ASTM F1091-20
(Specification)Standard Specification for Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy Surgical Fixation Wire (UNS R30605)
Standard Specification for Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy Surgical Fixation Wire (UNS R30605)
ABSTRACT
This specification covers the requirements for the manufacture of wrought cobalt-20chromium-15tungsten-10nickel surgical fixation wire. The starting material, dimensions, and tolerances shall conform to the requirements specified. The surgical fixation wire is usually furnished in the bright-annealed condition. Mechanical properties of the surgical fixation wire such as tensile strength and elongation shall be determined.
SCOPE
1.1 This specification covers the chemical, mechanical, and metallurgical requirements for the manufacture of wrought cobalt-20chromium-15tungsten-10nickel surgical fixation wire.
1.2 Units—The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.12 - Metallurgical Materials
Relations
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Jan-2024
- Effective Date
- 01-Jan-2024
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Mar-2023
- Effective Date
- 05-May-2020
- Effective Date
- 01-Sep-2019
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Jan-2018
- Effective Date
- 15-Jul-2016
- Effective Date
- 01-Jan-2016
- Effective Date
- 01-Jan-2016
- Effective Date
- 01-Feb-2015
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-May-2014
Overview
ASTM F1091-20 is the standard specification for wrought cobalt-20chromium-15tungsten-10nickel alloy surgical fixation wire (UNS R30605). Developed by ASTM International, this specification details the chemical, mechanical, and metallurgical requirements for the manufacture of surgical fixation wires commonly used in orthopedic and surgical implant applications. The wire, characterized by its high performance and biocompatibility, is typically provided in the bright-annealed condition, making it suitable for medical environments where reliability and material consistency are crucial.
Key Topics
- Material Composition: Specifies the use of wrought cobalt-20chromium-15tungsten-10nickel alloy (UNS R30605), conforming to precise chemical requirements and based on established standards.
- Mechanical Properties: The standard defines requirements such as tensile strength and elongation, ensuring that surgical fixation wire meets necessary performance benchmarks for medical use.
- Dimensional Tolerances: Detailed permissible variations for diameter, size tolerance, and out-of-round tolerance to guarantee strict dimensional control of the wire.
- Surface Condition: Requirements to minimize surface imperfections such as tool marks, nicks, scratches, and other defects, ensuring that wires are clean and free from foreign material.
- Testing and Certification: Mandates careful tension testing and adherence to USP requirements for nonabsorbable surgical sutures. Certification of conformity with all specified requirements must accompany each shipment.
- Quality Management: Suppliers must maintain a quality management system conforming to ISO 9001, ISO 13485, or comparable programs, ensuring traceability and consistency.
Applications
The primary application of ASTM F1091-20 specification is in the production of surgical fixation wire for orthopedic and general surgical procedures. Key use cases include:
- Surgical Fixation: Used in the stabilization and support of fractured bones and during reconstructive surgery.
- Implant Devices: Serves as a raw material for medical device manufacturers, particularly for products intended for long-term contact with bone and soft tissue.
- Medical Sutures: Qualified for applications requiring nonabsorbable surgical sutures in compliance with United States Pharmacopeia (USP) standards.
- Custom Medical Components: The specification allows for flexibility in dimensions and tolerances, supporting the fabrication of wires tailored to specific device designs.
Due to its excellent mechanical properties and long-term clinical experience demonstrating an acceptable biological response, this alloy wire is a preferred choice in applications demanding high strength, ductility, and corrosion resistance.
Related Standards
ASTM F1091-20 references and relates to several other key standards to ensure comprehensive coverage in medical and surgical applications:
- ASTM F90: Specification for wrought cobalt-20chromium-15tungsten-10nickel alloy for surgical implant applications.
- ASTM E8/E8M: Test methods for tension testing of metallic materials.
- ASTM A484/A484M: General requirements for stainless steel bars, billets, and forgings.
- ASTM A555/A555M: General requirements for stainless steel wire and wire rods.
- ASTM F86: Practice for surface preparation and marking of metallic surgical implants.
- USP Nonabsorbable Surgical Suture Standards: Ensures compatibility and safety for suture use.
- ISO 9001 & ISO 13485: Quality management system requirements, including those specific to medical devices.
Manufacturers and purchasers should always refer to the current versions of related standards and ensure compliance with both ASTM and applicable international requirements for medical devices.
Keywords: ASTM F1091-20, surgical fixation wire, cobalt-chromium-tungsten-nickel alloy, orthopedic wire, UNS R30605, mechanical properties, biocompatibility, medical device manufacturing, implant materials, standard specification.
Buy Documents
ASTM F1091-20 - Standard Specification for Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy Surgical Fixation Wire (UNS R30605)
REDLINE ASTM F1091-20 - Standard Specification for Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy Surgical Fixation Wire (UNS R30605)
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1091-20 is a technical specification published by ASTM International. Its full title is "Standard Specification for Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy Surgical Fixation Wire (UNS R30605)". This standard covers: ABSTRACT This specification covers the requirements for the manufacture of wrought cobalt-20chromium-15tungsten-10nickel surgical fixation wire. The starting material, dimensions, and tolerances shall conform to the requirements specified. The surgical fixation wire is usually furnished in the bright-annealed condition. Mechanical properties of the surgical fixation wire such as tensile strength and elongation shall be determined. SCOPE 1.1 This specification covers the chemical, mechanical, and metallurgical requirements for the manufacture of wrought cobalt-20chromium-15tungsten-10nickel surgical fixation wire. 1.2 Units—The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This specification covers the requirements for the manufacture of wrought cobalt-20chromium-15tungsten-10nickel surgical fixation wire. The starting material, dimensions, and tolerances shall conform to the requirements specified. The surgical fixation wire is usually furnished in the bright-annealed condition. Mechanical properties of the surgical fixation wire such as tensile strength and elongation shall be determined. SCOPE 1.1 This specification covers the chemical, mechanical, and metallurgical requirements for the manufacture of wrought cobalt-20chromium-15tungsten-10nickel surgical fixation wire. 1.2 Units—The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F1091-20 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1091-20 has the following relationships with other standards: It is inter standard links to ASTM F1091-12, ASTM A484/A484M-24, ASTM E8/E8M-24, ASTM A484/A484M-23a, ASTM A484/A484M-23, ASTM A484/A484M-20a, ASTM A484/A484M-19, ASTM A484/A484M-18a, ASTM A484/A484M-18, ASTM E8/E8M-16, ASTM A555/A555M-16, ASTM A484/A484M-16, ASTM E8/E8M-15, ASTM A484/A484M-14a, ASTM A484/A484M-14. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1091-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:F1091 −20
Standard Specification for
Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy
Surgical Fixation Wire (UNS R30605)
This standard is issued under the fixed designation F1091; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* Determine Conformance with Specifications
F86 Practice for Surface Preparation and Marking of Metal-
1.1 This specification covers the chemical, mechanical, and
lic Surgical Implants
metallurgical requirements for the manufacture of wrought
F90 Specification for Wrought Cobalt-20Chromium-
cobalt-20chromium-15tungsten-10nickel surgical fixation
15Tungsten-10NickelAlloy for Surgical ImplantApplica-
wire.
tions (UNS R30605)
1.2 Units—The values stated in either SI units or inch-
IEEE/ASTM SI 10 American National Standard for Use of
pound units are to be regarded separately as standard. The
theInternationalSystemofUnits(SI):TheModernMetric
values stated in each system are not necessarily exact equiva-
System
lents; therefore, to ensure conformance with the standard, each 3
2.2 USP Standards:
system shall be used independently of the other, and values
USP29 Nonabsorbable Surgical Suture, U.S. Pharmacopeia
from the two systems shall not be combined.
2.3 ISO Standards:
1.3 This standard does not purport to address all of the
ISO 9001 Quality Management Systems—Requirements
safety concerns, if any, associated with its use. It is the
ISO 13485 Medical Devices—Quality Management
responsibility of the user of this standard to establish appro-
Systems—Requirements for Regulatory Purposes
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use. 3. General Requirements for Delivery
1.4 This international standard was developed in accor-
3.1 In addition to the requirements of this specification, all
dance with internationally recognized principles on standard-
requirements of the current editions of Specification F90 shall
ization established in the Decision on Principles for the
apply.
Development of International Standards, Guides and Recom-
3.2 In cases where a conflict exists between this specifica-
mendations issued by the World Trade Organization Technical
tion and the standards listed in Section 2, this specification
Barriers to Trade (TBT) Committee.
shall take precedence.
2. Referenced Documents
4. Terminology
2.1 ASTM Standards:
4.1 Definitions of Terms Specific to This Standard:
A484/A484M Specification for General Requirements for
4.1.1 lot, n—the total number of mill products produced
Stainless Steel Bars, Billets, and Forgings
from the same melt heat under the same conditions at essen-
A555/A555M Specification for General Requirements for
tially the same time.
Stainless Steel Wire and Wire Rods
E8/E8M Test Methods for Tension Testing of Metallic Ma-
5. Ordering Information
terials
5.1 Inquiries and orders for material under this specification
E29 Practice for Using Significant Digits in Test Data to
shall include the following information:
5.1.1 Quantity;
This specification is under the jurisdiction of ASTM Committee F04 on
5.1.2 ASTM designation and date of issue;
Medical and Surgical Materials and Devices and is the direct responsibility of
5.1.3 Material requirements (see Section 6);
Subcommittee F04.12 on Metallurgical Materials.
5.1.4 Mechanical properties (see Section 7);
Current edition approved Feb. 1, 2020. Published April 2020. Originally
approved 1991. Last previous edition approved in 2012 as F1091 – 12. DOI:
10.1520/F1091-20.
2 3
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville,
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM MD 20852-1790, http://www.usp.org.
Standards volume information, refer to the standard’s Document Summary page on Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1091−20
5.1.5 Form (bar or wire); 7. Mechanical Requirements
5.1.6 Applicable dimension, including size, thickness,
7.1 Surgical fixation wire shall conform to the appropriate
width, and length (exact, random, or multiples) or drawing
mechanical properties specified in Table 1.
number;
7.2 Perform tension tests in accordance with Test Methods
5.1.7 Tolerances—Unless otherwise specified by purchaser,
E8/E8M using a 254 mm [10 in.] gauge length and crosshead
tolerances must meet the requirements of Specification A484/
speed of 254 mm⁄min [10 in.⁄min]. Should any of the test
A484M, Specification A555/A555M, or both, as applicable;
specimens not meet the specified requirements, test two addi-
5.1.8 Condition (see 6.1);
tional test pieces representative of the same lot, in the same
5.1.9 Finish (see 6.2);
manner, for each failed test piece. The lot shall be considered
5.1.10 Special tests (if any); and
in compliance only if all additional test pieces meet the
5.1.11 Other requirements.
specified requirements.
6. Materials and Manufacture
7.3 Tensile test results for which any specimen fractures
outside the gauge length shall be considered acceptable if the
6.1 Condition:
elongation meets the minimum requirements specified in Table
6.1.1 The starting material used to make fixation wire must
1. Refer to subsections 7.11.4 and 7.12.5 of Test Methods
meet Specification F90.
E8/E8M. If the elongation is less than the minimum
6.1.2 Surgical fixation wire shall conform to the specified
requirement, discard the test and retest. Retest one specimen
chemical requirements of Specification F90.
for each specimen that did not meet the minimum requirement.
6.2 Finish:
7.4 The wire shall meet the requirements of USP for
6.2.1 Surgical fixation wire is usually furnished in the
Nonabsorbable Surgical Sutures, when tested in accordance
bright-annealed condition. Other surface finishes shall be
with 7.2.
specified as agreed to between supplier and purchaser.
6.2.2 The surface of surgical fixation wire conforming to
8. Dimensional and Permissible Variations
this specification shall be processed to minimize imperfections
suchastoolmarks,nicks,scratches,cracks,cavities,spurs,and 8.1 Units of Measure:
other defects that would impair the serviceability of the wire. 8.1.1 Selection—This specification requires that the pur-
The surfaces shall be cleaned to minimize the presence of chaser selects the units (SI or inch-pound) to be used for
foreign material. product certification. In the absence of a stated selection of
6.2.3 The wire may be subjected to a passivation process if units on the purchase order, this selection may be expressed by
requested by the purchaser. Such passivation process shall be the purchaser in several alternate forms listed in order of
performed in accordance with Practice F86. precedence.
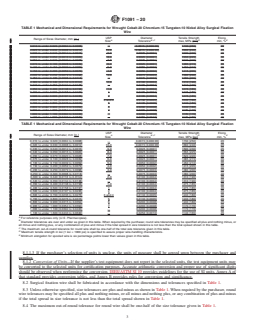
TABLE 1 Mechanical and Dimensional Requirements for Wrought Cobalt-20 Chromium-15 Tungsten-10 Nickel Alloy Surgical Fixation
Wire
USP Diameter Tensile Strength Elong.
Range of Sizes Diameter, mm [in.]
A B ,C D E
Size Tolerance max, MPa [ksi] min, %
0.010 to under 0.020 [0.0004 to 0.0008] . 0.0015 [0.000 06] 1730 [250] 20
0.020 to under 0.030 [0.0008 to 0.0012] 10-0 0.0015 [0.000 06] 1660 [240] 20
0.030
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F1091 − 12 F1091 − 20
Standard Specification for
Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy
Surgical Fixation Wire (UNS R30605)
This standard is issued under the fixed designation F1091; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This specification covers the chemical, mechanical, and metallurgical requirements for the manufacture of wrought
cobalt-20chromium-15tungsten-10nickel surgical fixation wire.
1.2 Units—The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated
in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used
independently of the other, and values from the two systems shall not be combined.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
A484/A484M Specification for General Requirements for Stainless Steel Bars, Billets, and Forgings
A555/A555M Specification for General Requirements for Stainless Steel Wire and Wire Rods
E8E8/E8M Test Methods for Tension Testing of Metallic Materials [Metric] E0008_E0008M
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
F86 Practice for Surface Preparation and Marking of Metallic Surgical Implants
F90 Specification for Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy for Surgical Implant Applications (UNS
R30605)
IEEE/ASTM SI 10 American National Standard for Use of the International System of Units (SI): The Modern Metric System
2.2 USP Standards:
Nonabsorbable Surgical Suture, USP29 Nonabsorbable Surgical Suture, U.S. Pharmacopeia
2.3 ISO Standard:Standards:
ISO 9001 Quality Management Systems—Requirements
ISO 13485 Medical Devices—Quality Management Systems—Requirements for Regulatory Purposes
3. General Requirements for Delivery
3.1 In addition to the requirements of this specification, all requirements of the current editions of Specification F90 shall apply.
3.2 In cases where a conflict exists between this specification and the standards listed in Section 2, this specification shall take
precedence.
This specification is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.12 on Metallurgical Materials.
Current edition approved Dec. 1, 2012Feb. 1, 2020. Published December 2012April 2020. Originally approved 1991. Last previous edition approved in 20082012 as
F1091 – 08.F1091 – 12. DOI: 10.1520/F1091-12.10.1520/F1091-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville, MD 20852-1790, http://www.usp.org.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1091 − 20
4. Terminology
4.1 Definitions of Terms Specific to This Standard:
4.1.1 lot, n—the total number of mill products produced from the same melt heat under the same conditions at essentially the
same time.
5. Ordering Information
5.1 Inquiries and orders for material under this specification shall include the following information:
5.1.1 Quantity,Quantity;
5.1.2 ASTM designation and date of issue,issue;
5.1.3 Material requirements (see Section 6), );
5.1.4 Mechanical properties (see Section 7),);
5.1.5 Form,Form (bar or wire);
5.1.6 Dimensional requirements, including diameter and diameter tolerance,Applicable dimension, including size, thickness,
width, and length (exact, random, or multiples) or drawing number;
5.1.7 Tolerances—Surface condition and handling,Unless otherwise specified by purchaser, tolerances must meet the
requirements of Specification A484/A484M, Specification A555/A555M, or both, as applicable;
5.1.8 Condition (see 6.1);
5.1.9 Finish (see 6.2);
5.1.10 Special tests (if applicable),any); and
5.1.11 Other requirements.
6. Material Requirements Materials and Manufacture
6.1 The starting material used to make fixation wire must meet Specification F90.
6.1 Surgical fixation wire shall conform to the specified chemical requirements of Specification F90. Condition:
6.1.1 The starting material used to make fixation wire must meet Specification F90.
6.1.2 Surgical fixation wire shall conform to the specified chemical requirements of Specification F90.
6.2 Finish:
6.2.1 Surgical fixation wire is usually furnished in the bright-annealed condition. Other surface finishes shall be specified as
agreed to between supplier and purchaser.
6.2.2 The surface of surgical fixation wire conforming to this specification shall be processed to minimize imperfections such
as tool marks, nicks, scratches, cracks, cavities, spurs, and other defects that would impair the serviceability of the wire. The
surfaces shall be cleaned to minimize the presence of foreign material.
6.2.3 The wire may be subjected to a passivation process if requested by the purchaser. Such passivation process shall be
performed in accordance with Practice F86.
7. Mechanical Requirements
7.1 Surgical fixation wire shall conform to the appropriate mechanical properties specified in Table 1.
7.2 Perform tension tests in accordance with Test Methods E8E8/E8M using a 254-mm (10-in.) gage254 mm [10 in.] gauge
length and crosshead speed of 254254 mm mm/min⁄min [10 in. (10 in./min). ⁄min]. Should any of the test specimens not meet the
specified requirements, test two additional test pieces representative of the same lot, in the same manner, for each failed test piece.
The lot shall be considered in compliance only if all additional test pieces meet the specified requirements.
7.3 Tensile test results for which any specimen fractures outside the gagegauge length shall be considered acceptable if the
elongation meets the minimum requirements specified in Table 1. Refer to subsections 7.11.4 and 7.11.57.12.5 of Test Methods
E8E8/E8M. If the elongation is less than the minimum requirement, discard the test and retest. Retest one specimen for each
specimen that did not meet the minimum requirement.
7.4 The wire shall meet the requirements of USP for Nonabsorbable Surgical Sutures, when tested in accordance with 7.2.
8. Dimensional Requirements and Permissible Variations
8.1 Units of Measure:
8.1.1 Selection—This specification requires that the purchaser selects the units (SI or inch-pound) to be used for product
certification. In the absence of a stated selection of units on the purchase order, this selection may be expressed by the purchaser
in several alternate forms listed in order of precedence.
8.1.1.1 If the purchaser and supplier have a history of using specific units, these units shall continue to be certified until
expressly changed by the purchaser.
8.1.1.2 In the absence of historic precedence, if the units used to define the product on the purchaser’s purchase order (PO),
specifications, and engineering drawing are consistent, these units shall be used by the supplier for product certification.
F1091 − 20
TABLE 1 Mechanical and Dimensional Requirements for Wrought Cobalt-20 Chromium-15 Tungsten-10 Nickel Alloy Surgical Fixation
Wire
USP Diameter Tensile Strength Elong.
Range of Sizes Diameter, mm (in.)
A B ,C D E
Size Tolerance max, MPa (ksi) min, %
0.010 to under 0.020 (0.0004 to 0.0008) . 0.0015 (0.000 06) 1730 (250) 20
0.020 to under 0.030 (0.0008 to 0.0012) 10-0 0.0015 (0.000 06) 1660 (240) 20
0.030 to under 0.040 (0.0012 to 0.0016) 9-0 0.0025 (0.0001) 1590 (230) 25
0.040 to under 0.050 (0.0016 to 0.0020) 8-0 0.0025 (0.0001) 1555 (225) 30
0.050 to under 0.070 (0.0020 to 0.0028) 7-0 0.0025 (0.0001) 1520 (220) 30
0.070 to under 0.100 (0.0028 to 0.0039) 6-0 0.0025 (0.0001) 1385 (215) 35
0.100 to under 0.150 (0.0039 to 0.0059) 5-0 0.0050 (0.0002) 1450 (210) 35
0.150 to under 0.200 (0.0059 to 0.0079) 4-0 0.0050 (0.0002) 1415 (205) 35
0.200 to under 0.250 (0.0079 to 0.0098) 3-0 0.0075 (0.0003) 1380 (200) 40
0.250 to under 0.300 (0.0098 to 0.0118) . 0.0075 (0.0003) 1380 (200) 40
0.300 to under 0.340 (0.0118 to 0.0134) 2-0 0.0100 (0.0004) 1310 (190) 40
0.340 to under 0.350 (0.0134 to 0.0138) . 0.0100 (0.0004) 1310 (190) 40
0.350 to under 0.400 (0.0138 to 0.0158) 1-0 0.0100 (0.0004) 1275 (185) 40
0.400 to under 0.500 (0.0158 to 0.0197) 1 0.0100 (0.0004) 1275 (185) 40
0.500 to under 0.600 (0.0197 to 0.0236) 2 0.0100 (0.0004) 1275 (185) 45
0.600 to under 0.700
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...