ASTM F1781-21
(Specification)Standard Specification for Elastomeric Flexible Hinge Finger Total Joint Implants
Standard Specification for Elastomeric Flexible Hinge Finger Total Joint Implants
ABSTRACT
This specification covers the biocompatibility and other performance requirements, and associated test methods for elastomeric flexible hinge finger total joint implants, used with and without metal grommets in the reconstruction of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints. This specification excludes those implants that do not have an across-the-joint elastomeric linkage, and is limited to implants made from one material in a single one-step molding procedure.
SCOPE
1.1 This specification covers elastomeric flexible hinge finger total joint implants, used with and without metal grommets, in the reconstruction of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints.
1.2 This specification excludes those implants that do not have an across-the-joint elastomeric linkage. This specification is limited to implants made from one material in a single, one-step molding procedure.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2021
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 01-Apr-2023
- Effective Date
- 15-Aug-2019
- Effective Date
- 01-Dec-2018
- Refers
ASTM F983-86(2018) - Standard Practice for Permanent Marking of Orthopaedic Implant Components - Effective Date
- 01-Feb-2018
- Effective Date
- 01-Mar-2017
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Apr-2014
- Refers
ASTM F601-13 - Standard Practice for Fluorescent Penetrant Inspection of Metallic Surgical Implants - Effective Date
- 01-Dec-2013
- Refers
ASTM F983-86(2013) - Standard Practice for Permanent Marking of Orthopaedic Implant Components - Effective Date
- 01-Oct-2013
- Effective Date
- 01-Jun-2013
- Effective Date
- 01-Jun-2013
- Effective Date
- 01-Apr-2013
- Refers
ASTM F86-12a - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 01-Dec-2012
- Refers
ASTM F86-12 - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 15-May-2012
- Effective Date
- 01-Dec-2011
Overview
ASTM F1781-21 is the internationally recognized standard specification for elastomeric flexible hinge finger total joint implants. Developed by ASTM International, this document establishes performance and biocompatibility requirements as well as relevant testing protocols for such implants, focusing on devices used in the reconstruction of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints. The standard applies to implants made from a single material using a one-step molding process, with or without metal grommets, and featuring an across-the-joint elastomeric linkage.
Key Topics
- Scope: Covers elastomeric finger joint implants specifically for MCP and PIP joint reconstruction.
- Material Requirements: Implants must be fabricated from strong, durable, and biocompatible elastomeric materials meeting established ASTM and ISO guidance.
- Manufacturing Practices: Devices must be made in compliance with recognized manufacturing standards, such as Good Manufacturing Practices (21 CFR 820).
- Testing and Evaluation: Performance is verified through mechanical property tests, fatigue testing under both high and low cycle conditions, and biocompatibility assessments.
- Performance Criteria:
- Fatigue resistance (evaluated through multi-million cycle mechanical simulations)
- Material hardness and tensile properties
- Proper range of motion and stability prior to implantation
- Labeling and Packaging: All critical implant dimensions, material specifications, and performance properties must be clearly stated in product labeling and documentation.
Applications
ASTM F1781-21 is essential for a range of stakeholders involved in the design, production, and clinical application of finger joint implants:
- Medical Device Manufacturers: Ensures production of implants that meet rigorous standards for safety, durability, and mechanical performance.
- Orthopedic Surgeons: Facilitates selection of implants with documented material properties and performance characteristics, supporting optimal outcomes in MCP and PIP joint reconstruction.
- Regulatory Affairs and Quality Departments: Provides a framework for demonstrating compliance with international regulatory requirements for orthopedic implants, including documented procedures for testing, labeling, and package marking.
- Testing Laboratories: Defines required mechanical and biocompatibility test methods for evaluating new or existing finger joint implant designs.
Related Standards
To fully address all requirements and best practices for elastomeric finger joint implants, ASTM F1781-21 references and is complemented by several related standards:
- ASTM D412, D624, D813, D1052, D2240: Test methods for evaluating mechanical and physical properties of elastomeric materials.
- ASTM F67: Specification for unalloyed titanium used in surgical implant applications (relevant for metal grommets).
- ASTM F86, F983: Practices for marking and surface preparation of metallic surgical implants.
- ASTM F2038, F2042: Guides for silicone elastomers, gels, and foams in medical applications.
- ASTM F748, F981: Guidance on compatibility and biological safety of biomaterials.
- ASTM F2503, F2943: Practices for labeling and presentation of information for medical devices, including MRI safety.
- ISO 10993-1: International standard for the biological evaluation of medical devices.
- 21 CFR 820, 21 CFR 888.6: US regulations on medical device quality systems and implant constraint classification.
Practical Value
ASTM F1781-21 enables reliable, long-term function and patient safety by:
- Defining clear material and manufacturing criteria to minimize risk of implant failure.
- Requiring comprehensive mechanical and biocompatibility testing to assure clinical performance.
- Providing standardized test protocols and labeling practices to support regulatory compliance.
- Promoting global harmonization in the evaluation and use of elastomeric finger joint implants for MCP and PIP joint reconstruction.
For professionals in the orthopedic device industry, this standard is a critical resource for ensuring the quality and safety of flexible hinge finger joint prostheses.
Buy Documents
ASTM F1781-21 - Standard Specification for Elastomeric Flexible Hinge Finger Total Joint Implants
REDLINE ASTM F1781-21 - Standard Specification for Elastomeric Flexible Hinge Finger Total Joint Implants
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1781-21 is a technical specification published by ASTM International. Its full title is "Standard Specification for Elastomeric Flexible Hinge Finger Total Joint Implants". This standard covers: ABSTRACT This specification covers the biocompatibility and other performance requirements, and associated test methods for elastomeric flexible hinge finger total joint implants, used with and without metal grommets in the reconstruction of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints. This specification excludes those implants that do not have an across-the-joint elastomeric linkage, and is limited to implants made from one material in a single one-step molding procedure. SCOPE 1.1 This specification covers elastomeric flexible hinge finger total joint implants, used with and without metal grommets, in the reconstruction of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints. 1.2 This specification excludes those implants that do not have an across-the-joint elastomeric linkage. This specification is limited to implants made from one material in a single, one-step molding procedure. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This specification covers the biocompatibility and other performance requirements, and associated test methods for elastomeric flexible hinge finger total joint implants, used with and without metal grommets in the reconstruction of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints. This specification excludes those implants that do not have an across-the-joint elastomeric linkage, and is limited to implants made from one material in a single one-step molding procedure. SCOPE 1.1 This specification covers elastomeric flexible hinge finger total joint implants, used with and without metal grommets, in the reconstruction of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints. 1.2 This specification excludes those implants that do not have an across-the-joint elastomeric linkage. This specification is limited to implants made from one material in a single, one-step molding procedure. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F1781-21 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1781-21 has the following relationships with other standards: It is inter standard links to ASTM F2503-23e1, ASTM F2943-14(2019), ASTM F2042-18, ASTM F983-86(2018), ASTM F67-13(2017), ASTM F748-16, ASTM F2943-14, ASTM F601-13, ASTM F983-86(2013), ASTM F67-13, ASTM F2503-13, ASTM F2943-13, ASTM F86-12a, ASTM F86-12, ASTM F2038-00(2011). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1781-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F1781 −21
Standard Specification for
Elastomeric Flexible Hinge Finger Total Joint Implants
This standard is issued under the fixed designation F1781; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope F86 Practice for Surface Preparation and Marking of Metal-
lic Surgical Implants
1.1 This specification covers elastomeric flexible hinge
F601 Practice for Fluorescent Penetrant Inspection of Me-
finger total joint implants, used with and without metal
tallic Surgical Implants
grommets, in the reconstruction of the metacarpophalangeal
F748 PracticeforSelectingGenericBiologicalTestMethods
(MCP) and proximal interphalangeal (PIP) joints.
for Materials and Devices
1.2 This specification excludes those implants that do not
F981 Practice for Assessment of Compatibility of Biomate-
have an across-the-joint elastomeric linkage.This specification
rials for Surgical Implants with Respect to Effect of
is limited to implants made from one material in a single,
Materials on Muscle and Insertion into Bone
one-step molding procedure.
F983 Practice for Permanent Marking of Orthopaedic Im-
1.3 The values stated in SI units are to be regarded as plant Components
F2038 GuideforSiliconeElastomers,Gels,andFoamsUsed
standard. No other units of measurement are included in this
standard. in Medical Applications Part I—Formulations and Un-
cured Materials
1.4 This international standard was developed in accor-
F2042 GuideforSiliconeElastomers,Gels,andFoamsUsed
dance with internationally recognized principles on standard-
in Medical Applications Part II—Crosslinking and Fabri-
ization established in the Decision on Principles for the
cation
Development of International Standards, Guides and Recom-
F2503 Practice for Marking Medical Devices and Other
mendations issued by the World Trade Organization Technical
Items for Safety in the Magnetic Resonance Environment
Barriers to Trade (TBT) Committee.
F2943 Guide for Presentation of End User Labeling Infor-
mation for Musculoskeletal Implants
2. Referenced Documents
2.2 Government Standards:
2.1 ASTM Standards:
21 CFR 820 Quality System Regulation
D412 Test Methods forVulcanized Rubber andThermoplas-
21 CFR 888.6 Degree of Constraint
tic Elastomers—Tension
MIL STD 177A Rubber Products, Terms for Visible De-
D624 Test Method for Tear Strength of Conventional Vul-
fects
canized Rubber and Thermoplastic Elastomers
2.3 ISO Standard:
D813 TestMethodforRubberDeterioration—CrackGrowth
ISO 10993-1 Biological Evaluation of Medical Devices—
D1052 Test Method for Measuring Rubber Deterioration—
Part 1: Evaluation and testing within a risk management
Cut Growth Using Ross Flexing Apparatus
process
D2240 Test Method for Rubber Property—Durometer Hard-
ness
3. Significance and Use
F67 Specification for Unalloyed Titanium, for Surgical Im-
3.1 The prostheses described in this specification are in-
plant Applications (UNS R50250, UNS R50400, UNS
tended for use in the proximal interphalangeal (PIP) and
R50550, UNS R50700)
metacarpophalangeal (MCP) joints.
4. Classification
This specification is under the jurisdiction of ASTM Committee F04 on
4.1 Constrained—A constrained joint prosthesis is used for
Medical and Surgical Materials and Devices and is the direct responsibility of
Subcommittee F04.22 on Arthroplasty.
joint replacement and prevents dislocation of the prosthesis in
Current edition approved Sept. 1, 2021. Published September 2021. Originally
approved in 1997. Last previous edition approved in 2015 as F1781 – 15. DOI:
10.1520/F1781-21. AvailablefromU.S.GovernmentPrintingOfficeSuperintendentofDocuments,
For referenced ASTM standards, visit the ASTM website, www.astm.org, or 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM www.access.gpo.gov.
Standards volume information, refer to the standard’s Document Summary page on Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1781 − 21
more than one anatomic plane and consists of either a single, hinge have been reported (1-3). The fatigue characteristics of
flexible, across-the-joint component or more than one compo- the implant shall be evaluated. Testing shall evaluate the
nent linked together or affined (21 CFR 888.6). resistance of the implant to failure under high cycle fatigue
conditions (that is, when cycled through the intended range of
motion) as well as under low cycle fatigue conditions (that is,
5. Materials and Manufacture
when subjected to pinch and grip loads, such as key pinch or
5.1 Proper material selection is necessary, but insufficient to
jar opening). Testing shall be performed at 37 62°Cina
ensuresuitablefunctioningofadevice.Alldevicesconforming
physiological solution (for example, saline, serum). Frequency
to this specification shall be fabricated from materials with
of fatigue testing shall be no greater than 3 Hz (see X1.2.5).
adequate mechanical strength, durability, and biocompatibility.
6.2.1 Testingshallbeconductedforaminimumof4million
All elastomeric components shall conform to Guides F2038
cycles under high cycle fatigue conditions and a minimum of
and F2042.
1330 cycles under low cycle fatigue conditions (4, 5) (see
5.2 Test and evaluation parameters that could be considered
X1.2.6) or until failure. Mode of failure shall be reported for
for the elastomeric implant materials are Test Methods D813,
each sample tested.
D1052, D2240, D412, and D624. Before implants can be
6.2.2 Justification shall be provided for the following test
manufactured from other elastomeric materials, manufacturers
parameters:
shall comply with 5.3.
6.2.2.1 Preconditioning of samples prior to testing (such as,
samples should be tested in a maximum shelf-life aging
5.3 Biocompatibility—Devices made from materials with
condition;testingofmaximumshelf-lifeagedsamplesmaynot
limited or no history of successful use for orthopedic implant
be necessary if justification can be provided to ensure the
applications shall be determined to exhibit acceptable biologi-
material properties will not be affected by shelf-aging).
cal response when tested in accordance with Practice F748 or
6.2.2.2 Test solution.
F981 or ISO 10993-1. While no known surgical implant
6.2.2.3 Frequency.
material has ever been shown to be completely free of adverse
6.2.2.4 Load during high cycle and low cycle fatigue
reactions in the human body, long-term clinical experience has
conditions.
shown an acceptable level of biological response can be
6.2.2.5 Kinematics (that is, flexion/extension for high cycle
expected if materials meeting the specification and guidelines
in 5.1 and 5.4 are used. However, the specification and conditions and kinematics selected for low cycle conditions).
6.2.2.6 Number of samples.
guidelines listed in 5.1 and 5.4 cover raw materials and not
finished medical devices, where the design and fabrication 6.2.2.7 Choice of potting medium, if applicable (given that
the elastomeric material does not allow osseointegration, the
process of the device can impact biological response. Hence,
for a device made from material(s) meeting the specification pottingmediumshallbeselectedsuchthatthetestcanreplicate
the pistoning effect of the implant’s stems into the intramed-
and guidelines in 5.1 and 5.4, its biocompatibility shall be
verified in accordance with Practice F748 or F981 or ISO ullary canal).
6.2.3 Examples of fatigue test methods are included in the
10993-1, unless justification can be provided for why design
published literature for reference (4-7).
andprocessingwillnotimpactthebiocompatibilityofthefinal,
6.2.4 Justification of fatigue performance may be based on
sterilized device.
comparison to physiological loading parameters expected to be
5.4 Titanium used as a material of construction for metal
encountered throughout the lifetime of the implant and/or
grommets shall conform to Specification F67. Metal grommets
comparisontoperformanceofalegallymarketeddeviceandin
shall match the shape of the implant and not interfere with the
accordance with the requirements of the regulatory regime in
flexible hinge implant function.
which the device is to be marketed.
5.5 When appropriate for metallic grommets, fluorescent
6.3 Range of Motion of the Device Before Implantation—
penetrant inspection shall be performed in accordance with
The implant shall be evaluated to determine the maximum
Practice F601.
flexion and extension possible before subluxation occurs or the
5.6 Design and manufacture shall follow 21 CFR 820.
motion is arrested by the implant (elastomer-to-elastomer
contact within the hinge).These results shall be reported in the
6. Performance Requirements
product labeling.
6.1 Guidelines for in-vitro Laboratory Testing—No ASTM
6.4 Durometer—The hardness of elastomeric material used
standards for testing finger implants have been developed.
in the implant shall be measured according to Test Method
Laboratory testing that simulates the conditions of use by a
D2240.
joint function simulator is desirable to compare materials and
6.5 The mechanical properties (such as tensile strength,
designs and to provide an indication of clinical performance.
percentage elongation, modulus, and tear strength) of the
Implant testing shall be done in keeping with the intended
elastomeric materials used in components shall be determined
function of the implant. Implants intended to fully or partially
according to Test Methods D412 and D624.
stabilize a joint shall be subjected to loads and kinematics
expected to be experienced during activities of daily living.
6.2 FatigueTesting—Clinical failure modes such as fracture
The boldface numbers in parentheses refer to a list of references at the end of
at the hinge and fracture at the junction of the distal stem and this standard.
F1781 − 21
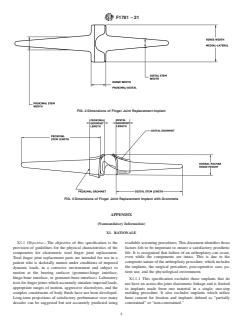
7. Dimensions 8.3 Consider Practice F2503 to identify potential hazards
produced by interactions between the device and the MR
7.1 The following dimensions of the implant shall be
environment,andfortermsthatmaybeusedtolabelthedevice
reported in labeling (see Figs. 1 and 2):
for safety in the MR environment.
7.1.1 Distal stem length,
7.1.2 Proximal stem length,
9. Labeling and Packaging
7.1.3 Hinge width in medial/lateral plane,
7.1.4 Hinge height in dorsal/palmar plane,
9.1 The maximum range of motion values as determined by
7.1.5 Distal stem width, 6.3 shall be included in the product labeling. The minimum
7.1.6 Proximal stem width,
limits for the mechanical properties of the elastomeric materi-
7.1.7 Distal-proximal hinge width, and al(s) used in components shall be included in the product
7.1.8 Angulation of the distal stem ax
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F1781 − 15 F1781 − 21
Standard Specification for
Elastomeric Flexible Hinge Finger Total Joint Implants
This standard is issued under the fixed designation F1781; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This specification covers elastomeric flexible hinge finger total joint implants, used with and without metal grommets, in the
reconstruction of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints.
1.2 This specification excludes those implants that do not have an across-the-joint elastomeric linkage. TheThis specification is
limited to implants made from one material in a single, one-step molding procedure.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D412 Test Methods for Vulcanized Rubber and Thermoplastic Elastomers—Tension
D624 Test Method for Tear Strength of Conventional Vulcanized Rubber and Thermoplastic Elastomers
D813 Test Method for Rubber Deterioration—Crack Growth
D1052 Test Method for Measuring Rubber Deterioration—Cut Growth Using Ross Flexing Apparatus
D2240 Test Method for Rubber Property—Durometer Hardness
F67 Specification for Unalloyed Titanium, for Surgical Implant Applications (UNS R50250, UNS R50400, UNS R50550, UNS
R50700)
F86 Practice for Surface Preparation and Marking of Metallic Surgical Implants
F601 Practice for Fluorescent Penetrant Inspection of Metallic Surgical Implants
F748 Practice for Selecting Generic Biological Test Methods for Materials and Devices
F981 Practice for Assessment of Compatibility of Biomaterials for Surgical Implants with Respect to Effect of Materials on
Muscle and Insertion into Bone
F983 Practice for Permanent Marking of Orthopaedic Implant Components
F2038 Guide for Silicone Elastomers, Gels, and Foams Used in Medical Applications Part I—Formulations and Uncured
Materials
F2042 Guide for Silicone Elastomers, Gels, and Foams Used in Medical Applications Part II—Crosslinking and Fabrication
F2503 Practice for Marking Medical Devices and Other Items for Safety in the Magnetic Resonance Environment
This specification is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.22 on Arthroplasty.
Current edition approved Oct. 1, 2015Sept. 1, 2021. Published December 2015September 2021. Originally approved in 1997. Last previous edition approved in 20092015
as F1781 – 03 (2009).F1781 – 15. DOI: 10.1520/F1781-15.10.1520/F1781-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1781 − 21
F2943 Guide for Presentation of End User Labeling Information for Musculoskeletal Implants
2.2 Government Standards:
21 CFR 820 Good Manufacturing Practices for Medical DevicesQuality System Regulation
21 CFR 888.6 Degree of Constraint
MIL STD 177A Rubber Products, Terms for Visible Defects
2.3 ISO Standard:
ISO 10993-1 Biological EvaluationsEvaluation of Medical Devices — Part Devices—Part 1: Evaluation and testing within a risk
management process
3. Significance and Use
3.1 The prostheses described in this specification are intended for use in the proximal interphalangeal (PIP) and metacarpopha-
langeal (MCP) joints.
4. Classification
4.1 Constrained—A constrained joint prosthesis is used for joint replacement and prevents dislocation of the prosthesis in more
than one anatomicalanatomic plane and consists of either a single, flexible, across-the-joint component,component or more than
one component linked together or affined.affined (21 CFR 888.6).
5. Materials and Manufacture
5.1 Proper material selection is necessary, but insufficient to ensure suitable functioning of a device. All devices conforming to
this specification shall be fabricated from materials with adequate mechanical strength, durability, and biocompatibility. All
elastomeric components shall conform to Guides F2038 and F2042.
5.2 Test and evaluation parameters that could be considered for the elastomeric implant materials are Test Methods D813, D1052,
D2240, D412, and D624. Before implants can be manufactured from other elastomeric materials, manufacturers shall comply with
5.3.
5.3 Biocompatibility—Flexible hinge implants shall be manufactured from Devices made from materials with limited or no history
of successful use for orthopedic implant applications shall be determined to exhibit acceptable biological response when tested in
accordance with Practice F748 or F981the materials or ISO 10993-1. While no known surgical implant material has ever been
shown to be completely free of adverse reactions in the human body, long-term clinical experience has shown an acceptable level
of biological response can be expected if materials meeting the specification and guidelines in 5.1 and 5.4 are used. However, the
specification and guidelines listed in 5.25.1 and 5.35.4. Before implants can be manufactured from other materials, their cover raw
materials and not finished medical devices, where the design and fabrication process of the device can impact biological response.
Hence, for a device made from material(s) meeting the specification and guidelines in 5.1 and 5.4, its biocompatibility shall be
demonstrated by producing an acceptable response after testing verified in accordance with PracticesPractice F748 or F981ISO
10993-1. or ISO 10993-1, unless justification can be provided for why design and processing will not impact the biocompatibility
of the final, sterilized device.
5.4 Titanium used as a material of construction for metal grommets shall conform to Specification F67. Metal grommets shall
match the shape of the implant and not interfere with the flexible hinge implant function.
5.5 When appropriate for metallic grommets, fluorescent penetrant inspection shall be performed in accordance with Practice
F601.
5.6 Design and manufacture shall follow 21 CFR 820.
6. Performance Requirements
6.1 Guidelines for in-vitro Laboratory Testing—No ASTM standards for testing finger implants have been developed. Laboratory
Available from U.S. Government Printing Office Superintendent of Documents, 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
www.access.gpo.gov.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
F1781 − 21
testing that simulates the conditions of use by a joint function simulator is desirable to compare materials and designs and to
provide an indication of clinical performance. Implant testing shall be done in keeping with the intended function of the implant.
Implants intended to fully or partially stabilize a joint shall be subjected to loads and kinematics expected to be experienced during
activities of daily living.
6.2 Fatigue Testing—Clinical failure modes such as fracture at the hinge and fracture at the junction of the distal stem and hinge
have been reported (1-3). The fatigue characteristics of material from which the elastomeric components are fabricated shall be
evaluated according to Test Methodthe implant shall be evaluated. Testing shall evaluate the resistance of the implant to failure
under D813. Any test should be designed to measure fatigue rate (for example, crack growth length) as a function of a million(s)
cycles.high cycle fatigue conditions (that is, when cycled through the intended range of motion) as well as under low cycle fatigue
conditions (that is, when subjected to pinch and grip loads, such as key pinch or jar opening). Testing shall be performed at 37
6 2 °C in a physiological solution (for example, saline, serum). Frequency of fatigue testing shall be no greater than 3 Hz (see
X1.2.5).
6.2.1 Testing shall be conducted for a minimum of 4 million cycles under high cycle fatigue conditions and a minimum of 1330
cycles under low cycle fatigue conditions (4, 5) (see X1.2.6) or until failure. Mode of failure shall be reported for each sample
tested.
6.2.2 Justification shall be provided for the following test parameters:
6.2.2.1 Preconditioning of samples prior to testing (such as, samples should be tested in a maximum shelf-life aging condition;
testing of maximum shelf-life aged samples may not be necessary if justification can be provided to ensure the material properties
will not be affected by shelf-aging).
6.2.2.2 Test solution.
6.2.2.3 Frequency.
6.2.2.4 Load during high cycle and low cycle fatigue conditions.
6.2.2.5 Kinematics (that is, flexion/extension for high cycle conditions and kinematics selected for low cycle conditions).
6.2.2.6 Number of samples.
6.2.2.7 Choice of potting medium, if applicable (given that the elastomeric material does not allow osseointegration, the potting
medium shall be selected such that the test can replicate the pistoning effect of the implant’s stems into the intramedullary canal).
6.2.3 Examples of fatigue test methods are included in the published literature for reference (4-7).
6.2.4 Justification of fatigue performance may be based on comparison to physiological loading parameters expected to be
encountered throughout the lifetime of the implant and/or comparison to performance of a legally marketed device and in
accordance with the requirements of the regulatory regime in which the device is to be marketed.
6.3 Range of Motion of the Device Before Implantation—The implant shall be evaluated to determine the maximum flexion and
extension possible before subluxation occurs or the motion is arrested by the implant (elastomer-to-elastomer contact within the
hinge). These results shall be reported in the product labeling.
6.3 Guidelines for in vitro Laboratory Testing—No ASTM standards for testing finger implants have been developed. Laboratory
testing that simulates the conditions of use, by a joint function simulator, is desirable to compare materials and designs and to
provide an indication of clinical performance. Implant testing shall be done in keeping with the implant’s intended function.
Implants intended to partially stabilize or stabilize a joint shall be subjected to the maximum destabilizing force or motion, or both,
anticipated in clinical application during flexural testing.
6.4 Durometer—The hardness of elastomeric components material used in the implant shall be measured according to Test Method
D2240.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
F1781 − 21
6.5 The mechanical properties (such as tensile strength, percentage elongation, modulus, and tear strength) of the elastomeric
materials used in components shall be determined according to Test Methods D412 and D624.
7. Dimensions
7.1 The following dimensions of finger and joint replacement components the implant shall be reported in labeling (see Figs. 1
and 2):
7.1.1 Distal stem length,
7.1.2 Proximal stem length,
7.1.3 Hinge width in medial/lateral plane,
7.1.4 Hinge height in dorsal/palmar plane,
7.1.5 Distal stem width,
7.1.6 Proximal stem width, and
7.1.7 Distal-proximal hinge width.width, and
7.1.8 Angulation of the distal stem axis with respect to the proximal stem axis.
7.2 The following additional dimensions of fingerthe implant with metal grommets shall be reported in labeling (see Fig. 3):
7.2.1 Distal stemgrommet length,
7.2.2 Proximal stemgrommet length,
7.2.3 Distal grommet length,medial/lateral width,
7.2.4 Proximal grommet length, medial/lateral width, and
7.2.5 Hinge height in dorsal/palmar plane.Minimum grommet thickness.
FIG. 1 Dimensions of Finger and Joint Replacement ComponentsImplant
F1781 − 21
FIG. 2 Dimensions
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...