ASTM F1223-20
(Test Method)Standard Test Method for Determination of Total Knee Replacement Constraint
Standard Test Method for Determination of Total Knee Replacement Constraint
SIGNIFICANCE AND USE
4.1 This test method, when applied to available products and proposed prototypes, is meant to provide a database of product functionality capabilities (in light of the suggested test regimens) that is hoped will aid the physician in making a more informed total knee replacement (TKR) selection.
4.2 A proper matching of TKR functional restorative capabilities and the recipient's (patient’s) needs is more likely to be provided by a rational testing protocol of the implant in an effort to reveal certain device characteristics pertinent to the selection process.
4.3 The TKR product designs are varied and offer a wide range of constraint (stability). The constraint of the TKR in the in vitro condition depends on several geometrical and kinematic interactions among the implant's components which can be identified and quantified. The degree of TKR's kinematic interactions should correspond to the recipient's needs as determined by the physician during clinical examination.
4.4 For mobile bearing knee systems, the constraint of the entire implant construct shall be characterized. Constraint of mobile bearings is dictated by design features at both the inferior and superior articulating interfaces.
4.5 The methodology, utility, and limitations of constraint/laxity testing are discussed.3, 4 The authors recognize that evaluating isolated implants (that is, without soft tissues) does not directly predict in vivo behavior, but will allow comparisons among designs. Constraint testing is also useful for characterizing implant performance at extreme ranges of motion which may be encountered in vivo at varying frequencies, depending on the patient’s anatomy, pre-operative capability, and post-operative activities and lifestyle.
SCOPE
1.1 This test method may be used to compare the constraint characteristics of total knee replacements (TKRs) with the intent of comparing new designs to existing clinically successful designs or to determine the constraint differences between two similar or dissimilar designs.
1.2 This test method covers the means by which a TKR constraint may be quantified according to motion delineated by the inherent articular design as determined under specific loading conditions in an in-vitro environment.
1.3 Tests deemed applicable to the constraint determination are antero-posterior draw, medio-lateral shear, rotary laxity, valgus-varus rotation, and distraction, as applicable. Also covered is the identification of geometrical parameters of the contacting surfaces which would influence this motion and the means of reporting the test results. (See Practices E4.)
1.4 This test method is not a wear test.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Jun-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 15-Jun-2020
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 15-Apr-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Jul-2008
- Effective Date
- 01-Jul-2008
- Effective Date
- 01-Aug-2007
- Effective Date
- 01-Jan-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Aug-2006
- Effective Date
- 01-Mar-2006
Overview
ASTM F1223-20: Standard Test Method for Determination of Total Knee Replacement Constraint is a crucial ASTM International standard used in the evaluation of total knee replacement (TKR) systems. This test method outlines procedures for quantifying the constraint characteristics of various TKR designs under specified loading conditions in an in vitro environment. The resulting data forms a vital database aimed at helping clinicians make informed choices when selecting knee implants suited to patient needs. While the standard does not directly assess in vivo performance or implant durability, it allows consistent relative comparisons among existing, new, or prototype knee implant designs.
Key Topics
- Constraint Testing: The standard defines constraint as a TKR’s resistance to specific displacements under load, influenced by its geometric and kinematic design.
- Degrees of Freedom: Focus is placed on five principal axes of motion relevant to TKR function: antero-posterior draw, medio-lateral shear, rotary laxity, valgus-varus rotation, and distraction.
- Methodology: Step-by-step guidance is given for measuring constraints, including specimen preparation, alignment, loading protocols, and key measurements (such as AP and ML displacements).
- Mobile Bearing Knee Systems: Special consideration is provided for the assessment of constraint in implants with mobile bearing components, covering both inferior and superior articulating surfaces.
- Applications of Data: The generated database guides matching implant function with patient requirements, assisting surgeons in device selection during preoperative planning.
Applications
The practical value of ASTM F1223-20 lies in its broad utility across the orthopedic sector:
- Implant Comparison and Selection: Manufacturers and researchers use these test results to compare new prosthesis designs with those that are well-established clinically, aiding regulatory submissions and iterative product development.
- Clinician Support: Surgeons and healthcare teams benefit from standardized constraint data, enabling better alignment of implant characteristics with individual patient factors such as remaining soft tissue stability and bone quality.
- Design Optimization: The standardized testing protocol highlights constraint-limiting features, helping engineers refine knee replacement prosthesis geometry for specific clinical indications.
- Database for Evidence-Based Practice: A consistent methodology allows for compilation of broad datasets, informing evidence-based guidelines and future standards development.
- Regulatory and Quality Compliance: Conformance with ASTM F1223-20 is often required to meet regulatory, accreditation, or hospital procurement criteria for total knee prostheses.
Related Standards
- ASTM F2083: Specification for Knee Replacement Prosthesis. Provides design and material specifications for knee implants, which often reference constraint testing as part of device classification or labeling.
- ASTM E4: Practices for Force Verification of Testing Machines. Ensures that testing equipment used in constraint assessment meets accuracy requirements.
- FDA Guidance: FDA device classification for knee prostheses recognizes multiple levels of constraint, and data according to ASTM F1223-20 can support regulatory filings.
- International Principles: This standard aligns with globally recognized procedures established by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Summary
ASTM F1223-20 underpins the reliable, repeatable evaluation of total knee replacement constraint, offering the orthopedic community clear protocols for comparing and characterizing implant designs. By providing a scientific basis for implant selection and standardizing test methods, it fosters better patient outcomes, supports regulatory compliance, and encourages innovation in TKR technology. For those involved in knee arthroplasty device development, testing, and clinical deployment, adherence to ASTM F1223-20 is essential for quality assurance and optimal patient care.
Buy Documents
ASTM F1223-20 - Standard Test Method for Determination of Total Knee Replacement Constraint
REDLINE ASTM F1223-20 - Standard Test Method for Determination of Total Knee Replacement Constraint
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1223-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Total Knee Replacement Constraint". This standard covers: SIGNIFICANCE AND USE 4.1 This test method, when applied to available products and proposed prototypes, is meant to provide a database of product functionality capabilities (in light of the suggested test regimens) that is hoped will aid the physician in making a more informed total knee replacement (TKR) selection. 4.2 A proper matching of TKR functional restorative capabilities and the recipient's (patient’s) needs is more likely to be provided by a rational testing protocol of the implant in an effort to reveal certain device characteristics pertinent to the selection process. 4.3 The TKR product designs are varied and offer a wide range of constraint (stability). The constraint of the TKR in the in vitro condition depends on several geometrical and kinematic interactions among the implant's components which can be identified and quantified. The degree of TKR's kinematic interactions should correspond to the recipient's needs as determined by the physician during clinical examination. 4.4 For mobile bearing knee systems, the constraint of the entire implant construct shall be characterized. Constraint of mobile bearings is dictated by design features at both the inferior and superior articulating interfaces. 4.5 The methodology, utility, and limitations of constraint/laxity testing are discussed.3, 4 The authors recognize that evaluating isolated implants (that is, without soft tissues) does not directly predict in vivo behavior, but will allow comparisons among designs. Constraint testing is also useful for characterizing implant performance at extreme ranges of motion which may be encountered in vivo at varying frequencies, depending on the patient’s anatomy, pre-operative capability, and post-operative activities and lifestyle. SCOPE 1.1 This test method may be used to compare the constraint characteristics of total knee replacements (TKRs) with the intent of comparing new designs to existing clinically successful designs or to determine the constraint differences between two similar or dissimilar designs. 1.2 This test method covers the means by which a TKR constraint may be quantified according to motion delineated by the inherent articular design as determined under specific loading conditions in an in-vitro environment. 1.3 Tests deemed applicable to the constraint determination are antero-posterior draw, medio-lateral shear, rotary laxity, valgus-varus rotation, and distraction, as applicable. Also covered is the identification of geometrical parameters of the contacting surfaces which would influence this motion and the means of reporting the test results. (See Practices E4.) 1.4 This test method is not a wear test. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This test method, when applied to available products and proposed prototypes, is meant to provide a database of product functionality capabilities (in light of the suggested test regimens) that is hoped will aid the physician in making a more informed total knee replacement (TKR) selection. 4.2 A proper matching of TKR functional restorative capabilities and the recipient's (patient’s) needs is more likely to be provided by a rational testing protocol of the implant in an effort to reveal certain device characteristics pertinent to the selection process. 4.3 The TKR product designs are varied and offer a wide range of constraint (stability). The constraint of the TKR in the in vitro condition depends on several geometrical and kinematic interactions among the implant's components which can be identified and quantified. The degree of TKR's kinematic interactions should correspond to the recipient's needs as determined by the physician during clinical examination. 4.4 For mobile bearing knee systems, the constraint of the entire implant construct shall be characterized. Constraint of mobile bearings is dictated by design features at both the inferior and superior articulating interfaces. 4.5 The methodology, utility, and limitations of constraint/laxity testing are discussed.3, 4 The authors recognize that evaluating isolated implants (that is, without soft tissues) does not directly predict in vivo behavior, but will allow comparisons among designs. Constraint testing is also useful for characterizing implant performance at extreme ranges of motion which may be encountered in vivo at varying frequencies, depending on the patient’s anatomy, pre-operative capability, and post-operative activities and lifestyle. SCOPE 1.1 This test method may be used to compare the constraint characteristics of total knee replacements (TKRs) with the intent of comparing new designs to existing clinically successful designs or to determine the constraint differences between two similar or dissimilar designs. 1.2 This test method covers the means by which a TKR constraint may be quantified according to motion delineated by the inherent articular design as determined under specific loading conditions in an in-vitro environment. 1.3 Tests deemed applicable to the constraint determination are antero-posterior draw, medio-lateral shear, rotary laxity, valgus-varus rotation, and distraction, as applicable. Also covered is the identification of geometrical parameters of the contacting surfaces which would influence this motion and the means of reporting the test results. (See Practices E4.) 1.4 This test method is not a wear test. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F1223-20 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1223-20 has the following relationships with other standards: It is inter standard links to ASTM F1223-14, ASTM E4-14, ASTM F2083-11, ASTM E4-10, ASTM F2083-10, ASTM E4-09a, ASTM E4-09, ASTM E4-08, ASTM F2083-08e1, ASTM F2083-08, ASTM F2083-07, ASTM E4-07, ASTM F2083-06b, ASTM F2083-06a, ASTM F2083-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1223-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:F1223 −20
Standard Test Method for
Determination of Total Knee Replacement Constraint
This standard is issued under the fixed designation F1223; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method may be used to compare the constraint
E4 Practices for Force Verification of Testing Machines
characteristics of total knee replacements (TKRs) with the
F2083 Specification for Knee Replacement Prosthesis
intent of comparing new designs to existing clinically success-
ful designs or to determine the constraint differences between
3. Terminology
two similar or dissimilar designs.
3.1 Definitions—Items in this category refer to the geo-
1.2 This test method covers the means by which a TKR
metricalandkinematicaspectsofTKRdesignsastheyrelateto
constraint may be quantified according to motion delineated by
their human counterparts:
the inherent articular design as determined under specific
3.1.1 anterior curvature, n—a condylar design which is
loading conditions in an in-vitro environment.
generally planar except for a concave—upward region anteri-
orly on the tibial component.
1.3 Tests deemed applicable to the constraint determination
3.1.2 anterior posterior (AP), adj—any geometrical length
are antero-posterior draw, medio-lateral shear, rotary laxity,
aligned with the AP orientation.
valgus-varus rotation, and distraction, as applicable. Also
covered is the identification of geometrical parameters of the
3.1.3 AP displacement, n—the relative linear translation
contacting surfaces which would influence this motion and the
between components in the AP direction.
means of reporting the test results. (See Practices E4.)
3.1.4 AP draw load, n—the force applied to the movable
component with its vector aligned in the AP direction causing
1.4 This test method is not a wear test.
or intending to cause an AP displacement.
1.5 The values stated in SI units are to be regarded as
3.1.5 biconcave, n—a condylar design with pronounced AP
standard. No other units of measurement are included in this
and MLcondylar radii seen as a “dish” in the tibial component
standard.
or a “toroid” in the femoral component.
1.6 This standard does not purport to address all of the
3.1.6 bearing surface, n—those regions of the component
safety concerns, if any, associated with its use. It is the
which are intended to contact its counterpart for load transmis-
responsibility of the user of this standard to establish appro-
sion.
priate safety, health, and environmental practices and deter-
3.1.7 condyle, n—entity designed to emulate the joint
mine the applicability of regulatory limitations prior to use.
anatomy and used as a bearing surface primarily for transmis-
1.7 This international standard was developed in accor-
sion of the joint reaction force with geometrical properties
dance with internationally recognized principles on standard-
which tend to govern the general kinematics of the TKR.
ization established in the Decision on Principles for the
3.1.8 distraction, n—the separation of the femoral compo-
Development of International Standards, Guides and Recom-
nent(s) from the tibial component(s) in the z-direction.
mendations issued by the World Trade Organization Technical
3.1.9 femoralsideconstraint,n—thatconstraintprovidedby
Barriers to Trade (TBT) Committee.
the superior articulating interfaces, determined by fixing the
inferior surface of the mobile bearing component during
testing.
This test method is under the jurisdiction ofASTM Committee F04 on Medical
and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.22 on Arthroplasty. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved June 15, 2020. Published August 2020. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1989. Last previous edition approved in 2014 as F1223 – 14. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F1223-20. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1223−20
3.1.10 flexion angle, n—the angulation of the femoral com- 3.1.26 superior articulating interfaces, n—any interface in
ponent (about an axis parallel to the y-axis) from the fully whichrelativemotionoccursbetweenthetopsideofthemobile
extended knee position to a position in which a “local” vertical bearing component and the femoral bearing component.
axis on the component points posteriorly.
3.1.27 tibial eminence, n—a raised geometrical feature
3.1.10.1 Discussion—For many implants, 0° of flexion can
separating the tibial condyles.
be defined as when the undersurface of the tibial component is
3.1.28 tibial side constraint, n—that constraint provided by
parallel to the femoral component surface that in vivo contacts
the inferior articulating interface.
the most distal surface of the femur.This technique may not be
3.1.29 valgus-varus constraint, n—degree of relative angu-
possibleforsomeimplantsthataredesignedtohaveaposterior
lar motion allowed between the femoral and tibial components
tilt of the tibial component. In these cases, the user shall
of post-in-well (or similar) designs in the coronal plane.
specify how the 0° of flexion position was defined.
3.2 Definitions of Terms Specific to This Standard:
3.1.11 hinge, n—a mechanical physical coupling between
3.2.1 constraint, n—the relative inability of a TKR to be
femoral and tibial components which provides a single axis
further displaced in a specific direction under a given set of
about which flexion occurs.
loading conditions as dictated by the TKR’s geometrical
3.1.12 hyperextension stop, n—a geometrical feature which
design. This motion is limited, as defined in this test, to the
arrests further progress of flexion angles of negative value.
available articular or bearing surfaces found on the tibial
3.1.13 inferior articulating interfaces, n—any interface in
component.Theactualrelativemotionvaluesshallbeprovided
which relative motion occurs between the underside of the
as indicators of this type of constraint.
mobile bearing component and the tibial tray.
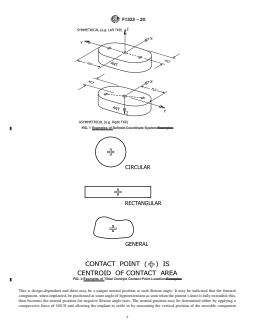
3.2.2 coordinate system (see Fig. 1),n—a set of Cartesian
3.1.14 internal-external rotation, n—the relative angulation
axes affixed to the stationary component.
of the moveable component about an axis parallel to the z-axis.
3.2.2.1 Discussion—Examples of symmetrical and asym-
3.1.15 joint reaction force, n—the applied load whose vec-
metrical designs are shown in Fig. 1.
tor is directed parallel to and collinear with the z-axis,
3.2.2.2 Discussion—The y-axis is parallel to the ML
generally considered parallel to tibial longitudinal axis.
direction, directed medially, and is coincident with the mated
3.1.15.1 Discussion—The applied load may be optionally
components’ contact points when the knee is in the neutral
offset medially yet continuing to be parallel to the z-axis.
position (see 7.2). The z-axis is located midway between the
mated components’ contact points (or in the case of a single
3.1.16 medio-lateral (ML), adj—referring to the orientation
contact point, located at that point) and aligned in the superior-
that is aligned with the y-axis in the defined coordinate system.
inferior direction of the distal component. A third axis, x,
3.1.17 ML condylar radius, n—the geometrical curvature of
mutually orthogonal to the two previous axes is directed
the component’s condyle in the frontal plane.
posteriorly. For determination of contact points, see AnnexA1
3.1.18 ML dimension, n—any geometrical length aligned
and Fig. 2. The contact point shall be located to a tolerance of
with the ML orientation.
61 mm. In the case of multiple contact points on a condyle, an
3.1.19 ML displacement, n—the relative linear translation
average location of the contact points shall be used.
between components in the ML direction.
3.1.20 ML shear load, n—the force applied to the moveable
component with its vector aligned in the ML direction and
causing or intending to cause an ML displacement.
3.1.21 mobile bearing component, n—the ultra-high mo-
lecular weight polyethylene (UHMWPE) component that, by
design, articulates against both the femoral bearing and the
tibial tray.
3.1.22 mobile bearing knee system, n—a knee prosthesis
system, comprised of a tibial component, a mobile bearing
component that can rotate or rotate and translate relative to the
tibial component, and a femoral component.
3.1.23 post-in-well feature, n—aTKR design which tends to
influence kinematics through the coupling of a prominent
eminence with a recess or housing in a mating component.
3.1.24 rotary laxity (RL),n—degree of relative angular
motionpermittedforamoveablecomponentaboutthez-axisas
governed by inherent geometry and load conditions.
3.1.25 rotary torque, n—the moment applied to the move-
ablecomponentwithitsvectoralignedtoanaxisparalleltothe
z-axis and causing or intending to cause an internal or external
rotation. FIG. 1Examples of Defined Coordinate System
F1223−20
3.2.5 set point, n—that numeric quantity assigned to an
input, such as a load.
3.2.6 movable component, n—that component identified
either through design or test equipment attributes as providing
the actual relative motion values.
3.2.6.1 Discussion—Depending upon the user’s fixtures and
the stationary component, it can be either the tibial or femoral
component.
3.2.7 stationary component, n—that component identified
either through design or test equipment attributes as being at
rest during that test and to which actual relative motion values
are referenced.
3.3 Symbols: Parameters:
3.3.1 TAP—overallAPtibial surface dimension (see Fig. 3).
3.3.2 TML—overall MLtibial surface dimension (see Fig. 4
and Fig. 5).
3.3.3 x, y, z—axes of neutral position coordinate system as
defined in Annex A1 and Fig. 1.
3.3.4 DIST—a “yes/no” response to distraction test at the
reported angle at which distraction is most likely to occur.
4. Significance and Use
4.1 This test method, when applied to available products
and proposed prototypes, is meant to provide a database of
product functionality capabilities (in light of the suggested test
regimens)thatishopedwillaidthephysicianinmakingamore
informed total knee replacement (TKR) selection.
4.2 A proper matching of TKR functional restorative capa-
FIG. 2Examples of Tibial Condyle Contact Point Location
bilities and the recipient’s (patient’s) needs is more likely to be
provided by a rational testing protocol of the implant in an
effort to reveal certain device characteristics pertinent to the
3.2.3 degrees of freedom, n—although the knee joint is
selection process.
noted to have 6 degrees of freedom (df), or directions in which
4.3 The TKR product designs are varied and offer a wide
relative motion is guided (three translations: AP, ML, vertical;
range of constraint (stability).The constraint of theTKR in the
three angulations: flexion, internal-external rotation, valgus-
in vitro condition depends on several geometrical and kine-
varus), the coupling effects due to geometrical features reduce
matic interactions among the implant’s components which can
this number to five which are the bases of this test method:AP
be identified and quantified. The degree of TKR’s kinematic
draw, ML shear, internal-external rotation, valgus-varus
interactions should correspond to the recipient’s needs as
rotation, and distraction.
determined by the physician during clinical examination.
3.2.4 neutral position (see 7.2),n—that position in which
the TKR is at rest with no relative linear or angular displace-
ments between components.
3.2.4.1 Discussion—This is design-dependent and there
may be a unique neutral position at each flexion angle. It may
be indicated that the femoral component, when implanted, be
positioned at some angle of hyperextension as seen when the
patient’s knee is fully extended; this, then becomes the neutral
position for negative flexion angle tests. The neutral position
may be determined either by applying a compressive force of
100 N and allowing the implant to settle or by measuring the
vertical position of the movable component with respect to the
stationary and using the low point of the component as the
neutral point. In those implants with a flat zone and no unique
low point, the midpoint of the flat zone can be used as the
neutral point. For those implants having a tibial component
NOTE 1—Sagittal plane section view. Cut taken through the condylar
with a posterior tilt, the user may use other means to define the
contact points. Anterior is to the left.
neutral point, but shall report on how it was found. FIG. 3AP Dimensions and Displacement Examples
F1223−20
cific tests shall be provided with a means of gradually applying
the load or torque to the set point of that test.
5.1.4 Displacement sensing devices shall be arranged so as
to measure relative motion between components in accordance
with the prescribed coordinate system.
5.1.5 Output graphs depicting the relationship of load and
displacement are required (see Fig. 6).
5.1.6 The moveable component shall be mounted on a
fixture with near zero friction or the effect of that friction shall
be subtracted from the applied force.
5.1.7 Tibial Tray Alignment—The tibial tray shall be
NOTE 1—Coronal plane section view. Cut taken though the condylar
mountedtoreflecttherecommendedamountofposteriorslope,
contact points. Medial is to the left.
if any.
FIG. 4ML Dimensions and Displacements Example
5.1.8 The femoral component alignment shall be mounted
accordingtothemanufacturer’sspecifications,suchthatduring
flexion both femoral condyles are in contact with the tibial
4.4 For mobile bearing knee systems, the constraint of the
condyles.
entire implant construct shall be characterized. Constraint of
5.2 Antero-Posterior Draw Test—The movable component
mobile bearings is dictated by design features at both the
shall be rigidly set in a fixture free to move in linear directions
inferior and superior articulating interfaces.
parallel to the x-axis only.
4.5 The methodology, utility, and limitations of constraint/
3,4
5.3 Medio-Lateral Shear Test—The movable component
laxity testing are discussed. The authors recognize that
shall be rigidly set in a fixture free to move in linear directions
evaluating isolated implants (that is, without soft tissues) does
parallel to the y-axis only.
not directly predict in vivo behavior, but will allow compari-
sons among designs. Constraint testing is also useful for
5.4 Rotary Laxity Test—The movable component shall be
characterizing implant performance at extreme ranges of mo-
rigidly set in a fixture free to move in angular displacements
tion which may be encountered in vivo at varying frequencies,
about an axis parallel to the z-axis only.
depending on the patient’s anatomy, pre-operative capability,
5.5 Distraction Test:
and post-operative activities and lifestyle.
5.5.1 The movable component shall be rigidly set in a
fixture free to move in only those directions tending to permit
5. Apparatus
such distraction. Should distraction be possible at more than
5.1 General:
one angle of flexion the test should be conducted at that angle
5.1.1 Thestationarycomponentshouldbefreetomoveonly
which would most likely permit the distraction.
in directions parallel to the z-axis and not permitted to rotate
5.5.2 The stationary component shall be rigidly set in a
about this axis in all but the distraction test. In the distraction
fixture or its position shall be rigidly simulated by the test
test it is fully fixed.
machine’s control system such that the stationary component is
not permitted to move in those directions allowed to the
NOTE 1—In order to test asymmetrical designs, which may be asym-
metrical about the sagittal or frontal planes, it may be necessary to allow movable component.
additional degrees of freedom in addition to those discussed in 5.1, 5.2,
5.6 Valgus-Varus Test:
5.3, and 5.4. For example, the anterior ridge of the tibial bearing insert
5.6.1 Install the tibial component in a fixture in which it is
may be thicker than the posterior ridge. Also the medial and lateral
surfaces may not be identical. As a result of this implant asymmetry,
either completely fixed, or free to translate linearly in a
condylar liftoff may occur. For example, during a rotary test, one may
medial-lateral (y) direction and anterior-posterior (x) direction.
need to allow valgus/varus angulation to ensure that both condyles remain
5.6.2 Install the femoral component in a fixture such that it
in contact. If one does allow additional degree(s) of freedom, these
is free to rotate about an axis parallel to the x-axis. If the tibial
changes to the test method shall be included in the report. For the
internal/external rotation test, asymmetrical designs may also require a component is fixed, then the femoral component shall be free
different center of rotation than as defined in Ssection 3 and AnnexA1.If
to translate medial laterally and anterior posteriorly. The
a different center of rotation is used, it shall be stated in the report section.
femoralcomponentshallbefreetoliftoffofonecondylewhile
5.1.2 The movable component shall be the displaced mem- the other condyle remains in contact.
ber when under loads specific to that test and shall be
6. Test Specimens
instrumented accordingly to obtain data pertinent to that test.
5.1.3 Load or torque actuators producing input vectors
6.1 TKR Specimens:
which tend to displace the movable component relative to the
6.1.1 The TKR should be the manufacturer’s designated
stationary component according to the guidelines of the spe-
“standard” or “medium” size as this is more suitable to the
loading regimes encountered in the tests.
6.1.2 Theimplantshallbeprocuredinitsoriginalpackaging
Walker PS, Haider H, “Characterizing the Motion of Total Knee Replacements
as supplied to the user by the manufacturer.
in Laboratory Tests,” Clin. Ortho. Rel. Res., 410, 2003, pp. 54–68.
6.1.3 If the implant is not available in its package state, the
Haider H, Walker PS, Measurements of Constraint of Total Knee Replacement,
Journal of Biomechanics, Vol. 38, No. 2, 2005, pp. 341–348. condition of the device shall meet all geometrical and material
F1223−20
NOTE 1—Transverse plane section view. Center of rotation for rotary
laxity test is about the neutral point.
FIG. 5Example of Neutral Point Determination
7. Sample Measurement
7.1 General—The constraint values refer to the relative
ability of the components to be displaced under the loads
applied while guided by the geometrical features inherent in
the component design. These features are herein identified as
being based solely on bearing surfaces, although certain
designs offer enhanced constraint (stability) due to other
structures. The tibial bearing surfaces are used as a reference
for relative displacement since the components should not
move beyond the limits of these features, this being disarticu-
lation.
7.2 Neutral Position (NP)—The neutral position is used as
the initial at-rest condition prior to any test as defined in 3.2.4.
It also corresponds to the measurement coordinate system
which is affixed to the stationary component and aligned along
anatomical planes. The location of the origin of the coordinate
system shall be determined as in A
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F1223 − 14 F1223 − 20
Standard Test Method for
Determination of Total Knee Replacement Constraint
This standard is issued under the fixed designation F1223; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the establishment of a database of total knee replacement (TKR) motion characteristics may be used
to compare the constraint characteristics of total knee replacements (TKRs) with the intent of developing guidelines for the
assignment of constraint criteria to TKR designs. (See the Rationale incomparing new designs to existing clinically successful
designs or to determine the constraint differences Appendix X1.)between two similar or dissimilar designs.
1.2 This test method covers the means by which a TKR constraint may be quantified according to motion delineated by the
inherent articular design as determined under specific loading conditions in an in vitroin-vitro environment.
1.3 Tests deemed applicable to the constraint determination are antero-posterior draw, medio-lateral shear, rotary laxity,
valgus-varus rotation, and distraction, as applicable. Also covered is the identification of geometrical parameters of the contacting
surfaces which would influence this motion and the means of reporting the test results. (See Practices E4.)
1.4 This test method is not a wear test.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Verification of Testing Machines
F2083 Specification for Knee Replacement Prosthesis
3. Terminology
3.1 Definitions—Items in this category refer to the geometrical and kinematic aspects of TKR designs as they relate to their
human counterparts:
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.22 on Arthroplasty.
Current edition approved May 15, 2014June 15, 2020. Published June 2014August 2020. Originally approved in 1989. Last previous edition approved in 20122014 as
F1223 – 08 (2012).F1223 – 14. DOI: 10.1520/F1223-14.10.1520/F1223-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1223 − 20
3.1.1 anterior curvature, n—a condylar design which is generally planar except for a concave—upward region anteriorly on the
tibial component.
3.1.2 anterior posterior (AP),n—adj—any geometrical length aligned with the AP orientation.
3.1.3 AP displacement, n—the relative linear translation between components in the AP direction.
3.1.4 AP draw load, n—the force applied to the movable component with its vector aligned in the AP direction causing or
intending to cause an AP displacement.
3.1.5 biconcave, n—a condylar design with pronounced AP and ML condylar radii seen as a “dish” in the tibial component or a
“toroid” in the femoral component.
3.1.6 bearing surface, n—those regions of the component which are intended to contact its counterpart for load transmission.
3.1.7 condyles,condyle, n—entity designed to emulate the joint anatomy and used as a bearing surface primarily for transmission
of the joint reaction force with geometrical properties which tend to govern the general kinematics of the TKR.
3.1.8 distraction, n—the separation of the femoral component(s) from the tibial component(s) in the z-direction.
3.1.9 femoral side constraint, n—that constraint provided by the superior articulating interfaces, determined by fixing the inferior
surface of the mobile bearing component during testing.
3.1.10 flexion angle, n—the angulation of the femoral component (about an axis parallel to the y-axis) from the fully extended knee
position to a position in which a “local” vertical axis on the component now points posteriorly.
3.1.10.1 Discussion—
For many implants, 0° of flexion can be defined as when the undersurface of the tibial component is parallel to the femoral
component surface that in vivo contacts the most distal surface of the femur. This technique may not be possible for some implants
that are designed to have a posterior tilt of the tibial component. In these cases, the user shall specify how the 0° of flexion position
was defined.
3.1.11 hinge, n—a mechanical physical coupling between femoral and tibial components which provides a single axis about which
flexion occurs.
3.1.12 hyperextension stop, n—a geometrical feature which arrests further progress of flexion angles of negative value.
3.1.13 inferior articulating interfaces, n—any interface in which relative motion occurs between the underside of the mobile
bearing component and the tibial tray.
3.1.14 internal-external rotation, n—the relative angulation of the moveable component about an axis parallel to the z-axis.
3.1.15 joint reaction force, n—the applied load whose vector is directed parallel to and collinear with the z-axis, generally
considered parallel to tibial longitudinal axis.
3.1.15.1 Discussion—
The applied load may be optionally offset medially yet continuing to be parallel to the z-axis.
3.1.16 medio-lateral (ML),n—adj—referring to the orientation that is aligned with the y-axis in the defined coordinate system.
3.1.17 ML condylar radius, n—the geometrical curvature of the component’s condyle in the frontal plane.
3.1.18 ML dimension, n—any geometrical length aligned with the ML orientation.
3.1.19 ML displacement, n—the relative linear translation between components in the ML direction.
F1223 − 20
3.1.20 ML shear load, n—the force applied to the moveable component with its vector aligned in the ML direction and causing
or intending to cause an ML displacement.
3.1.21 mobile bearing component, n—the ultra-high molecular weight polyethylene (UHMWPE) component that, by design,
articulates against both the femoral bearing and the tibial tray.
3.1.22 mobile bearing knee system, n—a knee prosthesis system, comprised of a tibial component, a mobile bearing component
that can rotate or rotate and translate relative to the tibial component, and a femoral component.
3.1.23 post-in-well feature, n—a TKR design which tends to influence kinematics through the coupling of a prominent eminence
with a recess or housing in a mating component.
3.1.24 rotary laxity (RL),n—degree of relative angular motion permitted for a moveable component about the z-axis as governed
by inherent geometry and load conditions.
3.1.25 rotary torque, n—the moment applied to the moveable component with its vector aligned to an axis parallel to the z-axis
and causing or intending to cause an internal or external rotation.
3.1.26 superior articulating interfaces, n—any interface in which relative motion occurs between the topside of the mobile bearing
component and the femoral bearing component.
3.1.27 tibial eminence, n—a raised geometrical feature separating the tibial condyles.
3.1.28 tibial side constraint, n—that constraint provided by the inferior articulating interface.
3.1.29 valgus-varus constraint, n—degree of relative angular motion allowed between the femoral and tibial components of
post-in-well designs (or similar designs)similar) designs in the coronal plane.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 constraint, n—the relative inability of a TKR to be further displaced in a specific direction under a given set of loading
conditions as dictated by the TKR’s geometrical design. This motion is limited, as defined in this test, to the available articular
or bearing surfaces found on the tibial component. The actual relative motion values shall be provided as indicators of this type
of constraint.
3.2.2 coordinate system (see Fig. 1),n—a set of arbitrary cartesian coordinates Cartesian axes affixed to the stationary component
and aligned such that the origin is located at the intersection of the component.y and z axes.
3.2.2.1 Discussion—
Examples of symmetrical and asymmetrical designs are shown in Fig. 1.
3.2.2.2 Discussion—
The y-axis is parallel to the ML direction, directed medially, and is coincident with the mated components’ contact points when
the knee is in the neutral position (see 7.2). The z-axis is located midway between the mated components’ contact points (or in
the case of a single contact point, located at that point) and aligned in the superior-inferior direction of the distal component. A
third axis, x, mutually orthogonal to the two previous axes is directed posteriorly. For determination of contact points, see Annex
A1 and Fig. 2. The contact point shall be located to a tolerance of 61 mm. In the case of multiple contact points on a condyle,
an average location of the contact points shall be used.
3.2.3 degrees of freedom, n—although the knee joint is noted to have 6 df, degrees of freedom (df), or directions in which relative
motion is guided (three translations: AP, ML, vertical; three angulations: flexion, internal-external rotation, valgus-varus), the
coupling effects due to geometrical features reduce this number to five which are the bases of this test method: AP draw, ML shear,
internal-external rotation, valgus-varus rotation, and distraction.
3.2.4 neutral position (see 7.2),n—that position in which the TKR is at rest with no relative linear or angular displacements
between components.
3.2.4.1 Discussion—
F1223 − 20
FIG. 1 Examples of Defined Coordinate System Examples
FIG. 2 Examples of Tibial Condyle Contact Point Location Examples
This is design-dependent and there may be a unique neutral position at each flexion angle. It may be indicated that the femoral
component, when implanted, be positioned at some angle of hyperextension as seen when the patient’s knee is fully extended; this,
then becomes the neutral position for negative flexion angle tests. The neutral position may be determined either by applying a
compressive force of 100 N and allowing the implant to settle or by measuring the vertical position of the movable component
F1223 − 20
with respect to the stationary and using the low point of the component as the neutral point. In those implants with a flat zone and
no unique low point, the midpoint of the flat zone can be used as the neutral point. For those implants having a tibial component
with a posterior tilt, the user may use other means to define the neutral point, but shall report on how it was found.
3.2.5 set point, n—that numeric quantity assigned to an input, such as a load.
3.2.6 movable component, n—that component identified either through design or test equipment attributes as providing the actual
relative motion values.
3.2.6.1 Discussion—
Depending upon the user’s fixtures and the stationary component, it can be either the tibial or femoral component.
3.2.7 stationary component, n—that component identified either through design or test equipment attributes as being at rest during
that test and to which actual relative motion values are referenced.
3.3 Symbols: Parameters:
3.3.1 TAP—overall AP tibial surface dimension.dimension (see Fig. 3).
3.3.2 TML—overall ML tibial surface dimension.dimension (see Fig. 4 and Fig. 5).
3.3.3 x, y, z—axes of neutral position coordinate system as defined in Annex A1 and Fig. 1.
3.3.4 DIST—a “yes/no” response to distraction test at the reported angle at which distraction is most likely to occur.
4. Significance and Use
4.1 This test method, when applied to available products and proposed prototypes, is meant to provide a database of product
functionality capabilities (in light of the suggested test regimens) that is hoped will aid the physician in making a more informed
total knee replacement (TKR) selection.
4.2 A proper matching of TKR functional restorative capabilities and the recipient’s (patient’s) needs is more likely to be provided
by a rational testing protocol of the implant in an effort to reveal certain device characteristics pertinent to the selection process.
4.3 The TKR product designs are varied and offer a wide range of constraint (stability). The constraint of the TKR in the in vitro
condition depends on several geometrical and kinematic interactions among the implant’s components which can be identified and
quantified. The degree of TKR’s kinematic interactions should correspond to the recipient’s needs as determined by the physician
during clinical examination.
4.4 For mobile bearing knee systems, the constraint of the entire implant construct shall be characterized. Constraint of mobile
bearings is dictated by design features at both the inferior and superior articulating interfaces.
NOTE 1—Sagittal plane section view. Cut taken through the condylar contact points. Anterior is to the left.
FIG. 43 AP Dimensions and Displacement Examples
F1223 − 20
NOTE 1—Coronal plane section view. Cut taken though the condylar contact points. Medial is to the left.
FIG. 54 ML Dimensions and Displacements Example
3,4
4.5 The methodology, utility, and limitations of constraint/laxity testing are discussed. The authors recognize that evaluating
isolated implants (that is, without soft tissues) does not directly predict in vivo behavior, but will allow comparisons among
designs. Constraint testing is also useful for characterizing implant performance at extreme ranges of motion which may be
encountered in vivo at varying frequencies, depending on the patient’s anatomy, pre-operative capability, and post-operative
activities and lifestyle.
5. Apparatus
5.1 General:
5.1.1 The stationary component should be free to move only in directions parallel to the z-axis and not permitted to rotate about
this axis in all but the distraction test. In the distraction test it is fully fixed.
NOTE 1—In order to test asymmetrical designs, which may be asymmetrical about the sagittal or frontal planes, it may be necessary to allow additional
degrees of freedom in addition to those discussed in 5.1, 5.2, 5.3, and 5.4. For example, the anterior ridge of the tibial bearing insert may be thicker than
the posterior ridge. Also the medial and lateral surfaces may not be identical. As a result of this implant asymmetry, condylar liftoff may occur. For
example, during a rotary test, one may need to allow valgus/varus angulation to ensure that both condyles remain in contact. If one does allow additional
degree(s) of freedom, these changes to the test method shall be included in the report. For the internal/external rotation test, asymmetrical designs may
also require a different center of rotation than as defined in Ssection 3 and Annex A1. If a different center of rotation is used, it shall be stated in the report
section.
5.1.2 The movable component shall be the displaced member when under loads specific to that test and shall be instrumented
accordingly to obtain data pertinent to that test.
5.1.3 Load or torque actuators producing input vectors which tend to displace the movable component relative to the stationary
component according to the guidelines of the specific tests shall be provided with a means of gradually applying the load or torque
to the set point of that test.
5.1.4 Displacement sensing devices shall be arranged so as to measure relative motion between components in accordance with
the prescribed coordinate system.
5.1.5 Output graphs depicting the relationship of load and displacement are required (see Fig. 36).
5.1.6 The moveable component shall be mounted on a fixture with near zero friction or the effect of that friction shall be subtracted
from the applied force.
5.1.7 Tibial Tray Alignment—The tibial tray shall be mounted to reflect the recommended amount of posterior slope, if any.
5.1.8 The femoral component alignment shall be mounted according to the manufacturer’s specifications, such that during flexion
both femoral condyles are in contact with the tibial condyles.
Walker PS, Haider H, “Characterizing the Motion of Total Knee Replacements in Laboratory Tests,” Clin. Ortho. Rel. Res., 410, 2003, pp. 54–68.
Haider H, Walker PS, Measurements of Constraint of Total Knee Replacement, Journal of Biomechanics, Vol. 38, No. 2, 2005, pp. 341–348.
F1223 − 20
NOTE 1—Transverse plane section view. Center of rotation for rotary NOTE 1—Transverse plane section view. Center of rotation for rotary
laxity test is about the neutral point. laxity test is about the neutral point.
NOTE 1—Transverse plane section view. Center of rotation for rotary
laxity test is about the neutral point.
FIG. 65 A,B Example of Neutral Point Determination Example
FIG. 36 Output Graph Examplegraph example showing AP displacement as a result of AP loading
NOTE 1—This figure corresponds to 3 cycles of loading. The graph’s y-axis is AP force and the graph’s x-axis is AP displacement. This figure
demonstrates the hysteresis effect that occurs due to loading in each direction.
5.2 Antero-Posterior Draw Test—The movable component shall be rigidly set in a fixture free to move in linear directions parallel
to the x-axis only.
5.3 Medio-Lateral Shear Test—The movable component shall be rigidly set in a fixture free to move in linear directions parallel
to the y-axis only.
F1223 − 20
5.4 Rotary Laxity Test—The movable component shall be rigidly set in a fixture free to move in angular displacements about an
axis parallel to the z-axis only.
5.5 Distraction Test:
5.5.1 The movable component shall be rigidly set in a fixture free to move in only those directions tending to permit such
distraction. Should distraction be possible at more than one angle of flexion the test should be conducted at that angle which would
most likely permit the distraction.
5.5.2 The stationary component shall be rigidly set in a fixture and or its position shall be rigidly simulated by the test machine’s
control system such that the stationary component is not permitted to move in those directions allowed to the movable component.
5.6 Valgus-Varus Test:
5.6.1 Install the tibial component in a fixture in which it is either completely fixed, or free to translate linearly in a medial-lateral
(y) direction and anterior-posterior (x) direction.
5.6.2 Install the femoral component in a fixture such that it is free to rotate in the coronal (about an axis parallel to the yzx ) plane.
-axis. If the tibial component is fixed, then the femoral component shall be free to translate medial laterally and anterior posteriorly.
The femoral component shall be free to lift off of one condyle while the other condyle remains in contact.
6. Test Specimens
6.1 TKR Specimens:
6.1.1 The TKR should be the manufacturer’s designated “standard” or “medium” size as this is more suitable to the loading
regimes encountered in the tests.
6.1.2 The implant shall be procured in its original packaging as supplied to the user by the manufacturer.
6.1.3 If the implant is not available in its package state, the condition of the device shall meet all geometrical and material
specifications, but may contain slight surface irregularities (that is, “cosmetic rejects”) not considered influential in those regions
of the device deemed critical to the specific test.
6.2 TKR Prototype—The implant shall be of quality as in 6.1.3.
7. Sample Measurement
7.1 General—The constraint values refer to the relative ability of the components to be displaced under the loads applied while
guided by the geometrical features inherent in the component design. These features are herein identifi
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...