ASTM F2978-20
(Guide)Standards Guide to Optimize Scan Sequences for Clinical Diagnostic Evaluation of Metal-on-Metal Hip Arthroplasty Devices using Magnetic Resonance Imaging
Standards Guide to Optimize Scan Sequences for Clinical Diagnostic Evaluation of Metal-on-Metal Hip Arthroplasty Devices using Magnetic Resonance Imaging
SIGNIFICANCE AND USE
5.1 Magnetic resonance imaging is ideally suited to image MOM hip arthroplasty due to its superior soft tissue contrast, multiplanar capabilities and lack of ionizing radiation. MR imaging is the most accurate imaging modality for the assessment of peri-prosthetic osteolysis and wear-induced synovitis (19, 20).
5.2 Before scanning a patient with a specific implant, the MR practitioner shall confirm that the device is MR Conditional and that the scan protocol to be used satisfies the conditions for safe scanning for the specific implant.
5.3 This guide can be used to identify the following adverse events.
5.3.1 Osteolysis—Magnetic resonance imaging is superior to conventional radiographs and computer tomography (CT) in the assessment of peri-prosthetic osteolysis and has been shown to be the most accurate method to locate and quantify the extent of peri-prosthetic osteolysis (19, 21). On MR imaging, osteolysis appears as well marginated intraosseous intermediate to slightly increased signal intensity lesions that contrast with the high signal intensity of the intramedullary fat. A characteristic line of low signal intensity surrounds the area of focal marrow replacement, distinguishing the appearance of osteolysis from tumoral replacement of bone or infection (22).
FIG. 4 Coronal (left) and Axial (right) FSE Images of a Left MOM Hip Arthroplasty
Note 1: There is focal osteolysis (white arrows) in the greater trochanter, which manifests as well-demarcated intermediate signal intensity, similar to that of skeletal muscle, replacing the normal high signal intensity fatty marrow. Images courtesy of Dr. Hollis Potter.
5.3.2 Component Loosening—While the data are preliminary, MR imaging can identify circumferential bone resorption that may indicate component loosening. Loosening may result from osteolysis, circumferential fibrous membrane formation or poor osseous integration of a non-cemented component. On MR imaging, component loosening typically manif...
SCOPE
1.1 This guide describes the recommended protocol for magnetic resonance imaging (MRI) studies of patients implanted with metal-on-metal (MOM) devices to determine if the periprosthetic tissues are likely to be associated with an adverse local tissue reaction (ALTR). Before scanning a patient with a specific implant, the MR practitioner shall confirm that the device is MR Conditional and that the scan protocol to be used satisfies the conditions for safe scanning for the specific implant. This guide assumes that the MRI protocol will be applied to MOM devices while they are implanted inside the body. It is also expected that standardized MRI safety measures will be followed during the performance of this scan protocol.
1.2 This guide covers the clinical evaluation of the tissues surrounding MOM hip replacement devices in patients using MRI. This guide is applicable to both total and resurfacing MOM hip systems.
1.3 The protocol contained in this guide applies to whole body magnetic resonance equipment, as defined in section 201.3.239 of IEC 60601-2-33, Ed. 3.2, with a whole body radiofrequency (RF) transmit coil as defined in section 201.3.240. The RF coil should have circulary polarized RF excitation (also commonly referred to as quadrature excitation) as defined in section 201.3.249 of IEC 60601-2-33, Ed. 3.2..
1.4 The values stated in SI units are to be regarded as standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. The user may consider all precautions and warnings provided in the MR system and hip implant labeling prior to determining the applicability of these protocols.
1.6 This international standard was developed in accordance with internatio...
General Information
- Status
- Published
- Publication Date
- 31-Aug-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Apr-2023
- Effective Date
- 15-Oct-2019
- Effective Date
- 15-Jun-2019
- Effective Date
- 15-Feb-2019
- Effective Date
- 01-Jun-2018
- Effective Date
- 15-Oct-2017
- Effective Date
- 01-Jul-2017
- Effective Date
- 01-May-2016
- Effective Date
- 01-May-2016
- Effective Date
- 01-Oct-2015
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-Jun-2013
- Refers
ASTM A340-03a(2011) - Standard Terminology of Symbols and Definitions Relating to Magnetic Testing - Effective Date
- 01-May-2011
- Effective Date
- 01-Oct-2008
Overview
ASTM F2978-20: Standards Guide to Optimize Scan Sequences for Clinical Diagnostic Evaluation of Metal-on-Metal Hip Arthroplasty Devices using Magnetic Resonance Imaging provides best practices for performing MRI on patients with metal-on-metal (MOM) hip implants. As an internationally recognized guide, it helps clinicians and imaging professionals enhance diagnostic accuracy while maintaining patient safety by detailing appropriate scan protocols and safety checks for both total and resurfacing MOM hip systems.
MRI is highly effective for evaluating peri-prosthetic tissues, given its superior soft tissue contrast, multiplanar imaging capabilities, and absence of ionizing radiation. The standard addresses how to recognize and assess adverse local tissue reactions, osteolysis, component loosening, wear-induced synovitis, infection, and other complications associated with MOM hip arthroplasty.
Key Topics
MR Conditional Assessment
Before scanning, practitioners must verify that the specific hip implant is MR Conditional and that the chosen protocol adheres to manufacturer safety guidelines.Recommended MRI Protocol
- Use of surface coil fast spin echo (FSE) sequences in three planes
- Inclusion of large field-of-view (FOV) short tau inversion recovery (STIR) sequences for broader pelvic assessment
- Optimization of scan parameters to reduce metal artifact, including usage of high receiver bandwidth and thinner slices
- Application of advanced metal artifact reduction techniques, such as MAVRIC and SEMAC, when available
Recognition of Adverse Events
The guide details imaging findings for identifying:- Peri-prosthetic osteolysis
- Component loosening
- Wear-induced synovitis
- Infections
- Adverse local tissue reactions (ALTR), including mass lesions or pseudotumors
- Signs of modular taper-associated ALTR
Reporting Standards
Radiology reports should describe the presence, severity, and distribution of:- Synovitis and solid synovial deposits
- Decompression into adjacent bursae
- Extracapsular disease
- Infection markers
- Osteolysis and loosening
- Any neurovascular or tendinous complications
Applications
Clinical Diagnostic Imaging
Clinicians rely on this standard for the safe, accurate diagnostic assessment of patients with MOM hip replacements. The imaging protocol enhances early detection of complications such as osteolysis, synovitis, and implant loosening.Pre- and Post-Operative Evaluation
The guide supports both immediate and delayed postoperative monitoring, offering critical information for timely intervention and revision planning.Safety Compliance
By following ASTM F2978-20, imaging centers ensure adherence to safety practices and regulatory considerations, minimizing risk for patients with metal implants.Research and Device Evaluation
Consistency in scan protocols facilitates reliable data for clinical studies and post-market surveillance of MOM hip devices.
Related Standards
- ASTM F2503 – Practice for Marking Medical Devices and Other Items for Safety in the Magnetic Resonance Environment
- IEC 60601-2-33 – Medical electrical equipment - Particular requirements for the safety of magnetic resonance equipment for medical diagnosis
- ASTM A340 – Terminology of Symbols and Definitions Relating to Magnetic Testing
Key search terms: metal-on-metal hip arthroplasty MRI protocol, MRI artifact reduction, peri-prosthetic tissue imaging, MR Conditional implants, adverse local tissue reaction imaging, ASTM F2978-20 compliance.
By implementing the guidance in ASTM F2978-20, healthcare professionals optimize MRI procedures for MOM hip arthroplasty, ensuring both safety and diagnostic precision for the evaluation of orthopedic implants.
Buy Documents
ASTM F2978-20 - Standards Guide to Optimize Scan Sequences for Clinical Diagnostic Evaluation of Metal-on-Metal Hip Arthroplasty Devices using Magnetic Resonance Imaging
REDLINE ASTM F2978-20 - Standards Guide to Optimize Scan Sequences for Clinical Diagnostic Evaluation of Metal-on-Metal Hip Arthroplasty Devices using Magnetic Resonance Imaging
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2978-20 is a guide published by ASTM International. Its full title is "Standards Guide to Optimize Scan Sequences for Clinical Diagnostic Evaluation of Metal-on-Metal Hip Arthroplasty Devices using Magnetic Resonance Imaging". This standard covers: SIGNIFICANCE AND USE 5.1 Magnetic resonance imaging is ideally suited to image MOM hip arthroplasty due to its superior soft tissue contrast, multiplanar capabilities and lack of ionizing radiation. MR imaging is the most accurate imaging modality for the assessment of peri-prosthetic osteolysis and wear-induced synovitis (19, 20). 5.2 Before scanning a patient with a specific implant, the MR practitioner shall confirm that the device is MR Conditional and that the scan protocol to be used satisfies the conditions for safe scanning for the specific implant. 5.3 This guide can be used to identify the following adverse events. 5.3.1 Osteolysis—Magnetic resonance imaging is superior to conventional radiographs and computer tomography (CT) in the assessment of peri-prosthetic osteolysis and has been shown to be the most accurate method to locate and quantify the extent of peri-prosthetic osteolysis (19, 21). On MR imaging, osteolysis appears as well marginated intraosseous intermediate to slightly increased signal intensity lesions that contrast with the high signal intensity of the intramedullary fat. A characteristic line of low signal intensity surrounds the area of focal marrow replacement, distinguishing the appearance of osteolysis from tumoral replacement of bone or infection (22). FIG. 4 Coronal (left) and Axial (right) FSE Images of a Left MOM Hip Arthroplasty Note 1: There is focal osteolysis (white arrows) in the greater trochanter, which manifests as well-demarcated intermediate signal intensity, similar to that of skeletal muscle, replacing the normal high signal intensity fatty marrow. Images courtesy of Dr. Hollis Potter. 5.3.2 Component Loosening—While the data are preliminary, MR imaging can identify circumferential bone resorption that may indicate component loosening. Loosening may result from osteolysis, circumferential fibrous membrane formation or poor osseous integration of a non-cemented component. On MR imaging, component loosening typically manif... SCOPE 1.1 This guide describes the recommended protocol for magnetic resonance imaging (MRI) studies of patients implanted with metal-on-metal (MOM) devices to determine if the periprosthetic tissues are likely to be associated with an adverse local tissue reaction (ALTR). Before scanning a patient with a specific implant, the MR practitioner shall confirm that the device is MR Conditional and that the scan protocol to be used satisfies the conditions for safe scanning for the specific implant. This guide assumes that the MRI protocol will be applied to MOM devices while they are implanted inside the body. It is also expected that standardized MRI safety measures will be followed during the performance of this scan protocol. 1.2 This guide covers the clinical evaluation of the tissues surrounding MOM hip replacement devices in patients using MRI. This guide is applicable to both total and resurfacing MOM hip systems. 1.3 The protocol contained in this guide applies to whole body magnetic resonance equipment, as defined in section 201.3.239 of IEC 60601-2-33, Ed. 3.2, with a whole body radiofrequency (RF) transmit coil as defined in section 201.3.240. The RF coil should have circulary polarized RF excitation (also commonly referred to as quadrature excitation) as defined in section 201.3.249 of IEC 60601-2-33, Ed. 3.2.. 1.4 The values stated in SI units are to be regarded as standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. The user may consider all precautions and warnings provided in the MR system and hip implant labeling prior to determining the applicability of these protocols. 1.6 This international standard was developed in accordance with internatio...
SIGNIFICANCE AND USE 5.1 Magnetic resonance imaging is ideally suited to image MOM hip arthroplasty due to its superior soft tissue contrast, multiplanar capabilities and lack of ionizing radiation. MR imaging is the most accurate imaging modality for the assessment of peri-prosthetic osteolysis and wear-induced synovitis (19, 20). 5.2 Before scanning a patient with a specific implant, the MR practitioner shall confirm that the device is MR Conditional and that the scan protocol to be used satisfies the conditions for safe scanning for the specific implant. 5.3 This guide can be used to identify the following adverse events. 5.3.1 Osteolysis—Magnetic resonance imaging is superior to conventional radiographs and computer tomography (CT) in the assessment of peri-prosthetic osteolysis and has been shown to be the most accurate method to locate and quantify the extent of peri-prosthetic osteolysis (19, 21). On MR imaging, osteolysis appears as well marginated intraosseous intermediate to slightly increased signal intensity lesions that contrast with the high signal intensity of the intramedullary fat. A characteristic line of low signal intensity surrounds the area of focal marrow replacement, distinguishing the appearance of osteolysis from tumoral replacement of bone or infection (22). FIG. 4 Coronal (left) and Axial (right) FSE Images of a Left MOM Hip Arthroplasty Note 1: There is focal osteolysis (white arrows) in the greater trochanter, which manifests as well-demarcated intermediate signal intensity, similar to that of skeletal muscle, replacing the normal high signal intensity fatty marrow. Images courtesy of Dr. Hollis Potter. 5.3.2 Component Loosening—While the data are preliminary, MR imaging can identify circumferential bone resorption that may indicate component loosening. Loosening may result from osteolysis, circumferential fibrous membrane formation or poor osseous integration of a non-cemented component. On MR imaging, component loosening typically manif... SCOPE 1.1 This guide describes the recommended protocol for magnetic resonance imaging (MRI) studies of patients implanted with metal-on-metal (MOM) devices to determine if the periprosthetic tissues are likely to be associated with an adverse local tissue reaction (ALTR). Before scanning a patient with a specific implant, the MR practitioner shall confirm that the device is MR Conditional and that the scan protocol to be used satisfies the conditions for safe scanning for the specific implant. This guide assumes that the MRI protocol will be applied to MOM devices while they are implanted inside the body. It is also expected that standardized MRI safety measures will be followed during the performance of this scan protocol. 1.2 This guide covers the clinical evaluation of the tissues surrounding MOM hip replacement devices in patients using MRI. This guide is applicable to both total and resurfacing MOM hip systems. 1.3 The protocol contained in this guide applies to whole body magnetic resonance equipment, as defined in section 201.3.239 of IEC 60601-2-33, Ed. 3.2, with a whole body radiofrequency (RF) transmit coil as defined in section 201.3.240. The RF coil should have circulary polarized RF excitation (also commonly referred to as quadrature excitation) as defined in section 201.3.249 of IEC 60601-2-33, Ed. 3.2.. 1.4 The values stated in SI units are to be regarded as standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. The user may consider all precautions and warnings provided in the MR system and hip implant labeling prior to determining the applicability of these protocols. 1.6 This international standard was developed in accordance with internatio...
ASTM F2978-20 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2978-20 has the following relationships with other standards: It is inter standard links to ASTM A340-23a, ASTM F2503-23e1, ASTM A340-19b, ASTM A340-19a, ASTM A340-19, ASTM A340-18, ASTM A340-17a, ASTM A340-17, ASTM A340-16e1, ASTM A340-16, ASTM A340-15, ASTM A340-14, ASTM F2503-13, ASTM A340-03a(2011), ASTM F2503-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2978-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2978 − 20
Standard Guide to
Optimize Scan Sequences for Clinical Diagnostic Evaluation
of Metal-on-Metal Hip Arthroplasty Devices using Magnetic
Resonance Imaging
This standard is issued under the fixed designation F2978; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope in the MR system and hip implant labeling prior to determining
the applicability of these protocols.
1.1 This guide describes the recommended protocol for
1.6 This international standard was developed in accor-
magnetic resonance imaging (MRI) studies of patients im-
dance with internationally recognized principles on standard-
planted with metal-on-metal (MOM) devices to determine if
ization established in the Decision on Principles for the
the periprosthetic tissues are likely to be associated with an
Development of International Standards, Guides and Recom-
adverselocaltissuereaction(ALTR).Beforescanningapatient
mendations issued by the World Trade Organization Technical
with a specific implant, the MR practitioner shall confirm that
Barriers to Trade (TBT) Committee.
the device is MR Conditional and that the scan protocol to be
used satisfies the conditions for safe scanning for the specific
2. Referenced Documents
implant. This guide assumes that the MRI protocol will be
2.1 ASTM Standards:
applied to MOM devices while they are implanted inside the
A340 Terminology of Symbols and Definitions Relating to
body.ItisalsoexpectedthatstandardizedMRIsafetymeasures
Magnetic Testing
will be followed during the performance of this scan protocol.
F2503 Practice for Marking Medical Devices and Other
1.2 This guide covers the clinical evaluation of the tissues
Items for Safety in the Magnetic Resonance Environment
surrounding MOM hip replacement devices in patients using
2.2 IEC Standard:
MRI. This guide is applicable to both total and resurfacing
IEC 60601-2-33:2010+AMD1:2013+AMD2:2015
MOM hip systems.
CSV Medical electrical equipment—Part 2: Particular re-
quirements for the basic safety and essential performance
1.3 The protocol contained in this guide applies to whole
of magnetic resonance equipment for medical diagnosis,
body magnetic resonance equipment, as defined in section
201.3.239 of IEC 60601-2-33, Ed. 3.2, with a whole body
radiofrequency (RF) transmit coil as defined in section
3. Terminology
201.3.240. The RF coil should have circulary polarized RF
3.1 Definitions—For the purposes of this standard the
excitation (also commonly referred to as quadrature excitation)
following definitions shall apply:
as defined in section 201.3.249 of IEC 60601-2-33, Ed. 3.2.
3.1.1 Magnetic Resonance Imaging (MRI)—diagnostic im-
1.4 The values stated in SI units are to be regarded as
aging technique that uses static and time-varying magnetic
standard.
fields to provide tomographic images of tissue by the magnetic
resonance of nuclei.
1.5 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the 3.1.2 MR-Conditional—an item with demonstrated safety in
responsibility of the user of this standard to establish appro-
theMRenvironmentwithindefinedconditions.Ataminimum,
priate safety, health, and environmental practices and deter-
address the conditions of the static magnetic field, the switched
mine the applicability of regulatory limitations prior to use.
gradient magnetic field and the radiofrequency fields. Addi-
The user may consider all precautions and warnings provided
tional conditions, including specific configurations of the item,
may be required (Practice F2503 – 13).
1 2
This guide is under the jurisdiction of ASTM Committee F04 on Medical and For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Surgical Materials and Devices and is the direct responsibility of Subcommittee contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
F04.22 on Arthroplasty. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Sept. 1, 2020. Published October 2020. Originally the ASTM website.
approved in 2013. Last previous edition approved in 2013 as F2978 – 13. DOI: Available from International Electrotechnical Commission (IEC), 3, rue de
10.1520/F2978-20. Varembé, P.O. Box 131, CH-1211 Geneva 20, Switzerland, http://www.iec.ch.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2978 − 20
3.1.3 Metal-on-Metal (MOM) hip replacement—a hip ar- high-resolution matrix will increase spatial resolution and
throplasty device in which the articulating surfaces of the trabecular detail in the face of the susceptibility artifact.
femoral head and the acetabular cup are fabricated from metal.
However, these techniques will also decrease the signal-to-
noise ratio. Orienting the frequency encoding direction along
4. Summary of Protocol
the long axis of the prosthesis can also be effective in
4.1 Surface coil fast spin echo (FSE), also known as turbo
decreasing artifacts but may not be feasible (4). In addition,
spin echo (TSE), sequences of the affected hip in three planes
view-angle tilting (VAT) gradients can be applied, which
and a larger field-of-view (FOV) short tau inversion recovery
applies a section-selection gradient during the signal readout
(STIR) sequence to include both hips and the surrounding
can be used (5).
pelvis are recommended. A large FOV sequence of the entire
4.4 Techniques to avoid when imaging in the presence of
pelvis should be included to assess for remote causes of pain,
metalincludeimagingathighfieldstrengths,useoffrequency-
such as pelvic or sacral fractures, which may be referred to the
selective fat suppression and use of gradient echo sequences.
hip.
Artifact due to susceptibility is directly proportional to the
4.2 With regards to the FSE, also known as turbo spin echo
main magnetic field (B ); therefore, imaging at field strengths
(TSE), surface coil imaging, an intermediate echo time, water-
of 1.5 T or less are preferable where appropriate. Users should
sensitive fast spin echo technique is effective in highlighting
validate use of higher field strength when applicable to show
osteolysis and detecting wear-induced synovitis. The fluid-
comparison to the 1.5 T for lack of increased artifact suscep-
sensitive inversion recovery sequence helps outline fluid col-
tibility. When fat suppression is required, inversion recovery
lectionsandwilldemonstratethepresenceofmarrowedemain
sequences are preferred over frequency-selective fat suppres-
the setting of implant loosening or peri-prosthetic fracture (1).
sion techniques, as they are less susceptible to magnetic field
4.3 Modifications of standard pulse sequence parameters
inhomogeneities. Standardized gradient echo imaging should
should be applied when imaging in the presence of metallic
be avoided, as these sequences lack the 180° refocusing pulse
implants. Options available to reduce susceptibility artifacts on
of spin echo sequences, resulting in rapid dephasing of spins
routine clinical scanners include increasing the amplitude of
and large areas of signal void in the presence of metal.
the readout gradient by the use of a wider receiver bandwidth
and thinner slices (2, 3). Decreasing voxel size by the use of a
4.5 Table 1 outlines a suggested protocol for imaging MOM
hip arthroplasty using a 1.5 Tesla (T) clinical scanner where
FSEandTSEareconsideredequalforparameterpicks(6).The
The boldface numbers in parentheses refer to a list of references at the end of
use of a 3 T MRI scanner is also an option but may not be
this standard.
A,B
TABLE 1 Suggested Protocol for Metal-on-Metal Hip Arthroplasty Imaging at a 1.5 T MRI Scanner
Timing Axial Coronal Sagittal
Coronal FIR Axial FSE/TSE
Parameters FSE/TSE FSE/TSE FSE/TSE
Body Body Surface Surface Surface
Coil
Coil Coil Coil Coil Coil
TR, msec 4500 – 5500 4500 4500 – 5800 4500 – 5500 5500 – 6500
TE, msec 21.4 - 32.0 18 24 - 30 24 - 30 23 - 30
TI, msec . . . 150 . . . . . . . . .
Echo train length 16 – 20 7 – 9 10 – 20 10 – 20 14 – 20
BW, kHz 83 – 125 83 – 125 83 – 125 83 – 125 83 – 125
FOV,cm 32–36 34–36 18 17–19 18–20
Matrix (or resolution in mm to be 512 × 256 256 × 192 512 × 352 512 × 256 - 288 512 × 352
calculated by user spec)
Slice thickness, mm 5 5 4 4 2.5 – 3
Interslice gap, mm 0 0 0 0 0
Number of averages 4 2 4 4 4
No phase wrap (Fold-over suppression, yes yes yes yes yes
by oversampling)
Swap phase yes yes yes yes yes
and frequency
Variable BW yes yes yes yes yes
Frequency direction anterior to right to right to anterior to anterior to
(read-out direction) posterior left left posterior posterior
A
Abbreviations:
BW – bandwidth.
FIR – fast inversion recovery.
FOV – field of view.
FSE – fast spin echo.
TSE – turbo spin echo.
KHz – kiloHertz.
TE – echo time.
TI – inversion time.
TR – repetition time.
B
Depending on the MRI system, the BW may be reported as half-bandwidth (maximum frequency), so a reported BW of 62.5 is actually acquired at 125 Hz over the entire
frequency range. For Table 1, to convert to Hz/pixel when implementing 512 frequency encoding steps, use the following formula: (kHz x 2000)/512.
F2978 − 20
preferable and the recommendation in 4.4 should be consid- 5. Significance and Use
ered. The protocol should be carefully considered for this
5.1 Magnetic resonance imaging is ideally suited to image
option before proceeding.
MOM hip arthroplasty due to its superior soft tissue contrast,
multiplanar capabilities and lack of ionizing radiation. MR
4.6 Examples of three-dimensional multispectral imaging
imaging is the most accurate imaging modality for the assess-
(3D-MSI) include multi-acquisition variable-resonance image
ment of peri-prosthetic osteolysis and wear-induced synovitis
combination (MAVRIC SL). Fourier transform-based spin-
(19, 20).
warp (WARP) and metal artifact reduction for orthopedic
implants (O-MAR XD) that results in an image with markedly
5.2 Before scanning a patient with a specific implant, the
reduced susceptibility artifact (7-11). All 3D-MSI techniques
MR practitioner shall confirm that the device is MR Condi-
utilize a VAT pulse to decrease frequency-encoding distortions
tional and that the scan protocol to be used satisfies the
and further utilize multiple frequency bins to mitigate through conditions for safe scanning for the specific implant.
plane distortions. Early studies have demonstrated decreased
5.3 This guide can be used to identify the following adverse
image distortion at the bone-implant interface and improved
events.
detection of peri-prosthetic osteolysis and synovitis when
5.3.1 Osteolysis—Magnetic resonance imaging is superior
compared to conventional fast spin echo techniques (12). A
to conventional radiographs and computer tomography (CT) in
published study of patients with either MOM resurfacing or
the assessment of peri-prosthetic osteolysis and has been
MOM total hip arthroplasty demonstrated synovitis using the
shown to be the most accurate method to locate and quantify
MAVRIC sequence in 77.4 % of resurfacing arthroplasty and
the extent of peri-prosthetic osteolysis (19, 21). On MR
86.2 % of total hip arthroplasty (THA) hips (13, 14). For
imaging, osteolysis appears as well marginated intraosseous
imaging MOM arthroplasty, the use of a MAVRIC sequence is
intermediate to slightly increased signal intensity lesions that
recommended in at least one plane (coronal or axial) when this
contrast with the high signal intensity of the intramedullary fat.
sequenceisavailable.Whencombinedwithaninversionpulse,
A characteristic line of low signal intensity surrounds the area
they provide optimal fat suppression in the presence of
of focal marrow replacement, distinguishing the appearance of
implants. Specific parameters are listed in Appendix X2. With
osteolysis from tumoral replacement of bone or infection (22).
regards to timing, these protocols have been successful in
5.3.2 Component Loosening—While the data are
assessing patients for both immediate and delayed
preliminary, MR imaging can identify circumferential bone
complications, including fracture, nerve impingement, and
resorption that may indicate component loosening. Loosening
tendon tears in the immediate post-operative period, and
may result from osteolysis, circumferential fibrous membrane
adverse tissue reactions, infection, and potential loosening in
formation or poor osseous integration of a non-cemented
the later post-operative period (6, 15, 16). Additional 3D MSI
component. On MR imaging, component loosening typically
sequences were developed and are available for such imaging,
manifests as circumferential increased signal intensity at the
includingsliceencodingformetalartifactcorrection(SEMAC)
metallic-boneorcement-boneinterfaceonfat-suppressedtech-
(9, 17), known commercially as advanced WARP (18) and niques (20). The finding of circumferential fibrous membrane
O-MAR XD. formation or osteolysis also indicates potential loosening; this
NOTE 1—Note the improved visualization of synovitis (white arrows)
and the bone-prosthesis interface (black arrow) on the MAVRIC image.
Images courtesy of Dr. Hollis Potter.
FIG. 1 Coronal FSE (left) and MAVRIC (right) Images of a Left
MOM Hip Arthroplasty
F2978 − 20
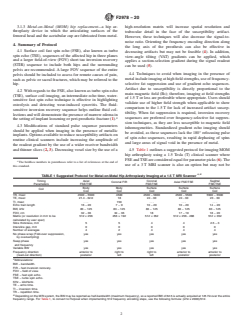
FIG. 2 T1 Weighted TSE (left) and High Bandwidth TSE and VAT (middle), and SEMAC (right) Images of a MOM Hip Arthroplasty
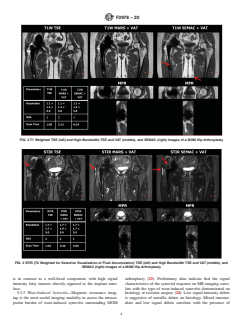
FIG. 3 STIR (T2 Weighted for Selective Visualization of Fluid Accumulation) TSE (left) and High Bandwidth TSE and VAT (middle), and
SEMAC (right) Images of a MOM Hip Arthroplasty
is in contrast to a well-fixed component, with high signal arthroplasty (23). Preliminary data indicate that the signal
intensity fatty marrow directly opposed to the implant inter- characteristics of the synovial response on MR imaging corre-
face. late with the type of wear-induced synovitis demonstrated on
5.3.3 Wear-Induced Synovitis—Magnetic resonance imag- histology at revision surgery (24). Low signal intensity debris
ing is the most useful imaging modality to assess the intraca- is suggestive of metallic debris on histology. Mixed interme-
psular burden of wear-induced synovitis surrounding MOM diate and low signal debris correlate with the presence of
F2978 − 20
NOTE 1—There is focal osteolysis (white arrows) in the greater
trochanter, which manifests as well-demarcated intermediate signal
intensity, similar to that of skeletal muscle, replacing the normal high
signal intensity fatty marrow. Images courtesy of Dr. Hollis Potter.
FIG. 4 Coronal (left) and Axial (right) FSE Images of a Left MOM
Hip Arthroplasty
mixed polymeric (polyethylene and/or polymethyl methacry- wear-induced synovitis, although aspiration is still required for
late) and metallic debris at histology. Magnetic resonance definitive diagnosis (22). The presence of a soft tissue
imaging can demonstrate decompression of synovitis or fluid collection, draining sinus or osteomyelitis further supports the
intoadjacentbursae,suchastheiliopsoasortrochantericbursa, diagnosis of infection on MR imaging.
which can present as soft tissue masses or with secondary 5.3.5 Adverse Local Tissue Re
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2978 − 13 F2978 − 20
Standards Guide to
Optimize Scan Sequences for Clinical Diagnostic Evaluation
of Metal-on-Metal Hip Arthroplasty Devices using Magnetic
Resonance Imaging
This standard is issued under the fixed designation F2978; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide describes the recommended protocol for magnetic resonance imaging (MRI) studies of patients implanted with
metal-on-metal (MOM) devices to determine if the periprosthetic tissues are likely to be associated with an adverse local tissue
reaction (ALTR). Before scanning a patient with a specific implant, the MR practitioner shall confirm that the device is MR
Conditional and that the scan protocol to be used satisfies the conditions for safe scanning for the specific implant. This guide
assumes that the MRI protocol will be applied to MOM devices while they are implanted inside the body. It is also expected that
standardized MRI safety measures will be followed during the performance of this scan protocol.
1.2 This guide covers the clinical evaluation of the tissues surrounding MOM hip replacement devices in patients using MRI. This
guide is applicable to both total and resurfacing MOM hip systems.
1.3 The protocol contained in this guide applies to whole body magnetic resonance equipment, as defined in section
2.2.103201.3.239 of IEC 60601-2-33, Ed. 3.0,3.2, with a whole body radiofrequency (RF) transmit coil as defined in section
2.2.100.201.3.240. The RF coil should have quadrature excitation.circulary polarized RF excitation (also commonly referred to as
quadrature excitation) as defined in section 201.3.249 of IEC 60601-2-33, Ed. 3.2.
1.4 The values stated in SI units are to be regarded as standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of
the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. The user may consider all precautions and warnings provided in the MR system and hip implant labeling
prior to determining the applicability of these protocols.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. The user may consider all precautions and warnings provided in the MR system and hip implant
labeling prior to determining the applicability of these protocols.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
This guide is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee F04.22
on Arthroplasty.
Current edition approved Dec. 1, 2013Sept. 1, 2020. Published May 2014October 2020. Originally approved in 2013. Last previous edition approved in 2013 as
F2978 – 13. DOI: 10.1520/F2978-13.10.1520/F2978-20.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2978 − 20
2. Referenced Documents
2.1 ASTM Standards:
A340 Terminology of Symbols and Definitions Relating to Magnetic Testing
F2503 Practice for Marking Medical Devices and Other Items for Safety in the Magnetic Resonance Environment
2.2 IEC Standard:
IEC 60601-2-33, Ed. 3.0IEC 60601-2-33:2010+AMD1:2013+AMD2:2015 CSV Medical Electrical Equipment—Partelectrical
equipment—Part 2: Particular Requirementsrequirements for the Safety of Magnetic Resonance Equipment for Medical
Diagnosis, 2010basic safety and essential performance of magnetic resonance equipment for medical diagnosis, 2015
3. Terminology
3.1 Definitions—For the purposes of this standard the following definitions shall apply:
3.1.1 Magnetic Resonance Imaging (MRI)—diagnostic imaging technique that uses static and time varying time-varying magnetic
fields to provide tomographic images of tissue by the magnetic resonance of nuclei.
3.1.2 MR - Conditional—MR-Conditional—an item that has been demonstrated to pose no known hazards in a specific MR
environment with specified conditions of use. Field conditions that define the specified MR environment include field strength,
spatial gradient,with demonstrated safety in the MR environment within defined conditions. At a dB/dtminimum, (time rate of
change of the magnetic field), radiofrequency (RF) fields, and specific absorption rate (SAR). address the conditions of the static
magnetic field, the switched gradient magnetic field and the radiofrequency fields. Additional conditions, including specific
configurations of the item, may be required (Practice F2503). – 13).
3.1.3 Metal-on-Metal (MOM) hip replacement—a hip arthroplasty device in which the articulating surfaces of the femoral head
and the acetabular cup are fabricated from metal.
4. Summary of Protocol
4.1 Surface coil fast spin echo (FSE) (FSE), also known as turbo spin echo (TSE), sequences of the affected hip in three planes
and a larger field-of-view (FOV) short tau inversion recovery (STIR) sequence to include both hips and the surrounding pelvis are
recommended. A large FOV sequence of the entire pelvis should be included to assess for remote causes of pain, such as pelvic
or sacral fractures, which may be referred to the hip.
4.2 With regards to the FSE FSE, also known as turbo spin echo (TSE), surface coil imaging, an intermediate echo time,
water-sensitive fast spin echo technique is effective in highlighting osteolysis and detecting wear-induced synovitis. The
fluid-sensitive inversion recovery sequence helps outline fluid collections and will demonstrate the presence of marrow edema in
the setting of implant loosening or peri-prosthetic fracture (1).
4.3 Modifications of standard pulse sequence parameters should be applied when imaging in the presence of metallic implants.
Options available to reduce susceptibility artifacts on routine clinical scanners include increasing the amplitude of the readout
gradient by the use of a wider receiver bandwidth and thinner slices (2, 3). Decreasing voxel size by the use of a high-resolution
matrix will increase spatial resolution and trabecular detail in the face of the susceptibility artifact. However, these techniques will
also decrease the signal-to-noise ratio. Orienting the frequency encoding direction along the long axis of the prosthesis can also
be effective in decreasing artifacts but may not be feasible (4). In addition, view-angle tilting (VAT) gradients can be applied, which
applies a section-selection gradient during the signal readout can be used (5).
4.4 Techniques to avoid when imaging in the presence of metal include imaging at high field strengths, use of frequency-selective
fat suppression and use of gradient echo sequences. Susceptibility artifact Artifact due to susceptibility is directly proportional to
the main magnetic field (B ); therefore, imaging at field strengths greater than 1.5 T should be avoided when possible. of 1.5 T
or less are preferable where appropriate. Users should validate use of higher field strength when applicable to show comparison
to the 1.5 T for lack of increased artifact susceptibility. When fat suppression is required, inversion recovery sequences are
preferred over frequency-selective fat suppression techniques, as they are less susceptible to magnetic field inhomogeneities.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from International Electrotechnical Commission (IEC), 3, rue de Varembé, P.O. Box 131, CH-1211 Geneva 20, Switzerland, http://www.iec.ch.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
F2978 − 20
Standardized gradient echo imaging should be avoided, as these sequences lack the 180° refocusing pulse of spin echo sequences,
resulting in rapid dephasing of spins and large areas of signal void in the presence of metal.
4.5 Table 1 outlines a suggested protocol for imaging MOM hip arthroplasty using a 1.5 Tesla (T) clinical scanner where FSE and
TSE are considered equal for parameter picks (6). The use of a 3 T MRI scanner is also an option but may not be preferable and
the recommendation in 4.4 should be considered. The protocol should be carefully considered for this option before proceeding.
4.6 Multi-acquisition Examples of three-dimensional multispectral imaging (3D-MSI) include multi-acquisition variable-
resonance image combination (MAVRIC SL) is a new technique SL). Fourier transform-based spin-warp (WARP) and metal
artifact reduction for orthopedic implants (O-MAR XD) that results in an image with markedly reducdreduced susceptibility
artifact (7-911). All 3D-MSI techniques utilize a VAT pulse to decrease frequency-encoding distortions and further utilize multiple
frequency bins to mitigate through plane distortions. Early studies have demonstrated decreased image distortion at the
bone-implant interface and improved detection of peri-prosthetic osteolysis and synovitis when compared to conventional fast spin
echo techniques (1012). A recently reported published study of patients with either MOM resurfacing or MOM total hip
arthroplasty demonstrated synovitis using the MAVRIC sequence in 77.4%77.4 % of resurfacing arthroplasty and 86.2%86.2 % of
total hip arthroplasty (THA) hips (1113, 14). For imaging MOM arthroplasty, the use of a MAVRIC sequence is recommended in
at least one plane (coronal or axial) when this sequence is available. When combined with an inversion pulse, they provide optimal
fat suppression in the presence of implants. Specific parameters are listed in Appendix X2. With regards to timing, this protocol
hasthese protocols have been successful in assessing patients for both immediate and delayed complications, including fracture,
nerve impingement, and tendon tears in the immediate postoperativepost-operative period, and adverse tissue reactions, infection,
and potential loosening in the later postoperativepost-operative period (6, 12-1415, 16). Additional prototype sequences are in
development and will become 3D MSI sequences were developed and are available for such imaging, including slice encoding for
metal artifact correction (SEMAC), which is a variant of the VAT principle that adds additional phase-encoding steps in the slice
A,B
TABLE 1 Suggested Protocol for Metal-on-Metal Hip Arthroplasty Imaging at a 1.5 T MRI Scanner
Timing Axial Coronal Sagittal
Coronal FIR Axial FSE
Parameters FSE/TSE FSE/TSE FSE/TSE
Timing Axial Coronal Sagittal
Coronal FIR Axial FSE/TSE
Parameters FSE/TSE FSE/TSE FSE/TSE
Body Body Surface Surface Surface
Coil
Coil Coil Coil Coil Coil
TR, msec 4,500 - 5 500 4,500 4,500 - 5,800 4,500-5,500 5,500-6,500
TR, msec 4500 – 5500 4500 4500 – 5800 4500 – 5500 5500 – 6500
TE, msec 21.4 - 32.0 18 24 - 30 24 - 30 23 - 30
TI, msec . . . 150 . . . . . . . . .
Number of echoes 16 - 20 7 - 9 10 - 20 10 - 20 14 - 20
Echo train length 16 – 20 7 – 9 10 – 20 10 – 20 14 – 20
BW, kHz 83 - 100 83 - 100 83 - 100 83 - 100 83 - 100
BW, kHz 83 – 125 83 – 125 83 – 125 83 – 125 83 – 125
FOV, cm 32 - 36 34 - 36 18 17 - 19 18 - 20
FOV, cm 32 – 36 34 – 36 18 17 – 19 18 – 20
Matrix (or resolution in mm to be 512 × 256 256 × 192 512 × 352 512 × 256 - 288 512 × 352
calculated by user spec)
Slice thickness, mm 5 5 4 4 2.5 - 3
Slice thickness, mm 5 5 4 4 2.5 – 3
Interslice gap, mm 0 0 0 0 0
Number of averages 4 2 4 - 5 4 - 5 4 - 5
Number of averages 4 2 4 4 4
No phase wrap (Fold-over suppression, yes yes yes yes yes
by oversampling)
Swap phase yes yes yes yes yes
and frequency
Variable BW yes yes yes yes yes
Frequency direction anterior to right to right to anterior to anterior to
(read-out direction) posterior left left posterior posterior
A
Abbreviations:
BW – bandwidth.
FIR – fast inversion recovery.
FOV – field of view.
FSE – fast spin echo.
TSE – turbo spin echo.
KHz – kiloHertz.
TE – echo time.
TI – inversion time.
TR – repetition time.
B
Depending Depending on the MRI system, the BW may be reported as half-bandwidth (maximum frequency), so a reported BW of 62.5 is actually acquired at 125 Hz
over the entire frequency range. For Table 1, to convert to Hz/pixel when implementing 512 frequency encoding steps, use the following formula: (kHz x 2000)/512.
F2978 − 20
dimension (SEMAC) (9, 1517). Currently, a commercially available sequence applies the SEMAC principle and is termed the
WARP sequence,), known commercially as advanced WARP (18 which is a high bandwidth protocol that includes the VAT
technique () and O-MAR XD.16).
5. Significance and Use
5.1 Magnetic resonance imaging is ideally suited to image MOM hip arthroplasty due to its superior soft tissue contrast,
multiplanar capabilities and lack of ionizing radiation. MR imaging is the most accurate imaging modality for the assessment of
peri-prosthetic osteolysis and wear-induced synovitis (17-1919, 20).
5.2 Before scanning a patient with a specific implant, the MR practitioner shall confirm that the device is MR Conditional and
that the scan protocol to be used satisfies the conditions for safe scanning for the specific implant.
5.3 This guide can be used to identify the following adverse events.
5.3.1 Osteolysis—Magnetic resonance imaging is superior to conventional radiographs and CT computer tomography (CT) in the
assessment of peri-prosthetic osteolysis and has been shown to be the most accurate method to locate and quantify the extent of
peri-prosthetic osteolysis (1719, 1821). On MR imaging, osteolysis appears as well marginated intraosseous intermediate to
slightly increased signal intensity lesions that contrast with the high signal intensity of the intramedullary fat. A characteristic line
of low signal intensity surrounds the area of focal marrow replacement, distinguishing the appearance of osteolysis from tumoral
replacement of bone or infection (2022).
5.3.2 Component Loosening—While the data are preliminary, MR imaging can identify circumferential bone resorption that may
indicate component loosening. Loosening may result from osteolysis, circumferential fibrous membrane formation or poor osseous
integration of a non-cemented component. On MR imaging, component loosening typically manifests as circumferential increased
signal intensity at the metallic-bone or cement-bone interface on fat-suppressed techniques (1920). The finding of circumferential
fibrous membrane formation or osteolysis also indicates potential loosening; this is in contrast to a well-fixed component, with high
signal intensity fatty marrow directly opposed to the implant interface.
5.3.3 Wear-Induced Synovitis—Magnetic resonance imaging is the most useful imaging modality to assess the intracapsular
burden of wear-induced synovitis surrounding MOM arthroplasty (2123). Preliminary data indicate that the signal characteristics
of the synovial response on MR imaging correlate with the type of wear-induced synovitis demonstrated on histology at revision
surgery (2224). Low signal intensity debris is suggestive of metallic debris on histology. Mixed intermediate and low signal debris
correlatescorrelate with the presence of mixed polymeric (polyethylene and/or polymethyl methacrylate) and metallic debris at
histology. Magnetic resonance imaging can demonstrate decompression of synovitis or fluid into adjacent bursae, such as the
NOTE 1—Note the improved visualization of synovitis (white arrows)
and the bone-prosthesis interface (black arrow) on the MAVRIC image.
Images courtesy of Dr. Hollis Potter.
FIG. 1 Coronal FSE (Left)(left) and MAVRIC (Right)(right) Images
of a Left MOM Hip Arthroplasty
F2978 − 20
FIG. 2 T1 Weighted TSE (left) and High Bandwidth TSE and VAT (middle), and SEMAC (right) Images of a MOM Hip Arthroplasty
FIG. 3 STIR (T2 Weighted for Selective Visualization of Fluid Accumulation) TSE (left) and High Bandwidth TSE and VAT (middle), and
SEMAC (right) Images of a MOM Hip Arthroplasty
iliopsoas or trochanteric bursa, which can present as soft tissue masses or with secondary nerve compression. On occasion,
wear-induced synovitis can result in a chronic indolent pattern of erosion of the surrounding bone, even in the absence of focal
osteolytic lesions (6).
5.3.4 Infection—In the setting of infection, the synovium often demonstrates a hyperintense, lamellated appearance with adjacent
extracapsular soft tissue edema. These appearances help to distinguish the synovial pattern of infection from wear-induced
F2978 − 20
NOTE 1—There is focal osteolysis (white arrows) in the greater
trochanter, which manifests as well-demarcated intermediate signal
intensity, similar to that of skeletal muscle, replacing the normal high
signal intensity fatty marrow. Images courtesy of Dr. Hollis Potter.
FIG. 24 Coronal (Left)(left) and Axial (Right)(right) FSE Images of
a Left MOM Hip Arthroplasty
NOTE 1—Wear-induced synovitis decompresses into the abductor
musculature where there is low signal intensity debris (arrow), consistent
with metallic debris. Images courtesy of Dr. Hollis Potter.
FIG. 35 Axial (Left)(left) and Coronal (Right)(right) FSE Images of
a Left MOM Hip Arthroplasty
synovitis, although aspiration is still required for definitive diagnosis (1422). The presence of a soft tissue collection, drainin
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...