ASTM E2525-22
(Test Method)Standard Test Method for Evaluation of the Effect of Nanoparticulate Materials on the Formation of Mouse Granulocyte-Macrophage Colonies
Standard Test Method for Evaluation of the Effect of Nanoparticulate Materials on the Formation of Mouse Granulocyte-Macrophage Colonies
SIGNIFICANCE AND USE
5.1 Stem cells of hematopoietic origin are pluripotential and may be particularly sensitive to the effects of stimulation by nanoparticulate materials.
5.2 The effect of particles on macrophage responses has an extensive history and can be assessed by Practice F1903. The test method described here will assess the effect on stem cells which can be progenitor cells to the macrophage line.
SCOPE
1.1 This test method provides a protocol for quantitative analysis of the effect of nanoparticulate materials in physiologic solution (isotonic, pH 7.2 ± 0.2) on granulocyte-macrophage colony-forming units (CFU-GM).
1.2 CFU-GM reflects the number of viable bone marrow cells which differentiate into granulocytes and macrophages. A decrease in CFU-GM count is indicative of a test substance’s toxicity to bone marrow and is commonly used for the identification of drug products with myelosuppressive properties, a form of immunosuppression.
1.3 This test method employs murine bone marrow hematopoietic stem cells which proliferate and differentiate to form discrete cell clusters or colonies which are counted.
1.4 This test method is part of the in vitro preclinical characterization cascade for nanoparticulate materials for systemic administration in medical applications.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2022
- Technical Committee
- E56 - Nanotechnology
- Drafting Committee
- E56.03 - Environment, Health, and Safety
Relations
- Effective Date
- 01-Oct-2018
- Refers
ASTM F1903-10 - Standard Practice for Testing For Biological Responses to Particles <i>In Vitro</i> - Effective Date
- 01-Jun-2010
- Refers
ASTM F1903-98(2003) - Standard Practice for Testing for Biological Responses to Particles In Vitro - Effective Date
- 01-Nov-2003
- Effective Date
- 10-Jun-1998
Overview
ASTM E2525-22: Standard Test Method for Evaluation of the Effect of Nanoparticulate Materials on the Formation of Mouse Granulocyte-Macrophage Colonies provides a comprehensive, quantitative framework for assessing the impact of nanoparticulate materials on bone marrow stem cells. This in vitro test specifically evaluates how these nanoparticles affect granulocyte-macrophage colony-forming units (CFU-GM) derived from murine bone marrow. Decreasing CFU-GM counts can indicate the toxicity of a substance to bone marrow, a critical endpoint in preclinical safety and immunotoxicology assessment, particularly for drug development and nanoparticle evaluation for systemic administration in medical applications.
The procedure outlined in ASTM E2525-22 is a vital component of the preclinical characterization of nanomaterials, helping stakeholders identify potential myelosuppressive effects-where drugs or nanoformulations may suppress the immune system via bone marrow toxicity.
Key Topics
- CFU-GM Assay: Measures the effect of nanoparticulate materials on hematopoietic stem cells’ ability to form granulocyte and macrophage colonies, which are essential for immune system function.
- Nanoparticle Toxicity Evaluation: Detects toxicity or stimulation induced by nanoparticles in comparison with established positive and negative controls.
- Stem Cell Sensitivity: Focuses on pluripotential hematopoietic stem cells, which may be especially sensitive to nanoparticles, making this assay relevant for new medical materials.
- Myelosuppression Assessment: Used to reveal immunosuppression risk, supporting regulatory compliance and product safety.
- In Vitro Preclinical Screening: Allows early-stage screening of materials intended for systemic administration before animal or human studies.
Applications
ASTM E2525-22 is widely applicable across multiple sectors that develop or evaluate nanoparticulate materials, including:
- Pharmaceutical Development: Used by pharmaceutical companies to screen new drugs, especially those formulated with nanoparticulate delivery systems, for their effects on immune cell production and bone marrow health.
- Nanomedicine Research: Enables researchers to determine the safety profile of novel nanomaterials, aiding the translation of nanotechnology-based therapies into clinical use.
- Medical Device Evaluation: Assists medical device manufacturers in documenting the biocompatibility of nanoscale components or coatings for regulatory submissions.
- Toxicology and Safety Laboratories: Provides contract research organizations, government, and academic labs with a standardized approach to assess bone marrow toxicity of new substances or materials.
- Regulatory Compliance: Supports submissions to regulatory agencies by providing robust, internationally-recognized safety data for systemic administration products.
Employing this standard test method streamlines the comparison of different nanoparticulate materials under consistent, reproducible conditions, improving both safety and innovation outcomes.
Related Standards
For a holistic assessment of nanomaterial and particle safety, ASTM E2525-22 should be used alongside the following standards:
- ASTM F1903: Practice for Testing for Cellular Responses to Particles in vitro-focuses on macrophage responses, complementing the stem cell analysis in E2525-22.
- ANSI/AAMI ST72: Bacterial Endotoxins-Test Methodologies, Routine Monitoring, and Alternatives to Batch Testing-addresses sterility and pyrogenicity in biological assays.
- ASTM E56 Series: Standards from ASTM Committee E56 on Nanotechnology, which address various aspects of nanomaterial characterization and safety.
By following ASTM E2525-22, organizations benefit from increased confidence in the biological safety evaluation of nanoparticulate materials, supporting innovation, compliance, and risk management in the fast-evolving field of nanotechnology.
Keywords: ASTM E2525, nanoparticulate materials, CFU-GM, bone marrow toxicity, immunosuppression, myelosuppression, preclinical testing, stem cells, nanomedicine safety, in vitro assay, granulocyte-macrophage colony, pharmaceutical nanotechnology.
Buy Documents
ASTM E2525-22 - Standard Test Method for Evaluation of the Effect of Nanoparticulate Materials on the Formation of Mouse Granulocyte-Macrophage Colonies
Frequently Asked Questions
ASTM E2525-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Evaluation of the Effect of Nanoparticulate Materials on the Formation of Mouse Granulocyte-Macrophage Colonies". This standard covers: SIGNIFICANCE AND USE 5.1 Stem cells of hematopoietic origin are pluripotential and may be particularly sensitive to the effects of stimulation by nanoparticulate materials. 5.2 The effect of particles on macrophage responses has an extensive history and can be assessed by Practice F1903. The test method described here will assess the effect on stem cells which can be progenitor cells to the macrophage line. SCOPE 1.1 This test method provides a protocol for quantitative analysis of the effect of nanoparticulate materials in physiologic solution (isotonic, pH 7.2 ± 0.2) on granulocyte-macrophage colony-forming units (CFU-GM). 1.2 CFU-GM reflects the number of viable bone marrow cells which differentiate into granulocytes and macrophages. A decrease in CFU-GM count is indicative of a test substance’s toxicity to bone marrow and is commonly used for the identification of drug products with myelosuppressive properties, a form of immunosuppression. 1.3 This test method employs murine bone marrow hematopoietic stem cells which proliferate and differentiate to form discrete cell clusters or colonies which are counted. 1.4 This test method is part of the in vitro preclinical characterization cascade for nanoparticulate materials for systemic administration in medical applications. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Stem cells of hematopoietic origin are pluripotential and may be particularly sensitive to the effects of stimulation by nanoparticulate materials. 5.2 The effect of particles on macrophage responses has an extensive history and can be assessed by Practice F1903. The test method described here will assess the effect on stem cells which can be progenitor cells to the macrophage line. SCOPE 1.1 This test method provides a protocol for quantitative analysis of the effect of nanoparticulate materials in physiologic solution (isotonic, pH 7.2 ± 0.2) on granulocyte-macrophage colony-forming units (CFU-GM). 1.2 CFU-GM reflects the number of viable bone marrow cells which differentiate into granulocytes and macrophages. A decrease in CFU-GM count is indicative of a test substance’s toxicity to bone marrow and is commonly used for the identification of drug products with myelosuppressive properties, a form of immunosuppression. 1.3 This test method employs murine bone marrow hematopoietic stem cells which proliferate and differentiate to form discrete cell clusters or colonies which are counted. 1.4 This test method is part of the in vitro preclinical characterization cascade for nanoparticulate materials for systemic administration in medical applications. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2525-22 is classified under the following ICS (International Classification for Standards) categories: 07.100.10 - Medical microbiology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2525-22 has the following relationships with other standards: It is inter standard links to ASTM F1903-18, ASTM F1903-10, ASTM F1903-98(2003), ASTM F1903-98. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2525-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2525 − 22
Standard Test Method for
Evaluation of the Effect of Nanoparticulate Materials on the
Formation of Mouse Granulocyte-Macrophage Colonies
This standard is issued under the fixed designation E2525; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method provides a protocol for quantitative
F1903Practice for Testing for Cellular Responses to Par-
analysis of the effect of nanoparticulate materials in physi-
ticles in vitro
ologic solution (isotonic, pH 7.2 6 0.2) on granulocyte-
2.2 ANSI Standard:
macrophage colony-forming units (CFU-GM).
ANSI/ AAMI ST72 Bacterial Endotoxins—Test
1.2 CFU-GM reflects the number of viable bone marrow
Methodologies, Routine Monitoring, and Alternatives to
cellswhichdifferentiateintogranulocytesandmacrophages.A
Batch Testing
decrease in CFU-GM count is indicative of a test substance’s
3. Terminology
toxicity to bone marrow and is commonly used for the
identification of drug products with myelosuppressive
3.1 Abbreviations:
properties, a form of immunosuppression.
3.1.1 CFU-GM—colony forming unit of granulocyte and
macrophage
1.3 This test method employs murine bone marrow he-
3.1.2 DMSO—dimethyl sulfoxide
matopoietic stem cells which proliferate and differentiate to
3.1.3 DPBS—Dulbecco’s phosphate buffered saline
form discrete cell clusters or colonies which are counted.
3.1.4 FBS—fetal bovine serum
1.4 This test method is part of the in vitro preclinical
3.1.5 IMDM—Iscove’s media
characterization cascade for nanoparticulate materials for sys-
temic administration in medical applications. 3.1.6 LPS—ipopolysaccharide
1.5 The values stated in SI units are to be regarded as
4. Summary of Test Method
standard. No other units of measurement are included in this
4.1 The effect of nanoparticulate materials on the formation
standard.
of granulocyte and macrophage colonies is assessed. Bone
1.6 This standard does not purport to address all of the
marrow cells are obtained from mice and cultured in stimula-
safety concerns, if any, associated with its use. It is the tory media. The number of colony forming units following
responsibility of the user of this standard to establish appro- contactwithnanoparticlesiscountedvisuallyandcomparedto
baseline and positive control. This determines if the nanopar-
priate safety, health, and environmental practices and deter-
ticulate material in physiologic solution is stimulatory or
mine the applicability of regulatory limitations prior to use.
inhibitory to bone marrow stem cells. Aseptic procedures are
1.7 This international standard was developed in accor-
necessary.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
5. Significance and Use
Development of International Standards, Guides and Recom-
5.1 Stemcellsofhematopoieticoriginarepluripotentialand
mendations issued by the World Trade Organization Technical
may be particularly sensitive to the effects of stimulation by
Barriers to Trade (TBT) Committee.
nanoparticulate materials.
1 2
This test method is under the jurisdiction of ASTM Committee E56 on For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Nanotechnology and is the direct responsibility of Subcommittee E56.03 on contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Environment, Health, and Safety. Standards volume information, refer to the standard’s Document Summary page on
CurrenteditionapprovedJuly1,2022.PublishedJuly2022.Originallyapproved the ASTM website.
in 2008. Last previous edition approved in 2013 as E2525 – 08(2013), which was Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
withdrawn in April 2022 and reinstated July 2022. DOI: 10.1520/E2525-22. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2525 − 22
5.2 The effect of particles on macrophage responses has an 6.3.14 Biohazard safety cabinet approved for level II han-
extensive history and can be assessed by Practice F1903. The dling of biological material.
test method described here will assess the effect on stem cells
6.3.15 Inverted microscope.
which can be progenitor cells to the macrophage line.
6.3.16 Vortex.
6.3.17 Hemocytometer.
6. Reagents and Materials
6.4 Animals—MiceofthestrainC56BL/6,malesorfemales
6.1 Purity of Reagents—Reagent grade chemicals shall be
8weeks to 12 weeks old, are used. Use of the pooled cells
used in all tests. Unless otherwise indicated, it is intended that
derived from at least two (2) animals is highly recommended.
all reagents conform to the specifications of the Committee on
NOTE 2—The source of the equipment, where given, is shown for
Analytical Reagents of theAmerican Chemical Society where
information purposes only to aid laboratories initiating this procedure.
such specifications are available. Other grades may be used,
Equivalent equipment from other suppliers may be used.
provided it is first ascertained that the reagent is of sufficiently
high purity to permit its use without lessening the accuracy of
7. Procedure
the determination.
7.1 Aseptic Precautions are required.
6.2 Reagents and Supplies (see Note 1):
7.2 Reagent and Control Preparation:
6.2.1 MethoCult medium, (optimized for growth and enu-
7.2.1 MethoCult Medium—The MethoCult medium is sup-
meration of granulocyte-macrophage progenitor cells) Stem-
plied in 100mL size batches. It is recommended by the
Cell Technologies Inc cat.# 03534.
manufacturer that the medium be thawed at room temperature
6.2.2 FBS prescreened for hematopoietic stem cells, Stem-
or in a refrigerator overnight, vortexed to mix the ingredients,
Cell Technologies Inc cat.# 06200.
then left at a room temperature for approximately 5 min to
6.2.3 IMDM with 2% FBS, StemCell Technologies Inc
allowairbubblestodissipate.Ifusinganothersourceofmedia,
cat.# 07700.
follow the instructions indicated by the supplier. Use a 16-
6.2.4 Sterile distilled water.
gauge blunt-end needle to dispense 3mL aliquots of the
6.2.5 Cisplatin, [cis-diammineplatinum(II) dichloride],
MethoCult medium into sterile 15mL tubes. Store the ali-
(positive control) Sigma-Aldrich cat# P4394.
2+ 2+
quoted medium at a nominal temperature of –20°C. Before the
6.2.6 Sterile Ca /MG -free DPBS, (negative control)
test,thawtherequirednumberoftubesatroomtemperaturefor
Sigma-Aldrich cat.# D8537.
approximately 20 min and then keep on ice prior to use.
NOTE 1—The source of the reagents is shown for information purposes
Repeated freezing and thawing should be avoided.
onlytoaidlaboratoriesinitiatingthisprocedure.Equivalentreagentsfrom
7.2.2 50 mmol/L Cisplatin (Positive Control)—Cisplatin is
other suppliers may be used.
supplied in a lyophilized form. Reconstitute the lyophilized
6.3 Equipment (see Note 2)—Aseptic procedures are neces-
powder by adding the appropriate amount of DMSO to make a
sary and care should be used in acquiring sterile equipment as
stock solution with nominal concentration of 50 mmol/L.
needed.
Prepare small aliquots and store at a nominal temperature of
6.3.1 Pipettes covering the range of 0.05mL to 10 mL.
–80 °C. Prior to use in the assay, thaw an aliquot of the stock
6.3.2 35-mmculturedishesprescreenedtosupportstemcell
solution at room temperature and dilute in IMDM supple-
growth and differentiation, StemCell Technologies Inc cat.#
mented with 2% FBS to bring the Cisplatin concentration to 2
27100.
mmol/L. One hundred fifty (150) µL of this intermediate
6.3.3 Blunt-end 16-gauge needles, StemCell Technologies
solution is then added to 3 mL of culture medium. Final
Inc cat.# 03534.
concentrationofCisplatininthepositivecontrolsampleis100
6.3.4 100-mm Petri dishes.
µmol/L.
6.3.5 Plastic beakers.
7.3 Preparation of Study Samples:
6.3.6 Polypropylene tubes 50mL and 15mL.
7.3.1 This assay requires 1200 µL of nanoparticles, 150 µL
6.3.7 Centrifuge.
samples in duplicate for each of four (4) concentrations. The
6.3.8 Refrigerator, 2°C to 8°C.
nanoparticles subjected to the biological test environment
6.3.9 Freezer, –20°C.
should have been characterized as appropriate to allow ad-
6.3.10 Cell culture incubator with 5% CO and 95%
equate data interpretation and to help provide information to
humidity.
predict biological responses. For example, lot-to-lot variations
6.3.11 CO euthanasia box, or appropriate equipment ap-
inparticlesizeandsurfacecharacteristicsoftheparticlescould
proved by institution.
result in different assay results. For this assay, the particles
6.3.12 Scissors for tissue dissection.
shall be provided in physiologic solution (isotonic with pH
6.3.13 Forceps.
7.2 60.2) and this solution shall be defined. The preparation
shall be sterile and the level of LPS provided or determined by
the testing laboratory.ANSI/AAMI ST72 may be helpful. The
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
number of particles/mL and mg/mL shall be indicated.
listed by the American Chemical Society, see Analar Standards for Laboratory
7.3.2 The test sample shall be used at the highest concen-
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
tration possible
...



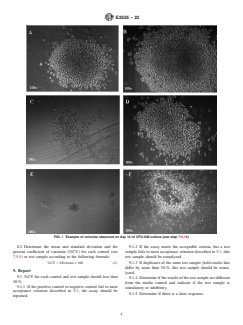
Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...