ASTM F2381-19
(Test Method)Standard Test Method for Evaluating Trans-Vinylene Yield in Irradiated Ultra-High Molecular Weight Polyethylene Fabricated Forms Intended for Surgical Implants by Infrared Spectroscopy
Standard Test Method for Evaluating Trans-Vinylene Yield in Irradiated Ultra-High Molecular Weight Polyethylene Fabricated Forms Intended for Surgical Implants by Infrared Spectroscopy
SIGNIFICANCE AND USE
4.1 Published literature shows that the yield of radiolytic reactions that occur during radiation treatment increases with radiation dose level. Measurement of the products of these reactions can be used as an internal dosimeter.
4.2 Trans-vinylene unsaturations are formed during ionization treatment by abstraction of a hydrogen molecule, and to a lesser extent by the recombination of two adjacent alkyl free radicals that reside on the same chain.
4.3 Previous work generated calibration curves of trans-vinylene absorption area as a function of absorbed radiation dose, yielding a linear relationship for both gamma- and electron beam-irradiated polyethylene.
4.4 This data can be used to determine received dose as a function of position, assuming a calibration curve (TVI versus radiation dose level) is known for the particular material and radiation conditions used, and can be used to determine uniformity of dose level in irradiated polyethylene.
SCOPE
1.1 This test method describes the measurement of the number of trans-vinylene groups in ultra-high molecular weight polyethylene (UHMWPE) intended for use in medical implants. The material is analyzed by infrared spectroscopy.
1.2 This test method is based on Guide F2102.
1.3 The applicability of the infrared method has been demonstrated in other literature reports. This particular method, using the intensity (area) of the C-H absorption centered at 1370 cm-1 to normalize for the sample’s thickness, will be validated by an Interlaboratory Study (ILS) conducted according to Practice E691.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2019
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.15 - Material Test Methods
Relations
- Effective Date
- 01-Oct-2019
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Nov-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Mar-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-May-2006
- Effective Date
- 01-May-2006
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Feb-2004
- Effective Date
- 10-May-2001
- Effective Date
- 10-May-2001
- Effective Date
- 10-Oct-1999
- Effective Date
- 10-May-1999
Overview
ASTM F2381-19, titled "Standard Test Method for Evaluating Trans-Vinylene Yield in Irradiated Ultra-High Molecular Weight Polyethylene Fabricated Forms Intended for Surgical Implants by Infrared Spectroscopy," provides a reliable protocol for measuring the number of trans-vinylene groups in ultra-high molecular weight polyethylene (UHMWPE) used in medical implants. Utilizing infrared spectroscopy, this test method supports the evaluation of material modifications induced by ionizing radiation, which plays a critical role in crosslinking UHMWPE for enhanced implant performance.
Accurate assessment of trans-vinylene yield is crucial for ensuring the quality, safety, and consistency of polyethylene components used in orthopedic and other surgical implants. This standard aligns with international standardization principles and leverages established calibration techniques to enable reproducible, objective measurement and quality assurance.
Key Topics
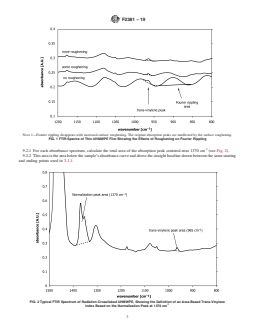
- Trans-Vinylene Index (TVI): The method defines the trans-vinylene index as the ratio of the absorption peak area centered near 965 cm⁻¹ to that at 1370 cm⁻¹, as measured by FTIR spectroscopy.
- Infrared Spectroscopy: The procedure relies on Fourier Transform Infrared (FTIR) spectrometers to detect specific absorption peaks indicating the presence of trans-vinylene groups.
- Radiation Dose Assessment: Calibration curves relate TVI to absorbed radiation dose, supporting both gamma and electron beam irradiation types.
- Sample Preparation: Detailed guidance is given on preparing thin sections of UHMWPE, emphasizing slice orientation and minimizing spectral artifacts.
- Internal Dosimetry: Measurement of trans-vinylene groups functions as an internal dosimeter, providing insight into the effective radiation dose received by the material.
- Uniformity of Treatment: Spatial TVI profiles allow users to assess the uniformity of irradiation and crosslinking within polyethylene forms.
- Traceable Reporting: The standard specifies requirements for detailed documentation of sample, apparatus, and data analysis parameters for quality and traceability.
Applications
Adhering to ASTM F2381-19 is essential for:
- Orthopedic and Surgical Implant Manufacturing: Providing a validated basis for assessing radiation-induced modifications in UHMWPE used as a bearing material in artificial joints, spinal implants, and other surgical devices.
- Quality Control: Offering robust, repeatable methodologies to confirm batch uniformity, validate irradiation procedures, and fulfill regulatory requirements.
- Research and Development: Assisting in the optimization of crosslinking processes to improve wear resistance, longevity, and biocompatibility of medical-grade polyethylene.
- Dose Mapping: Enabling detailed mapping of radiation dose distribution within complex component geometries, thereby ensuring the entire device meets design specifications for material stability.
- Comparative Material Studies: Allowing for comparison between different irradiation processes, sources, and post-processing treatments to determine optimal manufacturing protocols.
Related Standards
For comprehensive material evaluation and consistent application, ASTM F2381-19 should be considered alongside:
- ASTM F2102: Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants.
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method.
- ASTM E1421: Practice for Describing and Measuring Performance of Fourier Transform Mid-Infrared (FT-MIR) Spectrometers.
These related standards provide additional guidance on material analysis, validation, and instrument performance, supporting a holistic approach to UHMWPE implant component quality assurance. Adopting ASTM F2381-19 helps ensure compliance with international regulations and the delivery of safe, high-performance medical devices.
Buy Documents
ASTM F2381-19 - Standard Test Method for Evaluating Trans-Vinylene Yield in Irradiated Ultra-High Molecular Weight Polyethylene Fabricated Forms Intended for Surgical Implants by Infrared Spectroscopy
REDLINE ASTM F2381-19 - Standard Test Method for Evaluating Trans-Vinylene Yield in Irradiated Ultra-High Molecular Weight Polyethylene Fabricated Forms Intended for Surgical Implants by Infrared Spectroscopy
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2381-19 is a standard published by ASTM International. Its full title is "Standard Test Method for Evaluating Trans-Vinylene Yield in Irradiated Ultra-High Molecular Weight Polyethylene Fabricated Forms Intended for Surgical Implants by Infrared Spectroscopy". This standard covers: SIGNIFICANCE AND USE 4.1 Published literature shows that the yield of radiolytic reactions that occur during radiation treatment increases with radiation dose level. Measurement of the products of these reactions can be used as an internal dosimeter. 4.2 Trans-vinylene unsaturations are formed during ionization treatment by abstraction of a hydrogen molecule, and to a lesser extent by the recombination of two adjacent alkyl free radicals that reside on the same chain. 4.3 Previous work generated calibration curves of trans-vinylene absorption area as a function of absorbed radiation dose, yielding a linear relationship for both gamma- and electron beam-irradiated polyethylene. 4.4 This data can be used to determine received dose as a function of position, assuming a calibration curve (TVI versus radiation dose level) is known for the particular material and radiation conditions used, and can be used to determine uniformity of dose level in irradiated polyethylene. SCOPE 1.1 This test method describes the measurement of the number of trans-vinylene groups in ultra-high molecular weight polyethylene (UHMWPE) intended for use in medical implants. The material is analyzed by infrared spectroscopy. 1.2 This test method is based on Guide F2102. 1.3 The applicability of the infrared method has been demonstrated in other literature reports. This particular method, using the intensity (area) of the C-H absorption centered at 1370 cm-1 to normalize for the sample’s thickness, will be validated by an Interlaboratory Study (ILS) conducted according to Practice E691. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 Published literature shows that the yield of radiolytic reactions that occur during radiation treatment increases with radiation dose level. Measurement of the products of these reactions can be used as an internal dosimeter. 4.2 Trans-vinylene unsaturations are formed during ionization treatment by abstraction of a hydrogen molecule, and to a lesser extent by the recombination of two adjacent alkyl free radicals that reside on the same chain. 4.3 Previous work generated calibration curves of trans-vinylene absorption area as a function of absorbed radiation dose, yielding a linear relationship for both gamma- and electron beam-irradiated polyethylene. 4.4 This data can be used to determine received dose as a function of position, assuming a calibration curve (TVI versus radiation dose level) is known for the particular material and radiation conditions used, and can be used to determine uniformity of dose level in irradiated polyethylene. SCOPE 1.1 This test method describes the measurement of the number of trans-vinylene groups in ultra-high molecular weight polyethylene (UHMWPE) intended for use in medical implants. The material is analyzed by infrared spectroscopy. 1.2 This test method is based on Guide F2102. 1.3 The applicability of the infrared method has been demonstrated in other literature reports. This particular method, using the intensity (area) of the C-H absorption centered at 1370 cm-1 to normalize for the sample’s thickness, will be validated by an Interlaboratory Study (ILS) conducted according to Practice E691. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2381-19 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2381-19 has the following relationships with other standards: It is inter standard links to ASTM F2381-10, ASTM F2102-17, ASTM F2102-13, ASTM E691-13, ASTM E691-11, ASTM E1421-99(2009), ASTM E691-08, ASTM F2102-06e1, ASTM F2102-06, ASTM E691-05, ASTM E1421-99(2004), ASTM F2102-01e1, ASTM F2102-01, ASTM E1421-99, ASTM E691-99. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2381-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2381 − 19
Standard Test Method for
Evaluating Trans-Vinylene Yield in Irradiated Ultra-High
Molecular Weight Polyethylene Fabricated Forms Intended
for Surgical Implants by Infrared Spectroscopy
This standard is issued under the fixed designation F2381; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Determine the Precision of a Test Method
E1421 Practice for Describing and Measuring Performance
1.1 This test method describes the measurement of the
of Fourier Transform Mid-Infrared (FT-MIR) Spectrom-
number of trans-vinylene groups in ultra-high molecular
eters: Level Zero and Level One Tests
weight polyethylene (UHMWPE) intended for use in medical
F2102 Guide for Evaluating the Extent of Oxidation in
implants. The material is analyzed by infrared spectroscopy.
Polyethylene Fabricated Forms Intended for Surgical
1.2 This test method is based on Guide F2102.
Implants
1.3 The applicability of the infrared method has been
3. Terminology
demonstrated in other literature reports. This particular
3.1 Definitions:
method, using the intensity (area) of the C-H absorption
-1
centered at 1370 cm to normalize for the sample’s thickness, 3.1.1 trans-vinylene index (TVI)—trans-vinylene index is
defined as the ratio of the area of the absorption peak centered
will be validated by an Interlaboratory Study (ILS) conducted
-1
according to Practice E691. near 965 cm to the area of the absorption peak centered near
-1
1370 cm .
1.4 The values stated in SI units are to be regarded as
3.1.2 depth locator (DL)—measurement of the distance
standard. No other units of measurement are included in this
fromthearticularsurface,orsurfaceofinterest,thataspectrum
standard.
was collected and a corresponding TVI calculated.
1.5 This standard does not purport to address all of the
3.1.3 trans-vinylene index profile—trans-vinylene index
safety concerns, if any, associated with its use. It is the
profile is defined as the graphical representation of variation of
responsibility of the user of this standard to establish appro-
the sample’s trans-vinylene index with distance from its
priate safety, health, and environmental practices and deter-
articular surface or the surface of interest.This is a plot ofTVI
mine the applicability of regulatory limitations prior to use.
versus DL. Typically, the graph will show the profile through
1.6 This international standard was developed in accor-
the entire thickness of the sample.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
4. Significance and Use
Development of International Standards, Guides and Recom-
4.1 Published literature shows that the yield of radiolytic
mendations issued by the World Trade Organization Technical
reactions that occur during radiation treatment increases with
Barriers to Trade (TBT) Committee.
radiation dose level. Measurement of the products of these
2. Referenced Documents reactions can be used as an internal dosimeter.
2.1 ASTM Standards:
4.2 Trans-vinylene unsaturations are formed during ioniza-
E691 Practice for Conducting an Interlaboratory Study to
tion treatment by abstraction of a hydrogen molecule, and to a
lesser extent by the recombination of two adjacent alkyl free
radicals that reside on the same chain.
This test method is under the jurisdiction ofASTM Committee F04 on Medical
4.3 Previous work generated calibration curves of trans-
and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.15 on Material Test Methods.
vinylene absorption area as a function of absorbed radiation
Current edition approved Oct. 1, 2019. Published November 2019. Originally
dose, yielding a linear relationship for both gamma- and
approved 2004. Last previous edition approved in 2010 as F2381–10. DOI:
electron beam-irradiated polyethylene.
10.1520/F2381-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
4.4 This data can be used to determine received dose as a
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
function of position, assuming a calibration curve (TVI versus
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. radiation dose level) is known for the particular material and
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2381 − 19
radiation conditions used, and can be used to determine 7. Preparation of Apparatus
uniformity of dose level in irradiated polyethylene.
7.1 Prepare the infrared spectrometer for collection of a
transmission absorption spectrum from a thin film of the
5. Apparatus
UHMWPE sample according to the manufacturer’s
recommendations, Practice E1421, and the conditions de-
5.1 Infrared Spectrometer:
scribed in 5.1.
5.1.1 A calibrated infrared spectrometer capable of record-
ing a transmission absorption spectrum over a minimum range
8. Procedure
-1
of 900 to about 2000 cm using about 200 µm-thick films at a
-1
8.1 The test film (slice) shall be first configured in the
resolution of 4 cm and an aperture of approximately 200 by
spectrometer (after an appropriate background spectrum has
200 µm for a rectangular aperture, or 200 µm diameter for a
been collected) such that the aperture is positioned over the
circular aperture.
first 200 µm of the film starting at the surface of interest.
5.1.1.1 Other modes of collection (that is, reflection, attenu-
ated total reflection (ATR), and so forth) and aperture and
8.2 Subsequent spectra shall be collected sequentially at
sampling step sizes may be used to generate the sample’s
increments matching the aperture size (that is, about 200 µm)
absorption spectrum provided they can be demonstrated to
from the articular surface, or surface of interest, across the
produce equivalent results. Too large an aperture can result in
width of the film to the opposite surface.
a loss of profile accuracy.
8.3 Larger increments may be used; however, too large an
5.1.1.2 When a Fourier Transform Infrared (FTIR) spec-
increment size may result in a loss of profile accuracy.
trometer is used, a minimum of 32 scans shall be collected per
spectrum as a default. A fewer number of scans may be
9. Calculation of Results
performed if a user can verify that their FTIR spectrometer can
9.1 Trans-vinylene Peak Area:
reproducibly measure the TVI with less thana5%difference
9.1.1 For each absorbance spectrum, calculate the total area
in TVI compared with the default number of scans. In no case
-1
of the absorption peak centered near 965 cm (see Fig. 1).
shall a TVI be reported based on fewer than 8 scans per
9.1.2 This area is the area below the sample’s absorbance
spectrum.
curve and above the straight baseline drawn between the same
5.1.1.3 The FTIR instrument and sample compartment
starting and ending points used in 3.1.1.
should be purged with a moisture- and carbon-dioxide-free
9.2 Normalization of Peak Area:
inert gas (for example, nitrogen, helium, or argon) to minimize
9.2.1 For each absorbance spectrum, calculate the total area
spectral interference from these components.
-1
of the absorption peak centered near 1370 cm (see Fig. 2).
5.2 Specimen Holder—Equipment, such as an x-y table,
9.2.2 This area is the area below the sample’s absorbance
capable of accurately positioning the sample under the FTIR
curve and above the straight baseline drawn between the same
aperture with a minimum resolution at the scale of the aperture
starting and ending points used in 3.1.1.
dimensions.
9.3 Trans-vinylene Index (TVI)—
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2381 − 10 F2381 − 19
Standard Test Method for
Evaluating Trans-Vinylene Yield in Irradiated Ultra-High
Molecular Weight Polyethylene Fabricated Forms Intended
for Surgical Implants by Infrared Spectroscopy
This standard is issued under the fixed designation F2381; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method describes the measurement of the number of trans-vinylene groups in ultra-high molecular weight
polyethylene (UHMWPE) intended for use in medical implants. The material is analyzed by infrared spectroscopy.
1.2 This test method is based on Guide F2102.
1.3 The applicability of the infrared method has been demonstrated in other literature reports. This particular method, using the
-1
intensity (area) of the C-H absorption centered at 1370 cm to normalize for the sample’s thickness, will be validated by an
Interlaboratory Study (ILS) conducted according to Practice E691.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E1421 Practice for Describing and Measuring Performance of Fourier Transform Mid-Infrared (FT-MIR) Spectrometers: Level
Zero and Level One Tests
F2102 Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants
3. Terminology
3.1 Definitions:
3.1.1 trans-vinylene index (TVI)—a trans-vinylene index is defined as the ratio of the area of the absorption peak centered near
-1 -1
965 cm to the area of the absorption peak centered near 1370 cm .
3.1.2 depth locator (DL)—a measurement of the distance from the articular surface, or surface of interest, that a spectrum was
collected and a corresponding TVI calculated.
3.1.3 trans-vinylene index profile—a trans-vinylene index profile is defined as the graphical representation of variation of the
sample’s trans-vinylene index with distance from its articular surface or the surface of interest. This is a plot of TVI versus DL.
Typically, the graph will show the profile through the entire thickness of the sample.
4. Significance and Use
4.1 Published literature shows that the yield of radiolytic reactions that occur during radiation treatment increases with radiation
dose level. Measurement of the products of these reactions can be used as an internal dosimeter.
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.15 on Material Test Methods.
Current edition approved June 1, 2010Oct. 1, 2019. Published July 2010November 2019. Originally approved 2004. Last previous edition approved in 20042010 as
F2381–04.–10. DOI: 10.1520/F2381-10.10.1520/F2381-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2381 − 19
4.2 Trans-vinylene unsaturations are formed during ionization treatment by abstraction of a hydrogen molecule, and to a lesser
extent by the recombination of two adjacent alkyl free radicals that reside on the same chain.
4.3 Previous work generated calibration curves of trans-vinylene absorption area as a function of absorbed radiation dose,
yielding a linear relationship for both gamma- and electron beam-irradiated polyethylene.
4.4 This data can be used to determine received dose as a function of position, assuming a calibration curve (TVI versus
radiation dose level) is known for the particular material and radiation conditions used, and can be used to determine uniformity
of dose level in irradiated polyethylene.
5. Apparatus
5.1 Infrared Spectrometer:
5.1.1 A calibrated infrared spectrometer capable of recording a transmission absorption spectrum over a minimum range of 900
-1 -1
to about 2000 cm using about 200 μm-thick films at a resolution of 4 cm and an aperture of approximately 200 by 200 μm for
a rectangular aperture, or 200 μm diameter for a circular aperture.
5.1.1.1 Other modes of collection (that is, reflection, attenuated total reflection (ATR), and so forth) and aperture and sampling
step sizes may be used to generate the sample’s absorption spectrum provided they can be demonstrated to produce equivalent
results. Too large an aperture can result in a loss of profile accuracy.
5.1.1.2 When a Fourier Transform Infrared (FTIR) spectrometer is used, a minimum of 32 scans shall be collected per spectrum
as a default. A fewer number of scans may be performed if a user can verify that their FTIR spectrometer can reproducibly measure
the TVI with less than a 5 % difference in TVI compared with the default number of scans. In no case shall a TVI be reported
based on fewer than 8 scans per spectrum.
5.1.1.3 The FTIR instrument and sample compartment should be purged with a moisture- and carbon-dioxide-free inert gas (for
example, nitrogen, helium, or argon) to minimize spectral interference from these components.
5.2 Specimen Holder—Equipment, such as an x-y table, capable of accurately positioning the sample under the FTIR aperture
with a minimum resolution at the scale of the aperture dimensions.
5.3 Microtome—Equipment capable of producing films of thickness 200 μm or less of a sample perpendicular to the articular
surface or the surface of interest.
6. Sampling, Test Specimens, and Test Units
6.1 Using a microtome, or other appropriate device, prepare a thin slice of the sample about 200 μm thick. If the detected signal
from the FTIR is too weak with this thickness, a thicker sample may be used.
6.2 The slice shall typically be taken near the center of the sample’s articular surface or the surface of interest.
6.3 The orientation of the slice shall typically be perpendicular to the articular surface or the surface of interest.
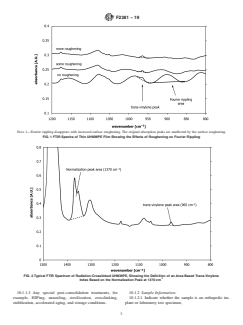
6.4 Waviness in the infrared spectrum caused by Fourier rippling on the lower wavelengths can interfere with the trans-vinylene
-1
absorbance at 965 cm . This rippling is caused by internal reflection of the infrared beam, and can be avoided by lightly rubbing
the sample film against 400 grit sandpaper until the film becomes translucent. This roughening procedure should be done slowly
to avoid heating the film, and should be performed until the area under a single peak due to Fourier rippling is less than 10 % of
the area under the trans-vinylene peak.
7. Preparation of Apparatus
7.1 Prepare the infrared spectrometer for collection of a transmission absorption spectrum from a thin film of the UHMWPE
sample according to the manufacturer’s recommendations, Practice E1421, and the conditions described in 5.1.
8. Procedur
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...