.26 0.888
424 C -olefin 78.90 126.24 0.874
426 C -olefin 79.08 126.24 0.874
428 Ethylcyclohexane 79.24 112.22 0.874
430 Propylcyclopentane 79.39 112.22 0.874
432 2-methyl-4-ethylhexane 79.59 128.26 0.888
434 2,6-dimethylheptane 79.74 128.26 0.888
436 C -olefin 79.85 126.24 0.874
438 1,1,4-trimethylcyclohexane 80.05 126.24 0.874
440 C -olefin 80.28 126.24 0.874
442 C -olefin 80.38 126.24 0.874
444 1,1,3-trimethylcyclohexane 80.52 126.24 0.874
446 2,5 & 3,5-dimethylheptane 80.69 128.26 0.888
448 C -olefin 80.88 126.24 0.874
450 3,3-DMheptane 81.00 128.26 0.888
452 C -paraffin 81.13 128.26 0.888
454 C -olefin 81.34 126.24 0.874
456 2,3,3-trimethylhexane 81.56 128.26 0.888
458 C -olefin 81.68 126.24 0.874
460 Ethylbenzene 81.96 106.08 0.827
462 C -olefin 82.00 126.24 0.874
464 1c,2t,4t-trimethylcyclohexane 82.31 126.24 0.874
466 C -olefin 82.33 126.24 0.874
468 2,3,4-trimethylhexane 82.63 128.26 0.888
470 C -olefin 82.73 126.24 0.874
472 3,3,4-trimethylhexane 82.89 128.26 0.888
474 M-Xylene 83.30 106.08 0.827
476 P-Xylene 83.43 106.08 0.827
478 2,3-dimethyheptane 83.57 128.26 0.888
480 3,5-dimethylheptane 83.83 128.26 0.888
482 3,4-dimethylheptane 83.91 128.26 0.888
484 C -olefin 84.08 126.24 0.874
486 3-methyl-3-ethylhexane 84.26 128.26 0.888
488 C -olefin 84.41 126.24 0.874
490 4-ethylheptane 84.52 128.26 0.888
492 4-methyloctane+C -olefin 84.70 128.26 0.888
494 2-methyloctane 84.84 128.26 0.888
496 C -olefin 85.01 126.24 0.874
498 C -paraffin 85.18 128.26 0.888
500 C -olefin 85.36 126.24 0.874
502 3-ethylheptane 85.51 128.26 0.888
504 3-methyloctane 85.69 128.26 0.888
506 C -paraffin 85.87 128.23 0.888
508 C -cyclohexane 85.91 126.24 0.874
510 1,1,2-trimethylcyclohexane 86.05 126.24 0.874
512 O-Xylene 86.27 106.08 0.827
514 C -olefin 86.47 126.24 0.874
516 C -paraffin 86.57 128.26 0.888
518 C -paraffin 86.75 128.26 0.888
520 C -olefin 86.90 126.24 0.874
522 Trans-1-ethyl-4- 87.08 126.24 0.874
methylcyclohexane
524 Cis-1-ethyl-4- 87.23 126.24 0.874
methylcyclohexane
526 C -paraffin 87.49 128.26 0.888
528 1-nonene 87.79 126.24 0.874
D6729 − 20
TABLE A1.1 Continued
Peak No. Compound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
530 Isobutylcyclopentane 88.00 126.24 0.874
532 C -paraffin 88.45 128.26 0.888
534 Trans-3-nonene 88.65 126.24 0.874
536 Cis-3-nonene 88.82 126.24 0.874
538 C -paraffin 89.09 128.26 0.888
540 n-nonane 89.24 128.26 0.888
542 C -olefin 89.41 140.27 0.874
544 Trans-2-nonene 89.47 126.24 0.874
546 1-methyl-1-ethylcyclohexane 89.61 126.24 0.874
548 1-methyl-2-propylcyclopentane 89.96 126.24 0.874
550 C -olefin 90.09 140.27 0.874
552 C -paraffin 90.18 142.28 0.887
554 C -paraffin 90.29 142.28 0.887
556 Isopropylbenzene 90.46 120.20 0.832
558 Cis-2-nonene 90.78 126.24 0.874
560 Tert-butylcyclopentane 90.80 126.24 0.874
562 C -olefins 90.88 126.24 0.874
564 C -Olefin 91.16 126.24 0.874
566 Isopropylcyclohexane 91.32 126.24 0.874
568 3,3,5-trimethylheptane 91.44 142.28 0.887
570 2,2-dimethyloctane 91.60 142.28 0.887
572 2,4-dimethyloctane 91.67 142.28 0.887
574 1-methyl-4- 91.82 140.27 0.874
isopropylcyclohexane
576 Sec-butylcyclopentane 92.20 126.24 0.874
578 Propylcyclohexane 92.40 126.24 0.874
580 2,5-dimethyloctane 92.59 142.28 0.887
582 Butylcyclopentane 92.89 126.24 0.874
584 2,6-dimethyloctane 93.04 142.28 0.887
586 3,6-dimethyloctane 93.43 142.28 0.887
588 1-methyl-2-ethylcyclohexane 93.59 126.24 0.874
590 C -olefin 93.79 140.27 0.874
592 Propylbenzene 93.96 120.20 0.832
594 3,3-dimethyloctane 94.27 142.28 0.887
596 3-methyl-5-ethylheptane 94.54 142.28 0.887
598 C -olefin 94.66 140.27 0.874
600 1-ethyl-3-methylbenzene 94.88 120.20 0.832
602 1-ethyl-4-methylbenzene 95.09 120.20 0.832
604 Naphthene 95.30 140.27 0.874
606 1,3,5-trimethylbenzene 95.73 120.20 0.832
608 2,3-dimethyloctane 95.94 142.28 0.887
610 5-methylnonane 96.13 142.28 0.887
612 4-methylnonane 96.29 142.28 0.887
614 2-methylnonane 96.49 142.28 0.887
616 1-ethyl-2-methylbenzene 96.77 120.20 0.832
618 3-ethyloctane 97.01 142.28 0.887
620 Naphthene 97.14 140.27 0.874
622 3-methylnonane 97.47 142.28 0.887
624 C -olefin 97.69 140.27 0.874
626 C -paraffin 97.83 142.28 0.887
628 C -paraffin 98.16 142.28 0.887
630 1,2,4-trimethylbenzene 98.49 120.20 0.832
632 C -paraffin 98.74
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6729 − 14 D6729 − 20
Standard Test Method for
Determination of Individual Components in Spark Ignition
Engine Fuels by 100 Metre Capillary High Resolution Gas
Chromatography
This standard is issued under the fixed designation D6729; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of individual hydrocarbon components of spark-ignition engine fuels and their
mixtures containing oxygenate blends (MTBE, ETBE, ethanol, and so forth) with boiling ranges up to 225 °C. Other light liquid
hydrocarbon mixtures typically encountered in petroleum refining operations, such as blending stocks (naphthas, reformates,
alkylates, and so forth) may also be analyzed; however, statistical data was obtained only with blended spark-ignition engine fuels.
1.2 Based on the cooperative study results, individual component concentrations and precision are determined in the range of
0.01 % mass to approximately 30 % mass. The procedure may be applicable to higher and lower concentrations for the individual
components; however, the user must verify the accuracy if the procedure is used for components with concentrations outside the
specified ranges.
1.3 The test method also determines methanol, ethanol, t-butanol, methyl t-butyl ether (MTBE), ethyl t-butyl ether (ETBE), t-amyl
methyl ether (TAME) in spark ignition engine fuels in the concentration range of 1 % mass to 30 % mass. However, the
cooperative study data provided sufficient statistical data for MTBE only.
1.4 Although a majority of the individual hydrocarbons present are determined, some co-elution of compounds is encountered. If
this test method is utilized to estimate bulk hydrocarbon group-type composition (PONA) the user of such data should be cautioned
that some error will be encountered due to co-elution and a lack of identification of all components present. Samples containing
significant amounts of olefinic or naphthenic (for example, virgin naphthas), or both, constituents above n-octane may reflect
significant errors in PONA type groupings. Based on the gasoline samples in the interlaboratory cooperative study, this procedure
is applicable to samples containing less than 25 % mass of olefins. However, some interfering coelution with the olefins above C
is possible, particularly if blending components or their higher boiling cuts such as those derived from fluid catalytic cracking
(FCC) are analyzed, and the total olefin content may not be accurate. Caution should also be exercised when analyzing olefin-free
samples using this test method as some of the paraffins may be reported as olefins since analysis is based purely on retention times
of the eluting components.
1.4.1 Total olefins in the samples may be obtained or confirmed, or both, if necessary, by Test Method D1319 (percent volume)
or other test methods, such as those based on multidimensional PONA type of instruments (Test Method D6839).
1.5 If water is or is suspected of being present, its concentration may be determined, if desired, by the use of Test Method D1744,
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.04.0L on Gas Chromatography Methods.
Current edition approved Oct. 1, 2014June 1, 2020. Published February 2015October 2020. Originally approved in 2001. Last previous edition approved in 20092014 as
D6729 – 04 (2009).D6729 – 14. DOI: 10.1520/D6729-14.10.1520/D6729-20.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6729 − 20
or equivalent. Other compounds containing oxygen, sulfur, nitrogen, and so forth, may also be present, and may co-elute with the
hydrocarbons. If determination of these specific compounds is required, it is recommended that test methods for these specific
materials be used, such as Test Methods D4815 and D5599 for oxygenates, and D5623 for sulfur compounds, or equivalent.
1.6 Annex A1 of this test method compares results of the test procedure with other test methods for selected components, including
olefins, and several group types for several interlaboratory cooperative study samples. Although benzene, toluene, and several
oxygenates are determined, when doubtful as to the analytical results of these components, confirmatory analyses can be obtained
by using specific test methods.
1.7 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.9 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1319 Test Method for Hydrocarbon Types in Liquid Petroleum Products by Fluorescent Indicator Adsorption
D1744 Test Method for Determination of Water in Liquid Petroleum Products by Karl Fischer Reagent (Withdrawn 2016)
D4815 Test Method for Determination of MTBE, ETBE, TAME, DIPE, tertiary-Amyl Alcohol and C to C Alcohols in
1 4
Gasoline by Gas Chromatography
D5599 Test Method for Determination of Oxygenates in Gasoline by Gas Chromatography and Oxygen Selective Flame
Ionization Detection
D5623 Test Method for Sulfur Compounds in Light Petroleum Liquids by Gas Chromatography and Sulfur Selective Detection
D6839 Test Method for Hydrocarbon Types, Oxygenated Compounds, and Benzene in Spark Ignition Engine Fuels by Gas
Chromatography
E355 Practice for Gas Chromatography Terms and Relationships
3. Terminology
3.1 Definitions—This test method makes reference to many common gas chromatographic procedures, terms, and relationships.
Detailed definitions can be found in Practice E355.
4. Summary of Test Method
4.1 Representative samples of the petroleum liquid are introduced into a gas chromatograph equipped with an open tubular
(capillary) column coated with the specified stationary phase. Helium carrier gas transports the vaporized sample through the
column, in which it is partitioned into individual components which are sensed with a flame ionization detector as they elute from
the end of the column. The detector signal is recorded digitally by way of an integrator or integrating computer. Each eluting
component is identified by comparing its retention time to that established by analyzing reference standards or samples under
identical conditions. The concentration of each component in mass percent is determined by normalization of the peak areas after
correction of selected components with detector response factors. The unknown components are reported individually and as a
summary total.
5. Significance and Use
5.1 Knowledge of the specified individual component composition (speciation) of gasoline fuels and blending stocks is useful for
refinery quality control and product specification. Process control and product specification compliance for many individual
hydrocarbons may be determined through the use of this test method.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
D6729 − 20
6. Apparatus
6.1 Gas Chromatograph, a gas chromatograph equipped with cryogenic column oven cooling and capable of producing repeatable
oven ramps from 0 °C to at least 300 °C is required. The following features are useful during the sample analysis phase: electronic
flow readout, electronic sample split-ratio readout, and electronic pneumatic control of flow. Though their use is not required,
careful review of this test method will demonstrate the usefulness of a gas chromatograph equipped with these features. These
features will replace the need to carry out the manual calculations that must be performed as listed in 8.1 and 8.2.
6.2 Inlet—a capillary split/splitless inlet system operated in the split mode is recommended. It must be operated in its linear range.
Refer to 8.4 to determine the proper split ratio.
6.2.1 Carrier Gas Pneumatic Control—Constant carrier gas pressure control was used by all cooperative study participants. This
may be either direct pressure to the inlet (injector) or by using a total flow/back pressure system.
6.2.2 Pneumatic Operation of the Chromatograph—The use of constant pressure was the mode of operating the gas
chromatography used by the participants in the interlaboratory cooperative study. Other carrier gas control methods such as
constant flow (pressure programming) may be used, but this may change the chromatography elution pattern unless the temperature
programming profile is also adjusted to compensate for the flow differences.
6.2.3 Temperature Control—The injector operated in the split mode shall be heated by a separate heating zone and heated to
temperatures of 200 °C to 275 °C.
6.3 Column, a fused silica capillary column, 100 m in length by 0.25 mm inside diameter, coated with a 0.5 μm film of bonded
dimethylpolysiloxane. The column must meet the resolution requirements expressed in 8.3. Columns from two different
commercial sources were used in the interlaboratory cooperative study.
6.4 Data System, a computer based chromatography data system capable of accurately and repeatedly measuring the retention time
and areas of eluting peaks. The system shall be able to acquire data at a rate of at least 10 Hz. Although it is not mandatory, a data
system which calculates column resolution (R) is extremely useful as it will replace the need to carry out the manual calculations
which must be performed as listed in 8.3.
6.4.1 Electronic Integrators, shall be capable of storing up to 400 components in the peak table and shall be able to acquire the
data at 10 Hz or faster speeds. They shall be capable of integrating peaks having peak widths at half height which are 1.0s wide.
The integrator must be capable of displaying the integration mode of partially resolved peaks. In addition, these integrators should
be able to download a commonly readable format of data (that is, ASCII) to a computer in order to facilitate data processing.
6.5 Sample Introduction—Sample introduction by way of a valve, automatic injection device, robotic arm or other automatic
means is highly recommended. An automatic sample introduction device is essential to the reproducibility of the analysis. Manual
injections are not recommended. All of the reproducibility data reported by this test method for the samples analyzed were gathered
using automatic injection devices.
6.6 Flame Ionization Detector (FID)—The gas chromatograph should possess a FID having a sensitivity of 0.0050.005 coulombs
coulombs/g ⁄g for n-butane. The linear dynamic range of the detector should be 10 or better. The detector is heated to 300 °C.
7. Reagents and Materials
7.1 Calibrating Standard Mixture—A spark ignition engine fuel standard of known composition and concentration by mass can
be used. In order to corroborate the identification of the sample, a typical chromatogram (Fig. 1) was obtained from reference
sample ARC96OX.
7.2 Gas Chromatograph Gases—All of the following gases shall have a purity of 99.999 % (V/V)(V ⁄V) or greater.
NOTE 1—Warning: Gases are compressed. Some are flammable and all gases are under high pressure.
Reference spark ignition sample No. ARC 960X obtained from the Alberta Research Council, Edmonton, Alberta, Canada. Other samples are available from suppliers.
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
7.2.1 Helium—The test data was developed with helium as the carrier gas. It is possible that other carrier gases may be used for
this test method. At this time, no data is available from this test method with other carrier gases.
7.2.2 Air, Hydrogen and Make-up Gas (Helium or Nitrogen), shall have a purity of 99.999 % (V/V) or greater.
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
8. Instrument Check Out Prior to Analysis
8.1 Setting:
8.1.1 Linear Gas Velocity—If the gas chromatograph is equipped with an electronic flow readout device, set the flow to 1.8
mL/min. This is achieved by setting the carrier gas flow rate by injection of cm/s methane or natural gas at 35 °C. Ensure that the
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
retention time is 7.00 min 6 0.05 min. This corresponds to a linear velocity of 25 cm/s to 26 cm/s. This is equivalent to retention
times of methane at 0 °C ranging from 6.5 min to 6.8 min.
8.1.2 If the gas chromatograph is not equipped with an electronic flow readout device, calculate the linear gas velocity in cm/s
using Eq 1.
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
column length ~cm!
linear gas velocity 5 V 5 (1)
retention time of methane s
~ !
8.1.3 The typical retention times for methane and linear gas velocity for helium are 6.5 cm ⁄s to 6.8 cm ⁄s and 24 cm ⁄s to 26 cm ⁄s,
respectively.
D6729 − 20
FIG. 1 Chromatogram for Reference Spiked Gasoline (continued)
8.2 Setting the Split Ratio—If the gas chromatograph is equipped with an electronic split-ratio readout device, set the split ratio
to a sample split of 200:1. If the gas chromatograph is not equipped with an electronic split-ratio readout device, one must first
calculate column flow rate and then proceed to calculating split ratio using Eq 2 and 3.
D6729 − 20
60 π r L T 2 P 2 P
~ ! ~ ! ~ !
ref i o
column flow rate 5 F 5 (2)
2 2
~T!3~P !~P 2 P !μ
ref i o
where:
F = flow rate as calculated by using the equation,
r = column radius, cm,
L = column length, cm,
P = inlet pressure,
i
P = outlet pressure,
o
P = reference pressure, 1 atm,
ref
T = temperature of the column oven,
T = temperature at the column outlet, and
ref
μ = linear velocity, cm/s.
split vent flow1F
split ratio 5 S 5 (3)
F
8.2.1 The column flow rate is calculated by the use of Eq 2. Use the results obtained from Eq 3 to adjust the split flow until a split
flow of approximately 200:1 is achieved.
8.3 Evaluation of Column Performance:
8.3.1 Prior to using the column described in Table 1, measure the resolution of the column under the conditions of Table 2. Check
that the resolution for the following pairs of components is obtained using Eq 4 to calculate the resolution of a pair of components:
2 t 2 t
~ !
R2 R1
R 5 (4)
1.699 ~W 1W !
h1 h2
TABLE 1 Chromatographic Operating Conditions, Column Requirements and Data Acquisition Requirements
Chromatographic Conditions Requirements
Injector settings
Injector temperature, °C 250
Split ratio 175:1 - 275:1
Liner deactivated glass
Injection volume, μL 0.2–0.5
Detector settings
A
FID detector temperature, °C 300–350
Gas flows
B
Hydrogen, mL/min 30–40
Air, mL/min 300–450
Nitrogen make up, mL/min 30
Column oven settings
Initial temperature, °C 0
Initial time, min 15
1st ramp rate, °C/min 1
Final temperature, °C 50
Final time, min 0
2nd ramp rate, °C/min 2
Final temperature, °C 130
Final time, min 0
3rd ramp rate, °C/min 4
Final temperature, °C 270
C
Final time, min 0
Column Requirements
Length, m 100
Inside diameter, mm 0.25
Liquid phase 100 % dimethylpolysiloxane
Film thickness, μ 0.5
Pressure, psig 40–50
Flow, mL/min 1.7–2.0
Linear gas velocity, cm/s 24.5
Data acquisition, Hz 10–20
Total analysis time, min 140–150
A
Set to 25 °C to 50°C above the highest column temperature.
B
Values to be set as recommended by instrument manufacturer.
C
Final temperature or time may be adjusted to ensure complete elution of the sample components.
D6729 − 20
TABLE 2 Resolution Performance Requirements
Concentration of
Component Minimum
Each
Pair Resolution
Component, W/W
Benzene 1.0 0.5 %–0.5 %
1-Methyl-cyclopentene
m-Xylene 0.4 2.0 %–2.0 %
p-Xylene
n-Tridecane 1.0 0.5 %–0.5 %
1-Methylnaphthalene
where:
R = resolution,
t = retention time of the first member of the pair,
R2
t = retention time of the second member of the pair,
R1
W = peak width at half height of the first member of the pair, and
h1
W = peak width at half height of the second member of the pair.
h2
8.3.1.1 Column resolution should be checked frequently by examining the resolution of these compounds.
8.3.2 Evaluation of the Baseline—Carry out a blank baseline run utilizing no solvent injection, by setting the GC in accordance
with the conditions of Table 1.
8.3.3 Subtract the baseline from a sample chromatogram and verify that the residual signal at the beginning of the chromatogram
does not differ from the end of the chromatogram by more than 2 %.
8.4 Evaluation of Splitter Linearity—Using the reference gasoline sample, inject this sample according to the schedule listed in
Table 3.
8.4.1 Select from the chromatogram about 10 to 15 components, which have concentrations in the range of 0.01 % mass to 30 %
mass. Tabulate for each split ratio the concentrations of the 10 to 15 components. Verify that for each component selected, its
concentration does not vary by more than 3 %.
9. Procedure
9.1 Set the operating conditions of the gas chromatograph as shown in Table 1. These conditions will elute all components up to
and including pentadecane (nC ).
9.2 All of the parameters in Table 1 can be marginally changed to optimize for sample types and optimize for characteristics of
each gas chromatographic system. The final boiling point of samples should not exceed nC and the column resolution (R)
performance requirements listed in Table 2 should not be compromised.
9.3 Obtain a representative sample following the guidelines of Practice D4057 and any other applicable guidelines. Take
precautions to minimize the loss of light ends from volatile samples. The sample container may be cooled prior to transfer of
sample into it. Cool the sample to less than 4 °C, maintain at that approximate temperature until the autosampler is loaded and
analysis begins.
9.4 Preparation/Storage:
TABLE 3 Injection Schedule
Injection Volume, Injection Temperature,
Split Ratio
μL °C
100:1 0.1 250
200:1 0.5 250
300:1 1.0 250
D6729 − 20
A
TABLE 4 Predominant Compounds and Identified Coeluting Compounds
NOTE 1—The response factor of the predominant compound will be used for the analyte and this analyte will be used for the calculations.
Peak Number Predominant Coeluting
(from Annex A1) Compound Compound(s)
164 3,3-dimethylpentane 5-methyl-1-hexene
186 2-methylhexane C -olefin
278 2,5-dimethylhexane C -olefin
286 3,3-dimethylhexane C -olefin
B
304 toluene 2,3,3-trimethylpentane
324 1,1,2-trimethylcyclopentane C -triolefin
326 C -diolefin C -paraffin
8 8
492 4-methyloctane C -olefin
796 1,2,3,4-tetramethylbenzene C -aromatic
A
This is not an exhaustive list. Due to the possibility of coeluting peaks in other areas, the user is cautioned in the interpretation of the data.
B
In most alkylated gasolines, a split may occur between toluene and 233 TMC5.
TABLE 5 Response Factors of Oxygenated Compounds
Relative Response Factors
Analytes
RRF C = 1.000 RRF CH = 1.000
7 4
Methanol 2.996 2.672
Ethanol 2.087 1.862
t-Butanol (TBA) 1.302 1.161
Methyl-t-butyl ether (MTBE) 1.577 1.407
Ethyl-t-butyl ether (ETBE) 1.407 1.255
t-Amyl methyl ether (TAME) 1.356 1.210
9.4.1 Samples Stored in Vials—Cool the original sample to less than 4 °C prior to taking a sample aliquot or prior to filling the
sample vials. The sample aliquot container, or the vial, or both, can also be cooled prior to the transfer of the original sample.
Syringes may also be cooled along with the sample for manual injections.
9.4.2 Samples Stored in Pressurized Containers—It is recommended that they be kept away from direct heat or light. No other
sample preparations are necessary for samples stored in pressurized containers. Avoid storage at temperatures greater than 25 °C.
Store pressure containers in accordance with the manufacturer’s instructions.
9.5 It is recommended that a quality assurance (QA) sample similar to the reference material gasoline sample be run at regular
intervals (see Fig. 1). An interval of once per week or after every 15 samples is recommended. The quantitation results use
statistical quality control charts can track benzene. Other components of interest in the reference sample can be tracked in a similar
manner. By monitoring these components over an extended period of time, the performance of the column and the chromatographic
system can be determined.
10. Data Analysis
10.1 Compound Identification—Prepare a table listing all of the retention times of the components in the sample. Compare the
retention time of each peak with that of the reference gasoline. Pay particular attention to the fact that columns can be overloaded,
and peaks can shift in retention time. Observe the peak pattern so that proper identification is made by comparison with the
reference material.
10.2 Consistency in peak identification can be achieved by using software (data handling software, spreadsheet software, and so
forth). Alternatively, a retention index system can be used.
log~T ! 2 log~T !
i n
R1 5 100n1100 (5)
~ ! F G
i
log TN 2 log T
~ ! ~ !
n
where:
(R1) = retention index of component I bracketed by the N-paraffin, n in its lower boundary and N-paraffin N in its upper
i
boundary,
T = adjusted retention time of component i (retention time of component i minus the retention time of methane),
i
T = adjusted retention time of N-paraffin n, and
n
D6729 − 20
TN = retention time of N-paraffin N.
10.3 Determine the hydrocarbon response factors by using the following equation.
MW 1
i
RRF 5 3 (6)
CH4
N MW
c CH4
where:
RRF = relative response factor of each component with respect to methane (RRF =1.000),
CH4 CH4
MW = molecular weight of the component, i,
i
N = number of carbon atoms in the molecule, and
c
MW = molecular weight of methane (16.04276).
CH4
10.4 Convert the acquired areas to corrected areas by multiplying each area by its corresponding relative response factor as
indicated in the following equation.
A 5 A RRF (7)
~ ! ~ !
c I I
i
where:
(A ) = corrected area,
c i
A = acquired area for an individual component, and
i
RRF = relative response factor (weight basis).
10.4.1 The percent mass (% W) is calculated as follows:
A
~ !
C i
% W 5 3100 (8)
i i5n
A
c
(
i
i51
where:
% W = percent mass of the component i in the mixture, and
i5n
= summation of all the corrected areas for the components analyzed.
A
(
c
i
i51
10.4.1.1 The subscript i indicates that the operations are carried out for each individual component in the matrix.
10.5 In the case of unidentified components, utilize a relative response factor of 0.800 (relative to methane).
11. Oxygenates
11.1 A cooperative study for linearity was performed for methanol, ethanol, t-butanol, methyl-t-butyl ether (MTBE), ethyl-t-butyl
ether (ETBE), and t-amyl methyl ether (TAME) in concentration ranges from 1.0 % mass up to 30 % mass (Annex A2). The
average relative response factors for the oxygenates were calculated from the study and are listed in Table A2.1. They have been
incorporated into the IHA Method. The percent standard deviation of these relative response values was as high as 7 %. MTBE
was the only oxygenate that was present in sufficient number of samples to meet the ASTM requirements for round robin testing
in accordance with RR:D02-1007. Therefore the statistical data for MTBE should be taken from Table A1.2.
12. Expression of Results
12.1 Report the concentration of each components as percent (m/m) to the nearest 0.001 % mass.
12.2 The data for individual components may by grouped by summing the concentration of compounds in each particular group
Sevcik, J., Detectors in Gas Chromatography, Elsevier, NY, 1976, p. 94.
D6729 − 20
type such as paraffin, isoparaffin, olefin, aromatic, naphthene, oxygenates, and unknowns. Commercially available software may
be used to provide this function, as well as the calculation of other properties of petroleum liquids.
13. Precision and Bias
13.1 The repeatability and reproducibility precision estimates are quoted in Annex A1.
13.2 Precision Statement Outline—(> Analyte Qualification Process):
13.2.1 For each analyte to qualify for a precision statement, it must be present in at least six samples, and detected by at least six
laboratories, at least once, in accordance with RR:D02-1007 requirements.
13.2.2 The (repeatability standard deviation)/mean value for each analyte/sample combination must be less than or equal to 0.1,
in accordance with LOQ requirements which, while not a standard, is what CS94 is recommending.
13.3 A brief explanation of headers in Table A1.2 follows:
13.3.1 ID: self explanatory,
13.3.2 r : lower 95 % confidence limit of r ,
min est
13.3.3 r : repeatability estimate in percentage of concentration,
est
13.3.4 r : upper 95 % confidence limit of r ,
max est
13.3.5 R , R , R : same as above except for reproducibility,
min est max
13.3.6 C : lower concentration limit that rest, R is applicable, and
min est
13.3.7 C : upper concentration limit that rest, R is applicable.
max est
13.4 The summaries for the paraffins, isoparaffins, C benzene, and oxygenates follow the same procedure that was used for the
analytes and are listed in Table A1.3.
13.5 Bias—The bias of this test method cannot be determined since an appropriate standard reference material is not available.
14. Keywords
14.1 gas chromatograph; gasoline; individual hydrocarbon analysis; oxygenated fuels; spark-ignition engine fuels
ANNEXES
(Mandatory Information)
A1. HYDROCARBON DATA
A1.1 Table A1.1 presents the component retention times and properties.
Supporting data describing the interlaboratory cooperative study to determine precision and bias has been filed at ASTM International Headquarters and may be obtained
by requesting RR: RR:D02-1519. Contact ASTM Customer Service at service@astm.org.
Supporting data have been filed at ASTM International Headquarters and may be obtained by requesting Research Report RR:D02-1007. Contact ASTM Customer
Service at service@astm.org.
D6729 − 20
A1.2 Table A1.2 represents the repeatability and reproducibility precision estimates prepared by statisticians of CS94 in
accordance with RR:D02-1007RR:D02-1519. The analyte qualification process for precision statements is outlined as follows:
A1.2.1 For each analyte to qualify for a precision statement, it must be present in at least six samples, and detected by at least
six laboratories, at least once, in accordance with RR:D02-1007 requirements.
A1.2.2 The (repeatability standard deviation)/mean value for each analyte/sample combination must be less than or equal to 0.1,
in accordance with LOQ requirements which, while not a standard, is what CS94 is recommending.
A1.3 Summary for Oxygenates:Warning—The statistical data could be done on the oxygenates but there was not an equal
number of all oxygenates in the round robin. MTBE was the largest contributor to the statistical results. The number of samples
that contained each oxygenate is as follows:
Oxygenate No. of Approximate
Type Samples Concentration Range
Ethanol 2 1 %, and 12 %
t-butanol 2 0.20 %, and 1.0 %
MTBE 6 1, 2, 4, 4, 8 and 16 %
ETBE 1 0.50 %
TAME 1 15.00 %
A1.4 The precision statement for the olefins and cycloparaffins is determined by taking the square root of the value determined
in the summary; multiply by the coefficient (r ) for repeatability and the coefficient (R ) for the reproducibility.
coef coef
Name r r r R R R C C
min coef max min coef max min max
Cyclo- 0.0726 0.08 0.098 0.286 0.384 0.586 2 10
paraffins
Olefins 0.1555 0.18 0.21 0.382 0.555 1.012 2 25
A1.5 The precision for the aromatics does not depend on level and is stated below in mass percent.
Name r r % r R R % R C C
min max min max min max
Aromatics 0.8549 0.98 1.155 2.151 2.706 3.651 15 50
A1.6 The summaries for the paraffins, isoparaffins, C benzene and oxygenates follow the same procedure that was used for the
analytes. The statistics for the grouping are shown in Table A1.3 as an indication of reproducibility and repeatability of reporting
the results as a group summary. However, there is a possibility that significant error could occur due to co-elution of peaks, the
presence of significant amounts of olefinic or naphthenic constituents, or both, above octane and the percent unknown in the
sample. If more accurate summary results are needed that are not covered by the above precision statement, for some or all of the
above families of components, please consider another ASTM test method.
D6729 − 20
TABLE A1.1 Component Retention Times and Properties
NOTE 1—The names used are from several other tables and changes have been made where the GCMS did not agree with the peak name or its retention
time.
NOTE 2—n-propanol will coelute with 3M-1-C5=.
NOTE 3—MTBE will coelute with 23DN-1C4=.
NOTE 4—MSBE will coelute with 1-hexene.
NOTE 5—ETBE will coelute with 23DM-13C4= =.
NOTE 6—isobutanol will coelute with 44DM-1-c5=.
NOTE 7—233TM pentane will coelute with toluene when the ratio with toluene is greater than 5.0:1.
NOTE 8—The coeluting olefins in Notes 2-6 will usually be below 1000 ppm.
NOTE 9—In some instances the chemical group is known, but the chemical structure is not known (for example, C -olefin; the position of the double
bond is not known).
NOTE 10—Relative response factors for six of the major oxygenated compounds have been determined by using the average results from seven
laboratories analyzing six samples in duplicate. These same samples were used to determine linearity of methanol, ethanol, t-butanol, MTBE, ETBE and
TAME from a concentration level ranging from 1 % mass up to 30 % mass.
Peak No. NewCompound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
1 Methane 6.74 16.04 1.000
2 Ethene 7.10 28.05 0.874
3 Ethane 7.21 30.07 0.937
4 Propene 7.41 42.05 0.874
5 Propane 7.87 44.11 0.916
6 Isobutane 8.26 58.12 0.906
7 Methanol 8.64 32.03 2.672
8 Isobutene 8.95 56.11 0.874
9 1-butene 8.99 56.11 0.874
10 1,3-butadiene 9.17 54.09 0.843
12 N-butane 9.28 58.12 0.906
14 Trans-2-butene 9.70 56.11 0.874
16 2,2-dimethylpropane 9.82 72.15 0.899
18 Cis-2-butene 10.33 56.11 0.874
20 1,2-butadiene 10.88 54.09 0.843
22 Ethanol 11.39 46.07 1.862
24 3-methyl-1-butene 12.21 70.13 0.874
26 Isopentane 13.57 72.15 0.899
28 1,4-pentadiene 14.25 68.12 0.849
30 2-Butyne (dimethylacetylene) 14.57 54.09 0.843
32 1-pentene 15.03 70.13 0.874
34 Isopropanol 15.28 60.11 1.950
36 2-methyl-1-butene 15.76 70.13 0.874
38 N-pentane 16.24 72.15 0.899
40 2-methyl-1,3-butadiene 16.73 68.12 0.849
42 Trans-2-pentene 17.23 70.13 0.874
44 3,3-dimethyl-1-butene 17.86 84.16 0.874
46 Cis-2-pentene 18.17 70.13 0.874
48 Tert-butanol (TBA) 18.51 74.12 1.161
50 2-methyl-2-butene 18.76 70.13 0.874
52 Trans-1,3-pentadiene 19.12 68.12 0.849
54 3-methyl-1,2-butadiene 19.48 68.12 0.849
56 Cyclopentadiene 19.76 67.10 0.824
56 Cyclopentadiene 19.76 66.10 0.824
58 Cis-1,3-pentadiene 20.25 68.12 0.849
60 1,2-pentadiene 20.51 68.12 0.849
62 2,2-dimethylbutane 20.69 86.18 0.895
64 Cyclopentene 23.16 68.12 0.849
66 4-methyl-1-pentene 24.30 84.16 0.874
68 3-methyl-1-pentene 24.38 84.16 0.874
70 n-propanol 24.68 60.11 1.770
72 Cyclopentane 24.86 70.13 0.874
74 2,3-dimethylbutane 25.57 86.18 0.895
76 2,3-dimethyl-1-butene 25.99 84.16 0.874
78 Methyl tert-butyl ether (MTBE) 26.18 88.09 1.407
80 Cis-4-methyl-2-pentene 26.48 84.16 0.874
82 2-methylpentane 26.66 86.18 0.895
84 Trans-4-methyl-2-pentene 72.09 84.16 0.874
84 Trans-4-methyl-2-pentene 27.09 84.16 0.874
86 Methyl ethyl ketone (MEK) 28.00 72.06 1.570
88 3-methylpentane 29.15 86.18 0.895
90 C -olefin 29.61 84.16 0.874
92 2-methyl-1-pentene 30.29 84.16 0.874
D6729 − 20
TABLE A1.1 Continued
Peak No. NewCompound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
94 1-hexene 30.52 84.16 0.874
96 Methyl sec-butyl ether (MSBE) 30.66 88.09 1.550
98 C -olefin 30.94 84.16 0.874
100 2-butanol 31.56 74.12 1.600
102 2ethyl-1-butene 32.47 84.16 0.874
104 N-hexane 32.75 86.18 0.895
106 Cis-3-hexene 33.41 84.16 0.874
108 Di-isopropyl ether (DIPE) 33.58 102.00 1.600
110 Trans-3-hexene+hexadiene 33.86 84.16 0.874
112 2-methyl-2-pentene 34.33 84.16 0.874
114 3-methylcyclopentene 34.57 82.10 0.853
116 Trans-3-methyl-2-pentene 34.71 84.16 0.874
118 Cis-2-hexene 35.62 84.16 0.874
120 3,3-dimethyl-1-pentene 36.04 98.19 0.874
122 Cis-3-methyl-2-pentene 36.92 84.16 0.874
124 Ethyl tert-butyl ether (ETBE) 37.07 102.18 1.255
126 2,3-dimethyl-1,3-butadiene 37.19 82.10 0.853
128 Methylcyclopentane 37.40 84.16 0.874
130 2,2-dimethylpentane 37.60 100.21 0.892
132 4,4-dimethyl-1-pentene 37.91 98.19 0.874
134 Isobutanol 38.06 74.12 1.500
136 2,3-dimethyl-2-butene 38.30 84.16 0.874
138 2,4-dimethylpentane 38.99 100.21 0.892
140 1,3,5-hexatriene 39.31 80.06 0.832
142 2,2,3-trimethylbutane 39.48 100.21 0.892
144 Methylcyclopentadiene 40.17 80.06 0.832
146 C -olefin 40.30 98.19 0.874
148 C -olefin 40.68 98.19 0.874
150 C -diolefin 41.20 96.18 0.856
152 4-methylcyclopentene 41.44 82.10 0.853
154 Methylenecyclopentane 42.08 82.10 0.853
156 Benzene 42.30 78.05 0.812
158 1-methyl-1-cyclopentene 42.46 82.10 0.853
160 C -olefin 43.06 98.19 0.874
162 C -methyl-3-hexene 43.37 98.19 0.874
162 Cis-2-methyl-3-hexene 43.37 98.19 0.874
164 3,3-dimethylpentane+5-methyl- 43.81 100.21 0.892
1-hexene
166 Cyclohexane 44.07 84.16 0.874
168 Trans-2methyl-3-hexene 44.82 98.19 0.874
168 Trans-2-methyl-3-hexene 44.82 98.19 0.874
170 3,3-dimethyl-1,4-pentadiene 45.44 96.18 0.856
172 N-butanol 45.58 74.12 1.500
174 Dimethylcyclopentadiene 45.69 94.17 0.838
176 t,2-ethyl-3-methyl-1-butene 45.97 98.19 0.874
178 4-methyl-1-hexene 46.27 98.19 0.874
180 C -olefin 46.55 98.19 0.874
182 3-methyl-1-hexene 46.78 98.19 0.874
184 4-methyl-2-hexene 46.92 98.19 0.874
186 2-methylhexane+C -olefin 47.29 100.21 0.892
188 2,3-dimethylpentane 47.51 100.21 0.892
190 Cyclohexene 47.65 82.10 0.853
192 Tert-amyl methyl ether (TAME) 48.10 102.18 1.210
194 C -olefin 48.46 98.19 0.874
196 C -olefin 48.64 98.19 0.874
198 3-methylhexane 49.05 100.21 0.892
200 C -olefin 49.47 98.19 0.874
202 C -olefin 49.62 98.19 0.874
204 Trans-1,3-dimethylcyclopentane 49.83 98.19 0.874
206 Cis-1,3-dimethylcyclopentane 50.40 98.19 0.874
208 Trans-1,2-dimethylcyclopentane 51.01 98.19 0.874
210 3-ethylpentane 51.21 100.10 0.892
212 C -olefin 51.43 98.19 0.874
214 2,2,4-trimethylpentane 51.61 114.23 0.890
216 C -olefin 51.75 98.19 0.874
218 1-heptene 52.05 98.19 0.874
220 C -olefin 52.18 98.19 0.874
222 2,3-dimethyl-1,3-pentadiene 52.69 96.18 0.874
222 2,3-dimethyl-1,3-pentadiene 52.69 96.18 0.856
224 C -diolefin 53.00 96.18 0.856
226 C -olefin 53.36 98.19 0.874
228 C -diolefin 53.81 96.18 0.856
230 C -diolefin 54.13 96.18 0.856
232 C -olefin 54.28 98.19 0.874
234 N-heptane 54.59 100.21 0.892
D6729 − 20
TABLE A1.1 Continued
Peak No. NewCompound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
236 Cis-3-heptene 54.81 98.19 0.874
238 2-methyl-2-hexene 55.10 98.19 0.874
240 Cis-methyl-3-hexene 55.35 98.19 0.874
240 Cis-3-methyl-3-hexene 55.35 98.19 0.874
242 Trans-3-heptene 55.72 98.19 0.874
244 3-ethyl-2-pentene 55.88 96.18 0.856
244 3-ethyl-2-pentene 55.88 96.19 0.874
246 1,5-dimethylcyclopentene 56.06 96.18 0.856
248 Trans-2-methyl-3-hexene 56.58 98.19 0.874
248 Cis-3-methyl-2-hexene 56.58 98.19 0.874
250 C -diolefin+C -triolefin 57.01 96.18 0.856
7 7
252 2,3-dimethyl-2-pentene 57.35 98.19 0.874
254 3-ethylpentene 57.57 98.19 0.874
256 Methylcyclohexane 57.79 98.19 0.874
258 C -olefin 58.28 98.19 0.874
260 1,1,3-trimethylcyclopentane 58.79 112.22 0.874
262 2,2-dimethylhexane 59.29 114.10 0.890
264 2,3,4-trimethyl-1,4-pentadiene 59.45 110.21 0.859
266 3,3-dimethyl-1,5-hexadiene 59.79 110.21 0.859
268 C -olefin 60.12 98.19 0.874
268 C -olefin 60.12 112.22 0.874
270 Ethylcyclopentane 60.60 98.19 0.874
272 3-methylcyclohexene 60.99 96.18 0.856
274 Methylcyclohexadiene 61.14 94.17 0.838
276 2,2,3-trimethylpentane 61.22 114.10 0.890
278 2,5-dimethylhexane+C -olefin 61.59 114.23 0.890
280 2,4-dimethylhexane 61.91 114.23 0.890
282 C -triolefin+C -olefin 62.28 112.24 0.856
7 8
284 Trans,cis-1,2,4- 62.68 112.22 0.874
trimethylcyclopentane
286 3,3-dimethylhexane+C -olefin 63.13 114.23 0.890
288 C -triolefin+C -olefin 63.39 112.24 0.856
7 8
290 C -olefin 63.69 112.22 0.874
292 Trans,cis-1,2,3- 64.27 112.22 0.874
trimethylcyclopentane
294 C -olefin 64.52 112.22 0.874
296 C -olefin 64.73 112.22 0.874
298 C -olefin 64.82 112.22 0.874
300 2,3,4-trimethylpentane 64.94 114.23 0.890
302 C -diolefin 65.25 96.18 0.856
304 Toluene 65.50 92.06 0.821
306 2,3,3-trimethylpentane 65.76 114.23 0.890
308 C -olefin 65.90 112.22 0.874
310 C -diolefin 66.12 110.21 0.859
312 C -olefin 66.48 112.22 0.874
314 C -olefin 66.65 112.22 0.874
316 C -olefin 67.08 112.22 0.874
318 C -diolefin+C -olefin 67.30 110.21 0.859
8 8
318 C -olefin 67.30 112.22 0.874
320 2,3-dimethylhexane 67.47 114.23 0.890
322 2-methyl-3-ethylpentane 67.71 114.23 0.890
324 1,1,2-trimethylcyclopentane+C - 68.04 112.22 0.874
triolefin
326 C -diolefin+C -paraffin 68.31 114.23 0.859
8 8
328 C -olefin 68.41 112.22 0.874
330 C -olefin 68.64 112.22 0.874
332 2-methylheptane 68.86 114.23 0.890
334 4-methylheptane 69.11 114.23 0.890
336 C -diolefin+C -olefin 69.41 112.22 0.874
8 7
338 C -olefin 69.70 112.22 0.874
340 Cis-1,3-dimethylcyclohexane 69.91 112.22 0.874
342 Trans-1,4-dimethylcyclohexane 70.01 112.22 0.874
344 3-methylheptane 70.23 114.23 0.890
346 3-ethylhexane 70.38 114.23 0.890
348 C -diolefin 70.51 110.21 0.874
350 C -olefin 70.72 112.22 0.874
352 C -olefin 70.92 112.22 0.874
354 1,1-dimethylcyclohexane 71.18 112.22 0.874
356 C -olefin 71.43 112.22 0.874
358 C -olefin 71.70 112.22 0.874
360 Cis-1-ethyl-3- 72.10 112.22 0.874
methylcyclopentane
362 2,2,5-trimethylhexane 72.23 128.26 0.888
364 Trans-1-ethyl-3- 72.46 112.22 0.874
methylcyclopentane
D6729 − 20
TABLE A1.1 Continued
Peak No. NewCompound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
366 Trans-1-ethyl-2- 72.68 112.22 0.874
methylcyclopentane
368 1-methyl-1-ethylcyclopentane 72.96 112.22 0.874
370 1-octene 73.16 112.22 0.874
372 C -olefin 73.26 112.22 0.874
374 Trans-1,2-dimethylcyclohexane 73.36 112.22 0.874
376 C -olefin 73.48 112.22 0.874
378 C -olefin 73.68 112.22 0.874
380 Trans-3-C -Olefin 74.08 112.11 0.874
380 C -olefin 74.08 112.11 0.874
382 C -olefins 74.45 112.22 0.874
384 Trans-1,3-dmcyclohexane 74.66 112.22 0.874
386 Cis-1,4-dmcycloHexane 74.79 112.22 0.874
388 N-octane 74.98 114.23 0.890
390 C -olefin 75.33 112.22 0.874
392 C -olefin 75.49 112.22 0.874
394 Trans-2-octene 75.62 112.22 0.874
396 Isopropylcyclopentane 75.72 112.22 0.874
398 C -olefin 75.85 126.24 0.874
400 C -olefin 75.89 126.24 0.874
402 C -olefin 75.90 126.24 0.874
404 C -olefin 76.08 126.24 0.874
406 2,2,4-trimethylhexane 76.31 128.26 0.888
408 2,4,4-trimethylhexane 76.62 128.26 0.888
410 C -olefins 76.86 126.24 0.874
412 2,3,5-trimethylhexane 77.29 128.26 0.888
414 Cis-2-octene 77.53 112.22 0.874
416 2,2,3,4-tetramethylpentane 77.77 128.26 0.888
418 2,2-dimethylheptane 78.02 128.26 0.888
420 Cis-1,2-dimethylcyclohexane 78.36 112.22 0.874
422 2,4-dimethylheptane 78.74 128.26 0.888
424 C -olefin 78.90 126.24 0.874
426 C -olefin 79.08 126.24 0.874
428 Ethylcyclohexane 79.24 112.22 0.874
430 Propylcyclopentane 79.39 112.22 0.874
432 2-methyl-4-ethylhexane 79.59 128.26 0.888
434 2,6-dimethylheptane 79.74 128.26 0.874
434 2,6-dimethylheptane 79.74 128.26 0.888
436 C -olefin 79.85 126.24 0.874
438 1,1,4-trimethylcyclohexane 80.05 126.24 0.874
440 C -olefin 80.28 126.24 0.874
442 C -olefin 80.38 126.24 0.874
444 1,1,3-trimethylcyclohexane 80.52 126.24 0.874
446 2,5 & 3,5-dimethylheptane 80.69 128.26 0.888
448 C -olefin 80.88 126.24 0.874
450 3,3-DMheptane 81.00 128.26 0.888
452 C -paraffin 81.13 128.26 0.888
454 C -olefin 81.34 126.24 0.874
456 2,3,3-trimethylhexane 81.56 128.26 0.888
458 C -olefin 81.68 126.24 0.874
460 Ethylbenzene 81.96 106.08 0.827
462 C -olefin 82.00 126.24 0.874
464 Trans-1,2,4- 82.31 126.24 0.874
trimethylcyclohexane
464 1c,2t,4t-trimethylcyclohexane 82.31 126.24 0.874
466 C -olefin 82.33 126.24 0.874
468 2,3,4-trimethylhexane 82.63 128.26 0.888
470 C -olefin 82.73 126.24 0.874
472 3,3,4-trimethylhexane 82.89 128.26 0.888
474 M-Xylene 83.30 106.08 0.827
476 P-Xylene 83.43 106.08 0.827
478 2,3-dimethyheptane 83.57 128.26 0.888
480 3,5-dimethylheptane 83.83 128.26 0.888
482 3,4-dimethylheptane 83.91 128.26 0.888
484 C -olefin 84.08 126.24 0.874
486 3-methyl-3-ethylhexane 84.26 128.26 0.888
488 C -olefin 84.41 126.24 0.874
490 4-ethylheptane 84.52 128.26 0.888
492 4-methyloctane+C -olefin 84.70 128.26 0.888
494 2-methyloctane 84.84 128.26 0.888
496 C -olefin 85.01 126.24 0.874
498 C -paraffin 85.18 128.26 0.888
500 C -olefin 85.36 126.24 0.874
502 3-ethylheptane 85.51 128.26 0.888
504 3-methyloctane 85.69 128.26 0.888
D6729 − 20
TABLE A1.1 Continued
Peak No. NewCompound Name Retention Time Molecular Mass, MWt Theoretical Mass, Rf, (C1)
506 C -paraffin 85.87 126.24 0.874
506 C -paraffin 85.87 128.23 0.888
508 Cis-1,2,4-trimethylcyclohexane 85.91 126.24 0.874
508 C -cyclohexane 85.91 126.24 0.874
510 1,1,2-trimethylcyclohexane 86.05 126.24 0.874
512 O-Xylene 86.27 106.08 0.827
514 C -olefin 86.47 126.24 0.874
516 C -paraffin 86.57 128.26 0.888
518 C -paraffin 86.75 128.26 0.888
520 C -olefin 86.90 126.24 0.874
522 Trans-1-ethyl-4- 87.08 126.24 0.874
methylcyclohexane
524 Cis-1-ethyl-4- 87.23 126.24 0.874
methylcyclohexane
526 C -paraffin 87.49 128.26 0.888
528 1-nonene 87.79 126.24 0.874
530 Isobutylcyclopentane 88.00 126.24 0.874
532 C -paraffin 88.45 128.26 0.888
534 Trans-3-nonene 88.65 126.24 0.874
536 Cis-3-nonene 88.82 126.24 0.874
538 C -paraffin 89.09 128.26 0.888
540 n-nonane 89.24 128.26 0.888
542 C -olefin 89.41 140.27 0.874
544 Trans-2-nonene 89.74 126.24 0.874
544 Trans-2-nonene 89.47 126.24 0.874
546 1-methyl-1-ethylcyclohexane 89.61 126.24 0.874
548 1-methyl-2-propylcyclopentane 89.96 126.24 0.874
550 C -olefin 90.09 140.27 0.874
552 C -paraffin 90.18 142.28 0.887
554 C -paraffin 90.29 142.28 0.887
556 Isopropylbenzene 90.46 118.08 0.832
556 Isopropylbenzene 90.46 120.20 0.832
558 Cis-2-nonene 90.78 126.24 0.874
560 Tert-butylcyclopentane 90.80 126.24 0.874
562 C -olefins 90.88 126.24 0.874
564 Nonene 91.16 126.24 0.874
564 C -Olefin 91.16 126.24 0.874
...



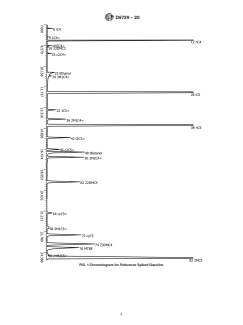



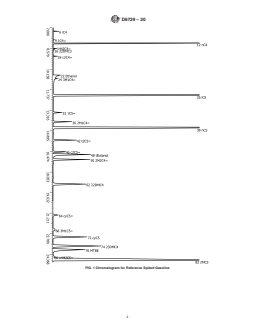
Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...