ASTM F2977-20
(Test Method)Standard Test Method for Small Punch Testing of Polymeric Biomaterials Used in Surgical Implants
Standard Test Method for Small Punch Testing of Polymeric Biomaterials Used in Surgical Implants
SIGNIFICANCE AND USE
4.1 Miniature specimen testing techniques are used to characterize the mechanical behavior of polymer stock materials and surgical implants after manufacture, sterilization, shelf aging, radiation crosslinking, thermal treatment, filler incorporation, and implantation (1-3). Furthermore, experimental materials can be evaluated after accelerated aging, fatigue testing, and hip, knee, or spine wear simulation. Consequently, the small punch test makes it possible to examine relationships between wear performance and mechanical behavior. This test method can also be used to rank the mechanical behavior relative to a reference control material.
4.2 Small punch testing results may vary with specimen preparation and with the speed and environment of testing. Consequently, where precise comparative results are desired, these factors must be carefully controlled.
SCOPE
1.1 This test method covers the determination of mechanical behavior of polymeric biomaterials by small punch testing of miniature disk specimens (0.5 mm in thickness and 6.4 mm in diameter). The test method has been established for characterizing surgical materials after ram extrusion or compression molding (1-3)2; for evaluating as-manufactured implants and sterilization method effects (4, 5); as well as for testing of implants that have been retrieved (explanted) from the human body (6, 7).
1.2 The results of the small punch test, namely the peak load, ultimate displacement, ultimate load, and work to failure, provide metrics of the yielding, ultimate strength, ductility, and toughness under multiaxial loading conditions. Because the mechanical behavior can be different when loaded under uniaxial and multiaxial loading conditions (8), the small punch test provides a complementary mechanical testing technique to the uniaxial tensile test. However, it should be noted that the small punch test results may not correlate with uniaxial tensile test results.
1.3 In addition to its use as a research tool in implant retrieval analysis, the small punch test can be used as a laboratory screening test to evaluate new materials with minimal material waste (1).
1.4 The small punch test has been applied to other polymers, including polymethyl methacrylate (PMMA) bone cement, polyacetal, and high density polyethylene (HDPE), ultra high molecular weight polyethylene (UHMWPE), and polyetheretherketone (PEEK) (2, 3, 5, 9, 10). This standard outlines general guidelines for the small punch testing of implantable polymers.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.15 - Material Test Methods
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Jan-2020
- Effective Date
- 01-Aug-2019
- Effective Date
- 15-Apr-2019
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Dec-2018
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Sep-2017
- Effective Date
- 15-Aug-2017
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-May-2014
- Effective Date
- 01-Nov-2013
- Effective Date
- 01-May-2013
Overview
ASTM F2977-20: Standard Test Method for Small Punch Testing of Polymeric Biomaterials Used in Surgical Implants provides a standardized approach for assessing the mechanical behavior of polymeric materials in the field of surgical implants. Developed by ASTM International, the standard specifies procedures for small punch testing of miniature disk-shaped specimens, enabling precise evaluation of mechanical properties after factors such as manufacturing, sterilization, aging, and clinical use. By focusing on miniature samples, this method minimizes material requirements and supports material development, quality control, and the study of retrieved implants.
Key Topics
- Specimen Preparation and Testing: The standard outlines detailed guidelines for specimen preparation, emphasizing the importance of specimen orientation, surface finish, and size control for accurate and repeatable results.
- Measurement Metrics: Through the small punch test, key mechanical properties are determined, including peak load, ultimate load, ultimate displacement, and work to failure. These metrics reflect a material’s yielding, ductility, strength, and toughness under multiaxial loading.
- Applicability: The method is applicable to a wide range of implantable polymers such as ultra-high molecular weight polyethylene (UHMWPE), polyetheretherketone (PEEK), polymethyl methacrylate (PMMA) bone cement, high-density polyethylene (HDPE), and polyacetal.
- Environmental and Test Control: The standard highlights the sensitivity of results to factors like testing speed, temperature, and environmental conditions. Rigorous control of these variables is essential for comparison and material ranking.
Applications
Small punch testing per ASTM F2977-20 is a versatile tool with significant practical applications in the medical device industry, research, and quality assurance:
- Material Development and Qualification: Evaluate the mechanical properties of experimental polymers and new formulations used in surgical implants, such as joint replacements, with minimal material consumption.
- Manufacturing Quality Control: Assess the effects of manufacturing methods (e.g., ram extrusion, compression molding) and post-processing (sterilization, crosslinking, thermal treatment) to ensure reliable mechanical performance.
- Aging and Wear Simulation: Test polymers after accelerated aging, shelf life simulation, and wear or fatigue testing (including hip, knee, and spine implant simulations) to predict in vivo performance.
- Implant Retrieval Analysis: Analyze explanted implants to correlate clinical performance and wear with changes in mechanical properties, aiding failure analysis and device improvement.
- Comparative Assessment: Rank the mechanical performance of materials relative to reference controls, supporting material selection and regulatory submissions.
- Lab Screening: Efficiently screen small quantities of new materials, avoiding unnecessary waste and expediting research workflows.
Related Standards
ASTM F2977-20 is supported and complemented by a set of related standards that provide additional guidance for material testing and implant evaluation:
- ASTM D695: Test Method for Compressive Properties of Rigid Plastics
- ASTM D883: Terminology Relating to Plastics
- ASTM E4: Practices for Force Verification of Testing Machines
- ASTM E83: Practice for Verification and Classification of Extensometer Systems
- ASTM E177: Practice for Use of the Terms Precision and Bias in ASTM Test Methods
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
- ASTM F1714: Guide for Gravimetric Wear Assessment of Prosthetic Hip Designs in Simulator Devices
- ASTM F2003: Practice for Accelerated Aging of UHMWPE after Gamma Irradiation in Air
- ASTM F2102: Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants
By implementing ASTM F2977-20, laboratories and manufacturers benefit from standardized, reliable assessment techniques tailored to the evolving needs of polymeric biomaterials in surgical implant applications. This enhances product safety, compliance, and innovation in the biomedical sector.
Buy Documents
ASTM F2977-20 - Standard Test Method for Small Punch Testing of Polymeric Biomaterials Used in Surgical Implants
REDLINE ASTM F2977-20 - Standard Test Method for Small Punch Testing of Polymeric Biomaterials Used in Surgical Implants
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2977-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Small Punch Testing of Polymeric Biomaterials Used in Surgical Implants". This standard covers: SIGNIFICANCE AND USE 4.1 Miniature specimen testing techniques are used to characterize the mechanical behavior of polymer stock materials and surgical implants after manufacture, sterilization, shelf aging, radiation crosslinking, thermal treatment, filler incorporation, and implantation (1-3). Furthermore, experimental materials can be evaluated after accelerated aging, fatigue testing, and hip, knee, or spine wear simulation. Consequently, the small punch test makes it possible to examine relationships between wear performance and mechanical behavior. This test method can also be used to rank the mechanical behavior relative to a reference control material. 4.2 Small punch testing results may vary with specimen preparation and with the speed and environment of testing. Consequently, where precise comparative results are desired, these factors must be carefully controlled. SCOPE 1.1 This test method covers the determination of mechanical behavior of polymeric biomaterials by small punch testing of miniature disk specimens (0.5 mm in thickness and 6.4 mm in diameter). The test method has been established for characterizing surgical materials after ram extrusion or compression molding (1-3)2; for evaluating as-manufactured implants and sterilization method effects (4, 5); as well as for testing of implants that have been retrieved (explanted) from the human body (6, 7). 1.2 The results of the small punch test, namely the peak load, ultimate displacement, ultimate load, and work to failure, provide metrics of the yielding, ultimate strength, ductility, and toughness under multiaxial loading conditions. Because the mechanical behavior can be different when loaded under uniaxial and multiaxial loading conditions (8), the small punch test provides a complementary mechanical testing technique to the uniaxial tensile test. However, it should be noted that the small punch test results may not correlate with uniaxial tensile test results. 1.3 In addition to its use as a research tool in implant retrieval analysis, the small punch test can be used as a laboratory screening test to evaluate new materials with minimal material waste (1). 1.4 The small punch test has been applied to other polymers, including polymethyl methacrylate (PMMA) bone cement, polyacetal, and high density polyethylene (HDPE), ultra high molecular weight polyethylene (UHMWPE), and polyetheretherketone (PEEK) (2, 3, 5, 9, 10). This standard outlines general guidelines for the small punch testing of implantable polymers. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 Miniature specimen testing techniques are used to characterize the mechanical behavior of polymer stock materials and surgical implants after manufacture, sterilization, shelf aging, radiation crosslinking, thermal treatment, filler incorporation, and implantation (1-3). Furthermore, experimental materials can be evaluated after accelerated aging, fatigue testing, and hip, knee, or spine wear simulation. Consequently, the small punch test makes it possible to examine relationships between wear performance and mechanical behavior. This test method can also be used to rank the mechanical behavior relative to a reference control material. 4.2 Small punch testing results may vary with specimen preparation and with the speed and environment of testing. Consequently, where precise comparative results are desired, these factors must be carefully controlled. SCOPE 1.1 This test method covers the determination of mechanical behavior of polymeric biomaterials by small punch testing of miniature disk specimens (0.5 mm in thickness and 6.4 mm in diameter). The test method has been established for characterizing surgical materials after ram extrusion or compression molding (1-3)2; for evaluating as-manufactured implants and sterilization method effects (4, 5); as well as for testing of implants that have been retrieved (explanted) from the human body (6, 7). 1.2 The results of the small punch test, namely the peak load, ultimate displacement, ultimate load, and work to failure, provide metrics of the yielding, ultimate strength, ductility, and toughness under multiaxial loading conditions. Because the mechanical behavior can be different when loaded under uniaxial and multiaxial loading conditions (8), the small punch test provides a complementary mechanical testing technique to the uniaxial tensile test. However, it should be noted that the small punch test results may not correlate with uniaxial tensile test results. 1.3 In addition to its use as a research tool in implant retrieval analysis, the small punch test can be used as a laboratory screening test to evaluate new materials with minimal material waste (1). 1.4 The small punch test has been applied to other polymers, including polymethyl methacrylate (PMMA) bone cement, polyacetal, and high density polyethylene (HDPE), ultra high molecular weight polyethylene (UHMWPE), and polyetheretherketone (PEEK) (2, 3, 5, 9, 10). This standard outlines general guidelines for the small punch testing of implantable polymers. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2977-20 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2977-20 has the following relationships with other standards: It is inter standard links to ASTM D883-24, ASTM D883-23, ASTM D883-20, ASTM D883-19c, ASTM D883-19a, ASTM D883-19, ASTM D883-18a, ASTM D883-18, ASTM F1714-96(2018), ASTM F2102-17, ASTM D883-17, ASTM E4-14, ASTM E177-14, ASTM F2102-13, ASTM E177-13. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2977-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2977 − 20
Standard Test Method for
Small Punch Testing of Polymeric Biomaterials Used in
Surgical Implants
This standard is issued under the fixed designation F2977; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
1.1 Thistestmethodcoversthedeterminationofmechanical
mine the applicability of regulatory limitations prior to use.
behavior of polymeric biomaterials by small punch testing of
1.6 This international standard was developed in accor-
miniature disk specimens (0.5 mm in thickness and 6.4 mm in
dance with internationally recognized principles on standard-
diameter). The test method has been established for character-
ization established in the Decision on Principles for the
izing surgical materials after ram extrusion or compression
2 Development of International Standards, Guides and Recom-
molding (1-3) ; for evaluating as-manufactured implants and
mendations issued by the World Trade Organization Technical
sterilization method effects (4, 5); as well as for testing of
Barriers to Trade (TBT) Committee.
implants that have been retrieved (explanted) from the human
body (6, 7).
2. Referenced Documents
1.2 The results of the small punch test, namely the peak
2.1 ASTM Standards:
load, ultimate displacement, ultimate load, and work to failure,
D695 Test Method for Compressive Properties of Rigid
providemetricsoftheyielding,ultimatestrength,ductility,and
Plastics
toughness under multiaxial loading conditions. Because the
D883 Terminology Relating to Plastics
mechanical behavior can be different when loaded under
E4 Practices for Force Verification of Testing Machines
uniaxial and multiaxial loading conditions (8), the small punch
E83 Practice for Verification and Classification of Exten-
test provides a complementary mechanical testing technique to
someter Systems
the uniaxial tensile test. However, it should be noted that the
E177 Practice for Use of the Terms Precision and Bias in
small punch test results may not correlate with uniaxial tensile
ASTM Test Methods
test results.
E691 Practice for Conducting an Interlaboratory Study to
1.3 In addition to its use as a research tool in implant
Determine the Precision of a Test Method
retrieval analysis, the small punch test can be used as a
F1714 Guide for GravimetricWearAssessment of Prosthetic
laboratory screening test to evaluate new materials with
Hip Designs in Simulator Devices
minimal material waste (1).
F1715 Guide for Wear Assessment of Prosthetic Knee De-
1.4 Thesmallpunchtesthasbeenappliedtootherpolymers, signs in Simulator Devices (Withdrawn 2006)
F2003 Practice for Accelerated Aging of Ultra-High Mo-
including polymethyl methacrylate (PMMA) bone cement,
polyacetal, and high density polyethylene (HDPE), ultra high lecular Weight Polyethylene after Gamma Irradiation in
Air
molecular weight polyethylene (UHMWPE), and polyethere-
therketone (PEEK) (2, 3, 5, 9, 10). This standard outlines F2102 Guide for Evaluating the Extent of Oxidation in
Polyethylene Fabricated Forms Intended for Surgical
general guidelines for the small punch testing of implantable
polymers. Implants
1.5 This standard does not purport to address all of the
3. Terminology
safety concerns, if any, associated with its use. It is the
3.1 Definitions:
This test method is under the jurisdiction ofASTM Committee F04 on Medical
and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.15 on Material Test Methods. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved April 1, 2020. Published June 2020. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
ɛ1
approved in 2013. Last previous edition approved in 2013 as F2977–13 . DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F2977-20. the ASTM website.
2 4
The boldface numbers in parentheses refer to the list of references at the end of The last approved version of this historical standard is referenced on
this standard. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2977 − 20
3.1.1 small punch test, n—a test wherein the specimen is of
miniature size relative to conventional mechanical test
specimens, is disk-shaped, and is loaded axisymmetrically in
bending by a hemispherical-head punch.
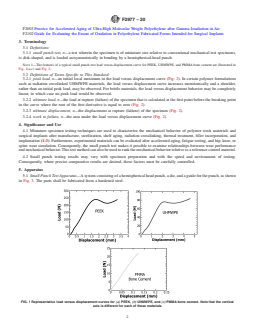
NOTE 1—The features of a typical small punch test load versus
displacement curve for PEEK, UHMWPE, and PMMA bone cement are
illustrated in Fig. 1(a-c) and Fig. 2.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 peak load, n—an initial local maximum in the load
versus displacement curve (Fig. 2). In certain polymer formu-
lations such as radiation crosslinked UHMWPE materials, the
load versus displacement curve increases monotonically and a
shoulder, rather than an initial peak load, may be observed. For
brittle materials, the load versus displacement behavior may be
completely linear, in which case no peak load would be
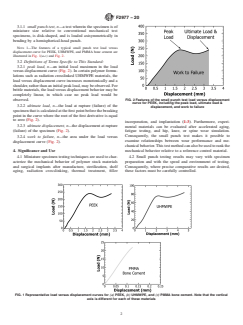
FIG. 2 Features of the small punch test load versus displacement
observed.
curve for PEEK, including the peak load, ultimate load &
3.2.2 ultimate load, n—the load at rupture (failure) of the
displacement, and work to failure
specimenthatiscalculatedatthefirstpointbeforethebreaking
point in the curve where the root of the first derivative is equal
to zero (Fig. 2).
incorporation, and implantation (1-3). Furthermore, experi-
3.2.3 ultimate displacement, n—the displacement at rupture
mental materials can be evaluated after accelerated aging,
(failure) of the specimen (Fig. 2).
fatigue testing, and hip, knee, or spine wear simulation.
Consequently, the small punch test makes it possible to
3.2.4 work to failure, n—the area under the load versus
examine relationships between wear performance and me-
displacement curve (Fig. 2).
chanicalbehavior.Thistestmethodcanalsobeusedtorankthe
4. Significance and Use mechanical behavior relative to a reference control material.
4.1 Miniature specimen testing techniques are used to char- 4.2 Small punch testing results may vary with specimen
acterize the mechanical behavior of polymer stock materials preparation and with the speed and environment of testing.
and surgical implants after manufacture, sterilization, shelf Consequently, where precise comparative results are desired,
aging, radiation crosslinking, thermal treatment, filler these factors must be carefully controlled.
FIG. 1 Representative load versus displacement curves for (a) PEEK, (b) UHMWPE, and (c) PMMA bone cement. Note that the vertical
axis is different for each of these materials
F2977 − 20
5. Apparatus 5.5 Micrometers—Suitable micrometers, reading to 0.0001
in. (0.0025 mm), shall be used to record the diameter and
5.1 Small Punch Test Apparatus—A system consisting of a
thickness of the specimens.
hemispherical head punch, a die, and a guide for the punch, as
5.6 Thermometer—Suitable thermometer or thermocouple,
shown in Fig. 3. The parts shall be fabricated from a hardened
steel. reading to 0.1°C, shall be used to record the test temperature
within the range 20° to 24°C.
5.1.1 Guide—Thefunctionoftheguideistoalignthepunch
relative to the specimen, which rests in a disk-shaped recess.
6. Test Specimens
The inner diameter of the guide bore shall be 0.1010 +0.0002/-
6.1 As the test results are known to be sensitive to prepa-
0.0000 in. (2.565 +0.005/0.000 mm), and the specimen recess
ration technique, the specimens described in 6.2 and 6.3 shall
shall be 0.0200 +0.0004/-0.0000 in. (0.508 +0.010/-0.000 mm)
be used. The specimens may be prepared by machining
in depth and 0.2520 6 0.0005 in. (6.401 6 0.013 mm) in
operations from materials in sheet, rod, plate, or implant form.
diameter.
All machining operations shall be done carefully so that
5.1.2 Die—Thefunctionofthedieistoconstrainthesample
smooth surfaces result. Great care shall be taken in machining
during testing. The inner diameter of the die bore shall be
the faces so that smooth, parallel surfaces result.
0.1500 6 0.0005 in. (3.810 6 0.013 mm).
NOTE 2—Although specimen fabrication methods other than machining
5.1.3 Punch—The hemispherical head punch shall have a
(e.g.,microtoming)maybeused,theuseofalternatespecimenpreparation
diameter of 0.1000 in. (2.540 mm), with a tolerance of
methods have not yet been shown to provide equivalent test results to
+0.0000/-0.0002 in. (+0.000/-0.005 mm).
machined specimens.
5.2 Testing Machine—Any suitable testing machine as de-
6.2 If specimens are prepared from stock materials, the
scribed in Method D695, consisting of a drive mechanism and
orientation of the test specimen with respect to the manufac-
a load indicator. A load cell should be used in which the peak
turing direction (e.g., perpendicular to the extrusion or com-
load and ultimate load fall within the 10-90% capacity of the
pression molding axis) shall be recorded, along with the
equipment. The accuracy of the machine shall be verified at
distance from the surface of the stock material. If the speci-
least once per year, as specified by Method D695 and Practice
mens are machined directly from actual implants, the orienta-
E4.
tion and depth from the articulating surface shall be recorded.
5.3 Compressometer—This instrument, described in section
6.3 The standard test specimen shall have a thickness of
5.2 from Method D695, can be used to determine the distance 0.0200 +0.0002/-0.0003 in. (0.508 +0.005/-0.008 mm) and a
between the die and the punch during the test. If the actuator
diameter of 0.250 +0.000/-0.005 in. (6.350 +0.000/-0.127
displacementofthetestingmachinecanbeshowntodetermine mm). For comparisons made at a single institution, alternate
punch displacement within 1% of the value measured by a
sample geometry may be specified. However, the results from
suitably calibrated compressometer (as defined in Practice thatinstitutionwilllikelynotbecomparabletotheresultsfrom
E83), actuator displacement shall be used as reference.
other institutions.
5.4 Compression Platen—The punch shall rest on a com-
6.4 Specimens falling outside the dimensional tolerances
pression platen or tool for applying the load to the punch. specified in 6.3 shall be discarded.
FIG. 3 Schematic of the small punch test apparatus, including the die, guide, hemispherical head punch, and miniature disk shaped
specimen
F2977 − 20
7. Number of Test Specimens 11. Calculations
7.1 A minimum of five specimens per material condition is 11.1 Calculate the initial peak load (if applicable), the
recommended, however, due to the high level of repeatability ultimate load, ultimate displacement, and work to failure as
of the test as reported in the literature, four specimens per shown in Fig. 1.The units for the initial peak load and ultimate
material condition may be sufficient to establish significant load shall be in N. Ultimate displacement shall be reported in
differences between material groups. The user should conduct mm, and the work to failure shall be reported in mJ.
NOTE 3—The ultimate load, ultimate displacement, and work to failure
the appropriate initial material evaluations followed by statis-
will be calculated based on the highest load achieve
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: F2977 − 13 F2977 − 20
Standard Test Method for
Small Punch Testing of Polymeric Biomaterials Used in
Surgical Implants
This standard is issued under the fixed designation F2977; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Editorial corrections were made to 5.1 in February 2020.
1. Scope
1.1 This test method covers the determination of mechanical behavior of polymeric biomaterials by small punch testing of
miniature disk specimens (0.5 mm in thickness and 6.4 mm in diameter). The test method has been established for characterizing
surgical materials after ram extrusion or compression molding (1-3) ; for evaluating as-manufactured implants and sterilization
method effects (4, 5); as well as for testing of implants that have been retrieved (explanted) from the human body (6, 7).
1.2 The results of the small punch test, namely the peak load, ultimate displacement, ultimate load, and work to failure, provide
metrics of the yielding, ultimate strength, ductility, and toughness under multiaxial loading conditions. Because the mechanical
behavior can be different when loaded under uniaxial and multiaxial loading conditions (8), the small punch test provides a
complementary mechanical testing technique to the uniaxial tensile test. However, it should be noted that the small punch test
results may not correlate with uniaxial tensile test results.
1.3 In addition to its use as a research tool in implant retrieval analysis, the small punch test can be used as a laboratory
screening test to evaluate new materials with minimal material waste (1).
1.4 The small punch test has been applied to other polymers, including polymethyl methacrylate (PMMA) bone cement,
polyacetal, and high density polyethylene (HDPE), ultra high molecular weight polyethylene (UHMWPE), and polyetherether-
ketone (PEEK) (2, 3, 5, 9, 10). This standard outlines general guidelines for the small punch testing of implantable polymers.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D695 Test Method for Compressive Properties of Rigid Plastics
D883 Terminology Relating to Plastics
E4 Practices for Force Verification of Testing Machines
E83 Practice for Verification and Classification of Extensometer Systems
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
F1714 Guide for Gravimetric Wear Assessment of Prosthetic Hip Designs in Simulator Devices
F1715 Guide for Wear Assessment of Prosthetic Knee Designs in Simulator Devices (Withdrawn 2006)
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.15 on Material Test Methods.
Current edition approved June 1, 2013April 1, 2020. Published August 2013.June 2020. Originally approved in 2013. Last previous edition approved in 2013 as
ɛ1
F2977–13 . DOI: 10.1520/F2977-13E01.10.1520/F2977-20.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2977 − 20
F2003 Practice for Accelerated Aging of Ultra-High Molecular Weight Polyethylene after Gamma Irradiation in Air
F2102 Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants
3. Terminology
3.1 Definitions:
3.1.1 small punch test, n—a test wherein the specimen is of miniature size relative to conventional mechanical test specimens,
is disk-shaped, and is loaded axisymmetrically in bending by a hemispherical-head punch.
NOTE 1—The features of a typical small punch test load versus displacement curve for PEEK, UHMWPE, and PMMA bone cement are illustrated in
Fig. 1(a-c) and Fig. 2.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 peak load, n—an initial local maximum in the load versus displacement curve (Fig. 2). In certain polymer formulations
such as radiation crosslinked UHMWPE materials, the load versus displacement curve increases monotonically and a shoulder,
rather than an initial peak load, may be observed. For brittle materials, the load versus displacement behavior may be completely
linear, in which case no peak load would be observed.
3.2.2 ultimate load, n—the load at rupture (failure) of the specimen that is calculated at the first point before the breaking point
in the curve where the root of the first derivative is equal to zero (Fig. 2).
3.2.3 ultimate displacement, n—the displacement at rupture (failure) of the specimen (Fig. 2).
3.2.4 work to failure, n—the area under the load versus displacement curve (Fig. 2).
4. Significance and Use
4.1 Miniature specimen testing techniques are used to characterize the mechanical behavior of polymer stock materials and
surgical implants after manufacture, sterilization, shelf aging, radiation crosslinking, thermal treatment, filler incorporation, and
implantation (1-3). Furthermore, experimental materials can be evaluated after accelerated aging, fatigue testing, and hip, knee, or
spine wear simulation. Consequently, the small punch test makes it possible to examine relationships between wear performance
and mechanical behavior. This test method can also be used to rank the mechanical behavior relative to a reference control material.
4.2 Small punch testing results may vary with specimen preparation and with the speed and environment of testing.
Consequently, where precise comparative results are desired, these factors must be carefully controlled.
5. Apparatus
5.1 Small Punch Test Apparatus—A system consisting of a hemispherical head punch, a die, and a guide for the punch, as shown
in Fig. 3. The parts shall be fabricated from a hardened steel.
FIG. 1 Representative load versus displacement curves for (a) PEEK, (b) UHMWPE, and (c) PMMA bone cement. Note that the vertical
axis is different for each of these materials
F2977 − 20
FIG. 2 Features of the small punch test load versus displacement curve for PEEK, including the peak load, ultimate load &
displacement, and work to failure
5.1.1 Guide—The function of the guide is to align the punch relative to the specimen, which rests in a disk-shaped recess. The
inner diameter of the guide bore shall be 0.1010 +0.0002/-0.0000 in. (2.565 +0.005/0.000 mm), and the specimen recess shall be
0.0200 +0.0004/-0.0000 in. (0.508 +0.010/-0.000 mm) in depth and 0.2520 6 0.0005 in. (6.401 6 0.013 mm) in diameter.
5.1.2 Die—The function of the die is to constrain the sample during testing. The inner diameter of the die bore shall be 0.1500
6 0.0005 in. (3.810 6 0.013 mm).
5.1.3 Punch—The hemispherical head punch shall have a diameter of 0.1000 in. (2.540 mm), with a tolerance of
+0.0000/-0.0002 in. (+0.000/-0.005 mm).
5.2 Testing Machine—Any suitable testing machine as described in Method D695, consisting of a drive mechanism and a load
indicator. A load cell should be used in which the peak load and ultimate load fall within the 10-90% capacity of the equipment.
The accuracy of the machine shall be verified at least once per year, as specified by Method D695 and Practice E4.
5.3 Compressometer—This instrument, described in section 5.2 from Method D695, can be used to determine the distance
between the die and the punch during the test. If the actuator displacement of the testing machine can be shown to determine punch
displacement within 1% of the value measured by a suitably calibrated compressometer (as defined in Practice E83), actuator
displacement shall be used as reference.
5.4 Compression Platen—The punch shall rest on a compression platen or tool for applying the load to the punch.
5.5 Micrometers—Suitable micrometers, reading to 0.0001 in. (0.0025 mm), shall be used to record the diameter and thickness
of the specimens.
5.6 Thermometer—Suitable thermometer or thermocouple, reading to 0.1°C, shall be used to record the test temperature within
the range 20° to 24°C.
6. Test Specimens
6.1 As the test results are known to be sensitive to preparation technique, the specimens described in 6.2 and 6.3 shall be used.
The specimens may be prepared by machining operations from materials in sheet, rod, plate, or implant form. All machining
operations shall be done carefully so that smooth surfaces result. Great care shall be taken in machining the faces so that smooth,
parallel surfaces result.
NOTE 2—Although specimen fabrication methods other than machining (e.g., microtoming) may be used, the use of alternate specimen preparation
methods have not yet been shown to provide equivalent test results to machined specimens.
6.2 If specimens are prepared from stock materials, the orientation of the test specimen with respect to the manufacturing
direction (e.g., perpendicular to the extrusion or compression molding axis) shall be recorded, along with the distance from the
surface of the stock material. If the specimens are machined directly from actual implants, the orientation and depth from the
articulating surface shall be recorded.
6.3 The standard test specimen shall have a thickness of 0.0200 +0.0002/-0.0003 in. (0.508 +0.005/-0.008 mm) and a diameter
of 0.250 +0.000/-0.005 in. (6.350 +0.000/-0.127 mm). For comparisons made at a single institution, alternate sample geometry may
be specified. However, the results from that institution will likely not be comparable to the results from other institutions.
6.4 Specimens falling outside the dimensional tolerances specified in 6.3 shall be discarded.
F2977 − 20
FIG. 3 Schematic of the small punch test apparatus, including the die, guide, hemispherical head punch, and miniature disk shaped
specimen
7. Number of Test Specimens
7.1 A minimum of five specimens per material condition is recommended, however, due to the high level of repeatability of the
test as reported in the literature, four specimens per material condition may be sufficient to establish significant differences between
material groups. The user should conduct the appropriate initial material evaluations followed by statistical analysis to determine
the appropriate sample size for testing.
8. Speed of Testing
8.1 The test results are sensitive to testing speed. Therefore, the speed of the testing machine shall be calibrated to within 1%
accuracy at least once per year, as specified in Standard D695.
-1
8.2 The reference speed for small punch testing shall be 0.5 mm min , unless otherwise indicated by the customer or
specification.
9. Conditioning
9.1 Condition the test specimens, die, and punch, at 23° 6 2°C for at least 1 h prior to
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...